Are you over 18 and want to see adult content?
More Annotations

A complete backup of www.espn.com/soccer/report?gameId=560962
Are you over 18 and want to see adult content?
A complete backup of www.tribunnews.com/nasional/2020/03/02/jangan-panik-kenali-dulu-gejala-cara-mencegah-virus-corona-dari-dema
Are you over 18 and want to see adult content?
A complete backup of nos.nl/l/2325403
Are you over 18 and want to see adult content?

A complete backup of nocutnews.co.kr/news/5299878
Are you over 18 and want to see adult content?
A complete backup of www.sortiraparis.com/loisirs/cinema/articles/205946-papi-sitter-de-philippe-guillard-bande-annonce-critique
Are you over 18 and want to see adult content?
Favourite Annotations
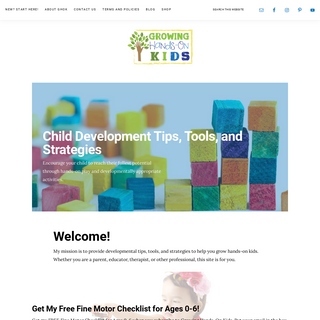
A complete backup of growinghandsonkids.com
Are you over 18 and want to see adult content?

A complete backup of the-north-face.ca
Are you over 18 and want to see adult content?
Text
PATIENT RESOURCES
To request additional information, and for full program terms and conditions, call. 1-888-347-3415. 1-888-347-3415 for more information. For residents of Arkansas, Massachusetts, or OUR FERTILITY OFFERINGS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program. WHO WE ARE – FERRING PHARMACEUTICALS Ferring was born in 1950 in two modest rooms in Malmö, Sweden. Dr Frederik Paulsen, Ferring’s founder, had a breakthrough idea: to create medicines on the body’s own terms, using peptide hormones produced by the pituitary gland to treat a range of health conditions. Today, Ferring is one of the world’s leading pharmaceuticalcompanies
MDR PHARMACEUTICAL CARE × MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients who ROXSAN PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional. ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries andPROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING PHARMACEUTICALS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PATIENT RESOURCES
To request additional information, and for full program terms and conditions, call. 1-888-347-3415. 1-888-347-3415 for more information. For residents of Arkansas, Massachusetts, or OUR FERTILITY OFFERINGS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program. WHO WE ARE – FERRING PHARMACEUTICALS Ferring was born in 1950 in two modest rooms in Malmö, Sweden. Dr Frederik Paulsen, Ferring’s founder, had a breakthrough idea: to create medicines on the body’s own terms, using peptide hormones produced by the pituitary gland to treat a range of health conditions. Today, Ferring is one of the world’s leading pharmaceuticalcompanies
MDR PHARMACEUTICAL CARE × MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients who ROXSAN PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional. ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries andPROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING PHARMACEUTICALS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PATIENT RESOURCES
To request additional information, and for full program terms and conditions, call. 1-888-347-3415. 1-888-347-3415 for more information. For residents of Arkansas, Massachusetts, or OUR FERTILITY OFFERINGS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program. WHO WE ARE – FERRING PHARMACEUTICALS Ferring was born in 1950 in two modest rooms in Malmö, Sweden. Dr Frederik Paulsen, Ferring’s founder, had a breakthrough idea: to create medicines on the body’s own terms, using peptide hormones produced by the pituitary gland to treat a range of health conditions. Today, Ferring is one of the world’s leading pharmaceuticalcompanies
MDR PHARMACEUTICAL CARE × MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients who ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries andPROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) ROXSAN PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional. WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING PHARMACEUTICALS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PATIENT RESOURCES
To request additional information, and for full program terms and conditions, call. 1-888-347-3415. 1-888-347-3415 for more information. For residents of Arkansas, Massachusetts, or OUR FERTILITY OFFERINGS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) WHO WE ARE – FERRING PHARMACEUTICALS Ferring was born in 1950 in two modest rooms in Malmö, Sweden. Dr Frederik Paulsen, Ferring’s founder, had a breakthrough idea: to create medicines on the body’s own terms, using peptide hormones produced by the pituitary gland to treat a range of health conditions. Today, Ferring is one of the world’s leading pharmaceuticalcompanies
PROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients who ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries and CONCORD PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional. WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING PHARMACEUTICALS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PATIENT RESOURCES
To request additional information, and for full program terms and conditions, call. 1-888-347-3415. 1-888-347-3415 for more information. For residents of Arkansas, Massachusetts, or OUR FERTILITY OFFERINGS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) WHO WE ARE – FERRING PHARMACEUTICALS Ferring was born in 1950 in two modest rooms in Malmö, Sweden. Dr Frederik Paulsen, Ferring’s founder, had a breakthrough idea: to create medicines on the body’s own terms, using peptide hormones produced by the pituitary gland to treat a range of health conditions. Today, Ferring is one of the world’s leading pharmaceuticalcompanies
PROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients who ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries and CONCORD PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional. WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING PHARMACEUTICALS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PATIENT RESOURCES
To request additional information, and for full program terms and conditions, call. 1-888-347-3415. 1-888-347-3415 for more information. For residents of Arkansas, Massachusetts, or OUR FERTILITY OFFERINGS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program. WHO WE ARE – FERRING PHARMACEUTICALS Ferring was born in 1950 in two modest rooms in Malmö, Sweden. Dr Frederik Paulsen, Ferring’s founder, had a breakthrough idea: to create medicines on the body’s own terms, using peptide hormones produced by the pituitary gland to treat a range of health conditions. Today, Ferring is one of the world’s leading pharmaceuticalcompanies
PROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries andPROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients who CONCORD PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional. WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING PHARMACEUTICALS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PATIENT RESOURCES
oneheart For patients who have been prescribed select Ferring fertility medications. Cash-paying and commercially insured patients may be eligible to receive a $200 rebate for out-of-pocket costs for purchases of select Ferring fertility medications made after June 1,2020.
OUR FERTILITY OFFERINGS Our Fertility Offerings: With the most complete reproductive medicine portfolio in the US, our products span the IVF journey, from stimulation to implantation. WHO WE ARE – FERRING PHARMACEUTICALS Who We Are: Here at Ferring, we believe that family building is a right—even in the face of biological barriers. And we’re committed to breaking down those barriers through science, innovation, andmedicine.
ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries andPROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients who ROXSAN PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional.PROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING REPRODUCTIVE HEALTH® Ferring Reproductive Health® FERRING PHARMACEUTICALS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PATIENT RESOURCES
oneheart For patients who have been prescribed select Ferring fertility medications. Cash-paying and commercially insured patients may be eligible to receive a $200 rebate for out-of-pocket costs for purchases of select Ferring fertility medications made after June 1,2020.
OUR FERTILITY OFFERINGS Our Fertility Offerings: With the most complete reproductive medicine portfolio in the US, our products span the IVF journey, from stimulation to implantation. WHO WE ARE – FERRING PHARMACEUTICALS Who We Are: Here at Ferring, we believe that family building is a right—even in the face of biological barriers. And we’re committed to breaking down those barriers through science, innovation, andmedicine.
ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries andPROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients who ROXSAN PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional.PROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING REPRODUCTIVE HEALTH® Ferring Reproductive Health® FERRING PHARMACEUTICALS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program.PATIENT RESOURCES
oneheart For patients who have been prescribed select Ferring fertility medications. Cash-paying and commercially insured patients may be eligible to receive a $200 rebate for out-of-pocket costs for purchases of select Ferring fertility medications made after June 1,2020.
OUR FERTILITY OFFERINGS MENOPUR® (menotropins for injection) INDICATION FOR USE. MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program. WHO WE ARE – FERRING PHARMACEUTICALS Ferring was born in 1950 in two modest rooms in Malmö, Sweden. Dr Frederik Paulsen, Ferring’s founder, had a breakthrough idea: to create medicines on the body’s own terms, using peptide hormones produced by the pituitary gland to treat a range of health conditions. ABOUT FERTILITY TESTING Fertility testing for women Ovarian reserve testing Ovarian reserve testing is performed to help determine the potential number of eggs remaining in the ovaries andPROGRAM APPLICATION
Ferring’s OneHeart Program (the “Program”) offers up to a $200 rebate for out-of-pocket cost incurred by eligible cash-paying or commercially insured patients whoPROGRAM APPLICATION
The HeartTomorrow Program offers MENOPUR for $68/vial (the “Price”) for eligible cash paying patients who are undergoing a controlled ovarian stimulation (“COS”) ROXSAN PHARMACY ARCHIVES The content on this website is for informational purposes only and is not intended to replace a discussion with a healthcare professional. WWW.FERRINGFERTILITY.COM 301 Moved Permanently. nginx FERRING REPRODUCTIVE HEALTH® Ferring Reproductive Health®Skip to content
* For US Healthcare Professionals * Important Safety Information*
MENOPUR®
(menotropins for injection)*
GANIRELIX ACETATE
Injection 250 mcg/0.5 mL*
NOVAREL®
(chorionic gonadotropin for injection, USP)*
INVOCELL®
Intravaginal Culture System*
ENDOMETRIN®
(progesterone) Vaginal Insert 100 mg * Prescribing Information*
MENOPUR®
(menotropins for injection)*
GANIRELIX ACETATE
Injection 250 mcg/0.5 mL*
NOVAREL®
(chorionic gonadotropin for injection, USP)*
INVOCELL®
Intravaginal Culture System*
ENDOMETRIN®
(progesterone) Vaginal Insert 100 mgMenu
* Home
* Who We Are
* Our Fertility Offerings* Patient Resources
Committed to Science. HELPING WISHES COME TRUE.Get to know us >
Stay up to date on the latest news and research from Ferring Reproductive Medicine and Maternal Health.Sign Up
×
LET’S STAY IN TOUCH* *Required
* Email Address*
* First Name*
* Last Name*
* I am a:
* Fertility Specialist* Nurse
* Other
* Please Specify:
Submit
WHO WE ARE
Get to know us, our legacy, and what we stand for.Learn More >
OUR FERTILITY OFFERINGS With the most complete reproductive medicine portfolio in the US, our products span the IVF journey, from stimulation to implantation.See Products >
PATIENT RESOURCES
Get educational materials and financial assistance information for patients undergoing fertility treatment.Get Support >
WHAT’S NEW?
Read the latest news from Ferring Reproductive Medicine and MaternalHealth. >
* FerringUSA.com
* Terms & Conditions* Privacy Policy
* Contact Us
FERRING, the FERRING PHARMACEUTICALS logo, and other Ferring product trade names are registered trademarks of Ferring B.V. save where indicated otherwise. INVOCELL is a registered trademark of INVOBioscience.
04/21 US-RMMH-2000068 v.4 © 2021 Ferring B.V.×
MENOPUR® (MENOTROPINS FOR INJECTION)INDICATION FOR USE
* MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program. IMPORTANT SAFETY INFORMATION * MENOPUR is contraindicated in women who have: a high FSH level indicating primary ovarian failure, presence of uncontrolled non-gonadal endocrinopathies, tumors of the pituitary gland or hypothalamus, sex hormone dependent tumors of the reproductive tract and accessory organs, abnormal uterine bleeding of undetermined origin, ovarian cysts or enlargement of undetermined origin, not due to polycystic ovary syndrome, or prior hypersensitivity to menotropins or MENOPUR. MENOPUR is not indicated in women who are pregnant and may cause fetal harm when administered to a pregnant woman * MENOPUR should only be used by physicians who are thoroughly familiar with infertility problems. MENOPUR is a potent gonadotropic substance capable of causing Ovarian Hyperstimulation Syndrome (OHSS), with or without pulmonary or vascular complications, in women undergoing therapy for infertility. Ovarian torsion has been reported after gonadotropin treatment. Serious pulmonary conditions and thromboembolic events have been reported with MENOPUR. There have been infrequent reports of ovarian neoplasms with MENOPUR. Multiple pregnancies, spontaneous abortion, congenital malformations and ectopic pregnancies have occurred following treatment with MENOPUR. * The most common adverse reactions (≥2%) in ART include: abdominal cramps; abdomen enlarged; abdominal pain; headache; injection site pain and reaction; injection site inflammation; OHSS.×
PRINTABLE INJECTION GUIDE A PDF of step-by-step instructions for preparing and injecting MENOPUR is available to download or print in the languages listed below. Select a Language English Spanish Mandarin Japanese KoreanDownload >
MENOPUR® (MENOTROPINS FOR INJECTION)INDICATION FOR USE
* MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program. IMPORTANT SAFETY INFORMATION * MENOPUR is contraindicated in women who have: a high FSH level indicating primary ovarian failure, presence of uncontrolled non-gonadal endocrinopathies, tumors of the pituitary gland or hypothalamus, sex hormone dependent tumors of the reproductive tract and accessory organs, abnormal uterine bleeding of undetermined origin, ovarian cysts or enlargement of undetermined origin, not due to polycystic ovary syndrome, or prior hypersensitivity to menotropins or MENOPUR. MENOPUR is not indicated in women who are pregnant and may cause fetal harm when administered to a pregnant woman * MENOPUR should only be used by physicians who are thoroughly familiar with infertility problems. MENOPUR is a potent gonadotropic substance capable of causing Ovarian Hyperstimulation Syndrome (OHSS), with or without pulmonary or vascular complications, in women undergoing therapy for infertility. Ovarian torsion has been reported after gonadotropin treatment. Serious pulmonary conditions and thromboembolic events have been reported with MENOPUR. There have been infrequent reports of ovarian neoplasms with MENOPUR. Multiple pregnancies, spontaneous abortion, congenital malformations and ectopic pregnancies have occurred following treatment with MENOPUR. * The most common adverse reactions (≥2%) in ART include: abdominal cramps; abdomen enlarged; abdominal pain; headache; injection site pain and reaction; injection site inflammation; OHSS.×
MENOPUR® (MENOTROPINS FOR INJECTION)INDICATION FOR USE
* MENOPUR® (menotropins for injection), administered subcutaneously, is indicated for the development of multiple follicles and pregnancy in the ovulatory patients participating in an Assisted Reproductive Technology (ART) program. IMPORTANT SAFETY INFORMATION * MENOPUR is contraindicated in women who have: a high FSH level indicating primary ovarian failure, presence of uncontrolled non-gonadal endocrinopathies, tumors of the pituitary gland or hypothalamus, sex hormone dependent tumors of the reproductive tract and accessory organs, abnormal uterine bleeding of undetermined origin, ovarian cysts or enlargement of undetermined origin, not due to polycystic ovary syndrome, or prior hypersensitivity to menotropins or MENOPUR. MENOPUR is not indicated in women who are pregnant and may cause fetal harm when administered to a pregnant woman * MENOPUR should only be used by physicians who are thoroughly familiar with infertility problems. MENOPUR is a potent gonadotropic substance capable of causing Ovarian Hyperstimulation Syndrome (OHSS), with or without pulmonary or vascular complications, in women undergoing therapy for infertility. Ovarian torsion has been reported after gonadotropin treatment. Serious pulmonary conditions and thromboembolic events have been reported with MENOPUR. There have been infrequent reports of ovarian neoplasms with MENOPUR. Multiple pregnancies, spontaneous abortion, congenital malformations and ectopic pregnancies have occurred following treatment with MENOPUR. * The most common adverse reactions (≥2%) in ART include: abdominal cramps; abdomen enlarged; abdominal pain; headache; injection site pain and reaction; injection site inflammation; OHSS.×
GANIRELIX ACETATE INJECTIONINDICATION FOR USE
* Ganirelix acetate injection is indicated for the inhibition of premature LH surges in women undergoing controlled ovarianhyperstimulation.
IMPORTANT SAFETY INFORMATION * Ganirelix acetate injection is contraindicated under the following conditions: Known hypersensitivity to ganirelix acetate or to any of its components, Known hypersensitivity to GnRH or any other GnRH analog, Known or suspected pregnancy. * Ganirelix acetate injection should be prescribed by physicians who are experienced in infertility treatment. Before starting treatment with ganirelix acetate, pregnancy must be excluded. Safe use of ganirelix acetate during pregnancy has not been established. * Special care should be taken in women with signs and symptoms of active allergic conditions. * Cases of hypersensitivity reactions, including anaphylactoid reactions, have been reported, as early as with the first dose, during postmarketing surveillance. In the absence of clinical experience, ganirelix acetate treatment is not advised in women with severe allergic conditions. * The packaging of this product contains natural rubber latex which may cause allergic reactions. * Most commonly reported adverse reactions (≥ 1%) reported in clinical trials were: Abdominal Pain (gynecological), Death Fetal, Headache, Ovarian Hyperstimulation Syndrome, Vaginal Bleeding, Injection Site Reaction, Nausea, Abdominal Pain (gastrointestinal). * During post marketing surveillance, rare cases of hypersensitivity reactions, including anaphylactoid reactions, have been reported, as early as with the first dose. * Major and minor congenital abnormalities have been reported in clinical follow-up studies of newborns of women administered ganirelix acetate injection. The causal relationship between these congenital anomalies and ganirelix acetate is unknown.×
NOVAREL®
(CHORIONIC GONADOTROPIN FOR INJECTION, USP)INDICATION FOR USE
* NOVAREL® is indicated for the induction of ovulation and pregnancy in the anovulatory, infertile woman in whom the cause of anovulation is secondary and not due to primary ovarian failure, and who has been appropriately pre-treated with human menotropins. IMPORTANT SAFETY INFORMATION * NOVAREL should not be used in patients with prior allergic reaction to HCG. HCG may cause fetal harm when administered to apregnant woman.
* NOVAREL should be used in conjunction with human menopausal gonadotropins only by physicians experienced with infertility problems who are familiar with the criteria for patient selection, contraindications, warnings, precautions, and adverse reactions described in the package insert for menotropins. The principal serious adverse reactions during this use are: (1) Ovarian hyperstimulation, a syndrome of sudden ovarian enlargement, ascites with or without pain, and/or pleural effusion; (2) Enlargement of preexisting ovarian cysts or rupture of ovarian cysts with resultant hemoperitoneum; (3) Multiple births, and (4) Arterial thromboembolism. * Anaphylaxis has been reported with urinary-derived HCG products. * Since androgens may cause fluid retention, HCG should be used with caution in patients with cardiac or renal disease, epilepsy, migraine,or asthma.
* HCG can cross react in the radioimmunoassay of gonadotropins, especially luteinizing hormone. Each individual laboratory should establish the degree of cross reactivity with their gonadotropin assay. Physicians should make the laboratory aware of patients on HCG if gonadotropin levels are requested. * Defects of forelimbs and of the central nervous system, as well as alterations in sex ratio, have been reported in mice on combined gonadotropin and HCG regimens. The dose of gonadotropin used was intended to induce superovulation. No mutagenic effect has been clearly established in humans. * It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk caution should be exercised when HCG is administered to a nursing woman. * Adverse reactions include headache, irritability, restlessness, depression, fatigue, edema, precocious puberty, gynecomastia, pain at the site of injection. Hypersensitivity reactions both localized and systemic in nature, including erythema, urticaria, rash, angioedema, dyspnea and shortness of breath, have been reported. The relationship of these allergic-like events to the polypeptide hormone or the diluent containing benzyl alcohol is not clear.×
PRINTABLE INJECTION GUIDE A PDF of step-by-step instructions for preparing and injecting NOVAREL is available to download or print in the languages below. Select a Language EnglishDownload >
NOVAREL®
(CHORIONIC GONADOTROPIN FOR INJECTION, USP)INDICATION FOR USE
* NOVAREL® is indicated for the induction of ovulation and pregnancy in the anovulatory, infertile woman in whom the cause of anovulation is secondary and not due to primary ovarian failure, and who has been appropriately pre-treated with human menotropins. IMPORTANT SAFETY INFORMATION * NOVAREL should not be used in patients with prior allergic reaction to HCG. HCG may cause fetal harm when administered to apregnant woman.
* NOVAREL should be used in conjunction with human menopausal gonadotropins only by physicians experienced with infertility problems who are familiar with the criteria for patient selection, contraindications, warnings, precautions, and adverse reactions described in the package insert for menotropins. The principal serious adverse reactions during this use are: (1) Ovarian hyperstimulation, a syndrome of sudden ovarian enlargement, ascites with or without pain, and/or pleural effusion; (2) Enlargement of preexisting ovarian cysts or rupture of ovarian cysts with resultant hemoperitoneum; (3) Multiple births, and (4) Arterial thromboembolism. * Anaphylaxis has been reported with urinary-derived HCG products. * Since androgens may cause fluid retention, HCG should be used with caution in patients with cardiac or renal disease, epilepsy, migraine,or asthma.
* HCG can cross react in the radioimmunoassay of gonadotropins, especially luteinizing hormone. Each individual laboratory should establish the degree of cross reactivity with their gonadotropin assay. Physicians should make the laboratory aware of patients on HCG if gonadotropin levels are requested. * Defects of forelimbs and of the central nervous system, as well as alterations in sex ratio, have been reported in mice on combined gonadotropin and HCG regimens. The dose of gonadotropin used was intended to induce superovulation. No mutagenic effect has been clearly established in humans. * It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk caution should be exercised when HCG is administered to a nursing woman. * Adverse reactions include headache, irritability, restlessness, depression, fatigue, edema, precocious puberty, gynecomastia, pain at the site of injection. Hypersensitivity reactions both localized and systemic in nature, including erythema, urticaria, rash, angioedema, dyspnea and shortness of breath, have been reported. The relationship of these allergic-like events to the polypeptide hormone or the diluent containing benzyl alcohol is not clear.×
5,000 USP Units 10,000 USP UnitsNOVAREL®
(CHORIONIC GONADOTROPIN FOR INJECTION, USP)INDICATION FOR USE
* NOVAREL® is indicated for the induction of ovulation and pregnancy in the anovulatory, infertile woman in whom the cause of anovulation is secondary and not due to primary ovarian failure, and who has been appropriately pre-treated with human menotropins. IMPORTANT SAFETY INFORMATION * NOVAREL should not be used in patients with prior allergic reaction to HCG. HCG may cause fetal harm when administered to apregnant woman.
* NOVAREL should be used in conjunction with human menopausal gonadotropins only by physicians experienced with infertility problems who are familiar with the criteria for patient selection, contraindications, warnings, precautions, and adverse reactions described in the package insert for menotropins. The principal serious adverse reactions during this use are: (1) Ovarian hyperstimulation, a syndrome of sudden ovarian enlargement, ascites with or without pain, and/or pleural effusion; (2) Enlargement of preexisting ovarian cysts or rupture of ovarian cysts with resultant hemoperitoneum; (3) Multiple births, and (4) Arterial thromboembolism. * Anaphylaxis has been reported with urinary-derived HCG products. * Since androgens may cause fluid retention, HCG should be used with caution in patients with cardiac or renal disease, epilepsy, migraine,or asthma.
* HCG can cross react in the radioimmunoassay of gonadotropins, especially luteinizing hormone. Each individual laboratory should establish the degree of cross reactivity with their gonadotropin assay. Physicians should make the laboratory aware of patients on HCG if gonadotropin levels are requested. * Defects of forelimbs and of the central nervous system, as well as alterations in sex ratio, have been reported in mice on combined gonadotropin and HCG regimens. The dose of gonadotropin used was intended to induce superovulation. No mutagenic effect has been clearly established in humans. * It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk caution should be exercised when HCG is administered to a nursing woman. * Adverse reactions include headache, irritability, restlessness, depression, fatigue, edema, precocious puberty, gynecomastia, pain at the site of injection. Hypersensitivity reactions both localized and systemic in nature, including erythema, urticaria, rash, angioedema, dyspnea and shortness of breath, have been reported. The relationship of these allergic-like events to the polypeptide hormone or the diluent containing benzyl alcohol is not clear.×
INVOCELL® INTRAVAGINAL CULTURE SYSTEMINDICATIONS FOR USE
INVOCELL® Intravaginal Culture System consists of the INVOCELL Culture Device and the INVOCELL Retention Device. The Culture Device is indicated for use in preparing, holding, and transferring human gametes or embryos during In Vitro Fertilization/Intravaginal Culture (IVF/IVC) and Intra-Cytoplasmic Sperm Injection Fertilization/ Intravaginal Culture (ICSI/IVC)procedures.
The Retention Device is indicated for use during the incubation period to aid in retention of the Culture Device in the vaginal cavity. The Culture and Retention Devices are not indicated for incubation periods exceeding 72 hours. SELECT IMPORTANT SAFETY INFORMATION * Culture and Retention Devices should not be used in patients with the inability to tolerate placement or wearing of a device within thevaginal cavity.
* Culture and Retention Devices are single use only. Do not use if product or package appears damaged. * Do not use Culture Device in patients with hypersensitivity to medical grade silicone or polystyrene or in patients with a severe case of vaginitis or with a history of toxic shock syndrome. Evaluate patients for any recent pelvic surgery to assure it will not affect intravaginal culture procedure. * Proper handling is extremely important to safe and effective use of Culture Device. Do not begin clinical use of INVOCELL Intravaginal Culture System without establishing competency by reading and practicing Instructions for Use. * It is recommended that INVOCELL Intravaginal Culture System be utilized with a mild ovarian stimulation protocol. The recommended upper limit on number of oocytes or ICSI fertilized embryos to be placed in Culture Device is seven. * Verify Culture Device is correctly locked before placement invaginal cavity.
* Patients should avoid any activity that may alter temperature of vaginal cavity and should avoid manipulation and removal of Culture and Retention Devices while in place.×
ENDOMETRIN®
(PROGESTERONE) VAGINAL INSERTINDICATION FOR USE
* ENDOMETRIN® (progesterone) Vaginal Insert is indicated to support embryo implantation and early pregnancy by supplementation of corpus luteal function as part of an Assisted Reproductive Technology (ART) treatment program for infertile women. IMPORTANT SAFETY INFORMATION * ENDOMETRIN should not be used in individuals with any of the following conditions: previous allergic reactions to progesterone or any of the ingredients of ENDOMETRIN, undiagnosed vaginal bleeding, known missed abortion or ectopic pregnancy, liver disease, known or suspected breast cancer, active arterial or venous thromboembolism or severe thrombophlebitis, or a history of these events. * The physician should be alert to earliest signs of myocardial infarction, cerebrovascular disorders, arterial or venous thromboembolism (venous thromboembolism or pulmonary embolism), thrombophlebitis, or retinal thrombosis. ENDOMETRIN should be discontinued if any of these are suspected. * Patients with a history of depression need to be closely observed. Consider discontinuation if symptoms worsen. * ENDOMETRIN should not be recommended for use with other vaginal products (such as antifungal products) as this may alter progesterone release and absorption from the vaginal insert. * The most common adverse reactions reported (greater than 5%) were post-oocyte retrieval pain, abdominal pain, nausea, and ovarian hyperstimulation syndrome.×
PRINTABLE ADMINISTRATION GUIDE PDF of step-by-step instructions for administering ENDOMETRIN. Available to download or print in the languages below. Select a Language English Spanish Simplified Chinese HindiDownload >
ENDOMETRIN®
(PROGESTERONE) VAGINAL INSERTINDICATION FOR USE
* ENDOMETRIN® (progesterone) Vaginal Insert is indicated to support embryo implantation and early pregnancy by supplementation of corpus luteal function as part of an Assisted Reproductive Technology (ART) treatment program for infertile women. IMPORTANT SAFETY INFORMATION * ENDOMETRIN should not be used in individuals with any of the following conditions: previous allergic reactions to progesterone or any of the ingredients of ENDOMETRIN, undiagnosed vaginal bleeding, known missed abortion or ectopic pregnancy, liver disease, known or suspected breast cancer, active arterial or venous thromboembolism or severe thrombophlebitis, or a history of these events. * The physician should be alert to earliest signs of myocardial infarction, cerebrovascular disorders, arterial or venous thromboembolism (venous thromboembolism or pulmonary embolism), thrombophlebitis, or retinal thrombosis. ENDOMETRIN should be discontinued if any of these are suspected. * Patients with a history of depression need to be closely observed. Consider discontinuation if symptoms worsen. * ENDOMETRIN should not be recommended for use with other vaginal products (such as antifungal products) as this may alter progesterone release and absorption from the vaginal insert. * The most common adverse reactions reported (greater than 5%) were post-oocyte retrieval pain, abdominal pain, nausea, and ovarian hyperstimulation syndrome.Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0