Are you over 18 and want to see adult content?
More Annotations

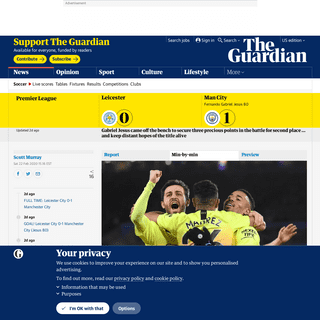
A complete backup of www.labaroviola.com/fiorentina-milan-gli-ultimi-cinque-precedenti-tra-i-due-club-in-serie-a-due-le-vittorie
Are you over 18 and want to see adult content?
Favourite Annotations

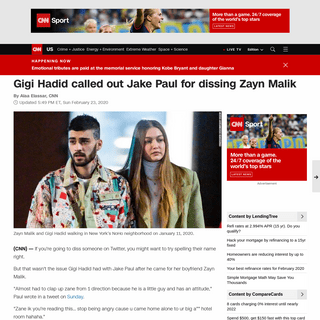
A complete backup of https://daysofadomesticdad.com/a-dads-moving-guide/
Are you over 18 and want to see adult content?
A complete backup of https://mashable.com/category/homemade-movies/
Are you over 18 and want to see adult content?
A complete backup of https://clean0074.pixnet.net/blog/post/29171090
Are you over 18 and want to see adult content?

A complete backup of https://tl.rulate.ru/book/39160/
Are you over 18 and want to see adult content?
A complete backup of https://cersilindonesia.wordpress.com/Deb-33/
Are you over 18 and want to see adult content?
Text
pumps.
ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks.XERIS PRODUCTS
XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US Progress for severe hypoglycemia and epilepsy are just the start. While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, andpumps.
ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
XERIS PRODUCTS
NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks. NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks. PRESS RELEASES FROM XERIS PHARMACEUTICALS Xeris Pharmaceuticals to Webcast Upcoming Presentations. 5/6/2021. Xeris Pharmaceuticals to Announce First Quarter 2021 Financial Results and Host Conference Call and Webcast on May 13, 2021. 5/6/2021. Xeris Pharmaceuticals Announces Extension of the Interest-Only Period of Its Debt Facility With Oxford Finance and Silicon Valley Bank. 4/29/2021. XERIS PHARMACEUTICALS' XERISOL™ & XERIJECT™ FORMULATION XeriSol ™ and XeriJect ™ provide distinct advantages over existing formulations. Both XeriSol and XeriJect offer the opportunity to: Eliminate reconstitution. Enable long-term room temperature stability. Significantly reduce injection volume. Eliminate the requirement for intravenous (IV) infusion. These attributes can lead to products that XERIS FINANCIAL INFORMATION Financials. Find relevant financial information about Xeris through our annual reports and proxy statements. Annual Reports. Proxy Statements. Select Section Annual Reports Proxy Statements. Never miss a financial update by signing up for alerts.XERIS PIPELINE
Xeris has a robust pipeline across its ready-to-use portfolios. Through a series of grant-supported collaborations, Xeris is exploring additional uses for ready-to-use glucagon in mild-to-moderate hypoglycemia. Additionally, Xeris is evaluating stable, liquid glucagon as a combination therapy inside a physiological, closed-loop,bi-hormonal
CONTACT US - XERIS PHARMACEUTICALS, INC. Contact Us. Please contact the Xeris Contact Center at 1-877-XERIS-37 to submit a medical/product information question and/or to report an adverse event, and/or product complaint. By entering your email address, you agree to receive emails from Xeris (including promotionaloffers and
XERIS RESEARCH AND DEVELOPMENT Xeris is developing its pipeline both through internal clinical development and external partnerships to advance the commercial potential of its products globally. Partner with us.SEC FILINGS
SEC Filings. Group: CHOOSE FROM LIST 3,4,5 Annual Filings Current Reports Mergers & Acquisitions Other Proxy Filings Quarterly Filings Registration Statements. Filing Year: - ANY - 2021 2020. Filing date. Form. Description. Filing Group. PARTNERING WITH XERIS PHARMACEUTICALS Partnering. Xeris is interested in establishing a portfolio of strategic collaborations with key partners who wish to codevelop products using Xeris’ platform technologies (XeriSol ™ and XeriJect ™ ). Currently, Xeris is working on a number of formulation feasibility programs in the areas of peptides, vaccines, and monoclonal antibodies. FINANCIAL CONFLICT OF INTEREST POLICY Financial conflict of interest (FCOI) means a significant financial interest that could directly and significantly affect the design, conduct, or reporting of PHS-funded research. 3.4. FCOI report means an Institution's report of a financial conflict of interest to a PHS Awarding Component. 3.5. XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US Progress for severe hypoglycemia and epilepsy are just the start. While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, andpumps.
ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks.XERIS PRODUCTS
XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US Progress for severe hypoglycemia and epilepsy are just the start. While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, andpumps.
ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks.XERIS PRODUCTS
NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks. PRESS RELEASES FROM XERIS PHARMACEUTICALS Xeris Pharmaceuticals to Webcast Upcoming Presentations. 5/6/2021. Xeris Pharmaceuticals to Announce First Quarter 2021 Financial Results and Host Conference Call and Webcast on May 13, 2021. 5/6/2021. Xeris Pharmaceuticals Announces Extension of the Interest-Only Period of Its Debt Facility With Oxford Finance and Silicon Valley Bank. 4/29/2021. XERIS PHARMACEUTICALS' XERISOL™ & XERIJECT™ FORMULATION XeriSol ™ and XeriJect ™ provide distinct advantages over existing formulations. Both XeriSol and XeriJect offer the opportunity to: Eliminate reconstitution. Enable long-term room temperature stability. Significantly reduce injection volume. Eliminate the requirement for intravenous (IV) infusion. These attributes can lead to products that XERIS FINANCIAL INFORMATION Financials. Find relevant financial information about Xeris through our annual reports and proxy statements. Annual Reports. Proxy Statements. Select Section Annual Reports Proxy Statements. Never miss a financial update by signing up for alerts.XERIS PIPELINE
Xeris has a robust pipeline across its ready-to-use portfolios. Through a series of grant-supported collaborations, Xeris is exploring additional uses for ready-to-use glucagon in mild-to-moderate hypoglycemia. Additionally, Xeris is evaluating stable, liquid glucagon as a combination therapy inside a physiological, closed-loop,bi-hormonal
CONTACT US - XERIS PHARMACEUTICALS, INC. Contact Us. Please contact the Xeris Contact Center at 1-877-XERIS-37 to submit a medical/product information question and/or to report an adverse event, and/or product complaint. By entering your email address, you agree to receive emails from Xeris (including promotionaloffers and
XERIS RESEARCH AND DEVELOPMENT Xeris is developing its pipeline both through internal clinical development and external partnerships to advance the commercial potential of its products globally. Partner with us.SEC FILINGS
SEC Filings. Group: CHOOSE FROM LIST 3,4,5 Annual Filings Current Reports Mergers & Acquisitions Other Proxy Filings Quarterly Filings Registration Statements. Filing Year: - ANY - 2021 2020. Filing date. Form. Description. Filing Group. PARTNERING WITH XERIS PHARMACEUTICALS Partnering. Xeris is interested in establishing a portfolio of strategic collaborations with key partners who wish to codevelop products using Xeris’ platform technologies (XeriSol ™ and XeriJect ™ ). Currently, Xeris is working on a number of formulation feasibility programs in the areas of peptides, vaccines, and monoclonal antibodies. FINANCIAL CONFLICT OF INTEREST POLICY Financial conflict of interest (FCOI) means a significant financial interest that could directly and significantly affect the design, conduct, or reporting of PHS-funded research. 3.4. FCOI report means an Institution's report of a financial conflict of interest to a PHS Awarding Component. 3.5. XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, and pumps. Uncover what tomorrow brings. ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
XERIS LEADERSHIP
Paul R. Edick. Chairman & Chief Executive Officer. Mr. Edick joined Xeris Pharmaceuticals as Chairman and Chief Executive Officer in January 2017. Prior to Xeris, Mr. Edick was Founding Partner of 3G Advisors, a consultancy to the pharmaceutical, healthcare, and healthcare investor communities. From 2010 to 2014. Paul R. Edick. XERIS PHARMACEUTICALS ANNOUNCES POSITIVE FINDINGS FROM THE Rebound hypoglycemia observed in the placebo arm with oral glucose tablet use; No rebound hypoglycemia observed in the RTU glucagon treatment arm CHICAGO --(BUSINESS WIRE)--May 22, 2020-- Xeris Pharmaceuticals, Inc. (Nasdaq: XERS), a specialtyXERIS PIPELINE
Xeris has a robust pipeline across its ready-to-use portfolios. Through a series of grant-supported collaborations, Xeris is exploring additional uses for ready-to-use glucagon in mild-to-moderate hypoglycemia. Additionally, Xeris is evaluating stable, liquid glucagon as a combination therapy inside a physiological, closed-loop,bi-hormonal
LOW BLOOD SUGAR TREATMENT WITH INSULIN PRAMLINTIDE Pramlintide is the drug that resembles amylin, a human hormone that slows the absorption of carbohydrates through its effects on stomach emptying. Pramlintide administration mimics the human body's natural interaction between insulin and amylin to regulate normal blood sugar levels with meals. Xeris is currently developing a ready-to-use XERIS PHARMACEUTICALS ANNOUNCES POSITIVE TOPLINE RESULTS All subjects successfully self-administered a mini dose of RTU glucagon Rebound hypoglycemia observed more frequently in the placebo arm and with oral glucose tablet use Subcutaneous RTU glucagon is safe and well-tolerated CHICAGO --(BUSINESS WIRE)--Dec. 10, 2019-- XERIS PHARMACEUTICALS GRANTED FAST TRACK DESIGNATION BY Xeris Pharmaceuticals Granted Fast Track Designation by the FDA for Its Novel Concentrated Diazepam Formulation (XP-0863) For InjectionOctober 20, 2020
XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, and pumps. Uncover what tomorrow brings. ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
XERIS LEADERSHIP
Paul R. Edick. Chairman & Chief Executive Officer. Mr. Edick joined Xeris Pharmaceuticals as Chairman and Chief Executive Officer in January 2017. Prior to Xeris, Mr. Edick was Founding Partner of 3G Advisors, a consultancy to the pharmaceutical, healthcare, and healthcare investor communities. From 2010 to 2014. Paul R. Edick. XERIS PHARMACEUTICALS ANNOUNCES POSITIVE FINDINGS FROM THE Rebound hypoglycemia observed in the placebo arm with oral glucose tablet use; No rebound hypoglycemia observed in the RTU glucagon treatment arm CHICAGO --(BUSINESS WIRE)--May 22, 2020-- Xeris Pharmaceuticals, Inc. (Nasdaq: XERS), a specialtyXERIS PIPELINE
Xeris has a robust pipeline across its ready-to-use portfolios. Through a series of grant-supported collaborations, Xeris is exploring additional uses for ready-to-use glucagon in mild-to-moderate hypoglycemia. Additionally, Xeris is evaluating stable, liquid glucagon as a combination therapy inside a physiological, closed-loop,bi-hormonal
LOW BLOOD SUGAR TREATMENT WITH INSULIN PRAMLINTIDE Pramlintide is the drug that resembles amylin, a human hormone that slows the absorption of carbohydrates through its effects on stomach emptying. Pramlintide administration mimics the human body's natural interaction between insulin and amylin to regulate normal blood sugar levels with meals. Xeris is currently developing a ready-to-use XERIS PHARMACEUTICALS ANNOUNCES POSITIVE TOPLINE RESULTS All subjects successfully self-administered a mini dose of RTU glucagon Rebound hypoglycemia observed more frequently in the placebo arm and with oral glucose tablet use Subcutaneous RTU glucagon is safe and well-tolerated CHICAGO --(BUSINESS WIRE)--Dec. 10, 2019-- XERIS PHARMACEUTICALS GRANTED FAST TRACK DESIGNATION BY Xeris Pharmaceuticals Granted Fast Track Designation by the FDA for Its Novel Concentrated Diazepam Formulation (XP-0863) For InjectionOctober 20, 2020
ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS PRODUCTS
Gvoke (glucagon injection) is not approved outside of the U.S. For more information, including how to access Gvoke, please contact Xeris Medical Team at 1-877-XERIS37 1-877-XERIS37 (1-877-937-4737).. Our medicines are approved in individual countries for specific uses, and the information shown is governed by local regulations. PRESS RELEASES FROM XERIS PHARMACEUTICALS Xeris Pharmaceuticals to Webcast Upcoming Presentations. 5/6/2021. Xeris Pharmaceuticals to Announce First Quarter 2021 Financial Results and Host Conference Call and Webcast on May 13, 2021. 5/6/2021. Xeris Pharmaceuticals Announces Extension of the Interest-Only Period of Its Debt Facility With Oxford Finance and Silicon Valley Bank. 4/29/2021. NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks.XERIS PIPELINE
Xeris has a robust pipeline across its ready-to-use portfolios. Through a series of grant-supported collaborations, Xeris is exploring additional uses for ready-to-use glucagon in mild-to-moderate hypoglycemia. Additionally, Xeris is evaluating stable, liquid glucagon as a combination therapy inside a physiological, closed-loop,bi-hormonal
XERIS FINANCIAL INFORMATION Financials. Find relevant financial information about Xeris through our annual reports and proxy statements. Annual Reports. Proxy Statements. Select Section Annual Reports Proxy Statements. Never miss a financial update by signing up for alerts.2019 10-K
Title: 2019 10-K Created Date: 3/11/2020 7:11:00 PM SEC FILING | XERIS PHARMACEUTICALS, INC. The summary risk factors described above should be read together with the text of the full risk factors below in the section entitled “Risk Factors” and the other information set forth in this Quarterly Report on Form 10-Q, including our condensed consolidated financial statements and the related notes, as well as in other documents that we file with the U.S. Securities and Exchange XERIS PHARMACEUTICALS ANNOUNCES NEW BOARD MEMBERS AND NEW Appointments reflect transition to publicly-listed company status. CHICAGO, July 30, 2018 (GLOBE NEWSWIRE) -- Xeris Pharmaceuticals, Inc. (NASDAQ:XERS), a specialty pharmaceutical company leveraging its novel technology platforms to develop and commercialize ready-to-use injectable and infusible drug formulations, announced today the appointment of four new independent members to DYNAMICS NAV 2017 ENVIRONMENT Remote Desktop Access: You can connect to the server in the 2017 Environment by following this link. Xeris Pharma NST: Access the 2017Environment using
XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, and pumps. Uncover what tomorrow brings. ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
PRESS RELEASES FROM XERIS PHARMACEUTICALS Xeris Pharmaceuticals to Webcast Upcoming Presentations. 5/6/2021. Xeris Pharmaceuticals to Announce First Quarter 2021 Financial Results and Host Conference Call and Webcast on May 13, 2021. 5/6/2021. Xeris Pharmaceuticals Announces Extension of the Interest-Only Period of Its Debt Facility With Oxford Finance and Silicon Valley Bank. 4/29/2021.XERIS PRODUCTS
XERIS INVESTOR EVENTS AND PRESENTATIONS Xeris-Strongbridge Conference Call. Supporting Materials. To listen to our webcast under 'events and presentations', click here. Dial-in Replay Number : (800) 585-8367. Dial-in Replay Number: (416) 621-4642.Passcode : 6359699.
XERIS LEADERSHIP
Paul R. Edick. Chairman & Chief Executive Officer. Mr. Edick joined Xeris Pharmaceuticals as Chairman and Chief Executive Officer in January 2017. Prior to Xeris, Mr. Edick was Founding Partner of 3G Advisors, a consultancy to the pharmaceutical, healthcare, and healthcare investor communities. From 2010 to 2014. Paul R. Edick. XERIS PHARMACEUTICALS ANNOUNCES POSITIVE FINDINGS FROM THE Rebound hypoglycemia observed in the placebo arm with oral glucose tablet use; No rebound hypoglycemia observed in the RTU glucagon treatment arm CHICAGO --(BUSINESS WIRE)--May 22, 2020-- Xeris Pharmaceuticals, Inc. (Nasdaq: XERS), a specialty LOW BLOOD SUGAR TREATMENT WITH INSULIN PRAMLINTIDE Pramlintide is the drug that resembles amylin, a human hormone that slows the absorption of carbohydrates through its effects on stomach emptying. Pramlintide administration mimics the human body's natural interaction between insulin and amylin to regulate normal blood sugar levels with meals. Xeris is currently developing a ready-to-use XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, and pumps. Uncover what tomorrow brings. ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
PRESS RELEASES FROM XERIS PHARMACEUTICALS Xeris Pharmaceuticals to Webcast Upcoming Presentations. 5/6/2021. Xeris Pharmaceuticals to Announce First Quarter 2021 Financial Results and Host Conference Call and Webcast on May 13, 2021. 5/6/2021. Xeris Pharmaceuticals Announces Extension of the Interest-Only Period of Its Debt Facility With Oxford Finance and Silicon Valley Bank. 4/29/2021.XERIS PRODUCTS
XERIS INVESTOR EVENTS AND PRESENTATIONS Xeris-Strongbridge Conference Call. Supporting Materials. To listen to our webcast under 'events and presentations', click here. Dial-in Replay Number : (800) 585-8367. Dial-in Replay Number: (416) 621-4642.Passcode : 6359699.
XERIS LEADERSHIP
Paul R. Edick. Chairman & Chief Executive Officer. Mr. Edick joined Xeris Pharmaceuticals as Chairman and Chief Executive Officer in January 2017. Prior to Xeris, Mr. Edick was Founding Partner of 3G Advisors, a consultancy to the pharmaceutical, healthcare, and healthcare investor communities. From 2010 to 2014. Paul R. Edick. XERIS PHARMACEUTICALS ANNOUNCES POSITIVE FINDINGS FROM THE Rebound hypoglycemia observed in the placebo arm with oral glucose tablet use; No rebound hypoglycemia observed in the RTU glucagon treatment arm CHICAGO --(BUSINESS WIRE)--May 22, 2020-- Xeris Pharmaceuticals, Inc. (Nasdaq: XERS), a specialty LOW BLOOD SUGAR TREATMENT WITH INSULIN PRAMLINTIDE Pramlintide is the drug that resembles amylin, a human hormone that slows the absorption of carbohydrates through its effects on stomach emptying. Pramlintide administration mimics the human body's natural interaction between insulin and amylin to regulate normal blood sugar levels with meals. Xeris is currently developing a ready-to-use NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks. XERIS PHARMACEUTICALS' XERISOL™ & XERIJECT™ FORMULATION XeriSol ™ and XeriJect ™ provide distinct advantages over existing formulations. Both XeriSol and XeriJect offer the opportunity to: Eliminate reconstitution. Enable long-term room temperature stability. Significantly reduce injection volume. Eliminate the requirement for intravenous (IV) infusion. These attributes can lead to products that CONTACT US - XERIS PHARMACEUTICALS, INC. Contact Us. Please contact the Xeris Contact Center at 1-877-XERIS-37 to submit a medical/product information question and/or to report an adverse event, and/or product complaint. By entering your email address, you agree to receive emails from Xeris (including promotionaloffers and
XERIS PIPELINE
Xeris has a robust pipeline across its ready-to-use portfolios. Through a series of grant-supported collaborations, Xeris is exploring additional uses for ready-to-use glucagon in mild-to-moderate hypoglycemia. Additionally, Xeris is evaluating stable, liquid glucagon as a combination therapy inside a physiological, closed-loop,bi-hormonal
XERIS RESEARCH AND DEVELOPMENT Xeris is developing its pipeline both through internal clinical development and external partnerships to advance the commercial potential of its products globally. Partner with us.SEC FILINGS
SEC Filings. Group: CHOOSE FROM LIST 3,4,5 Annual Filings Current Reports Mergers & Acquisitions Other Proxy Filings Quarterly Filings Registration Statements. Filing Year: - ANY - 2021 2020. Filing date. Form. Description. Filing Group. XERIS FINANCIAL INFORMATION Financials. Find relevant financial information about Xeris through our annual reports and proxy statements. Annual Reports. Proxy Statements. Select Section Annual Reports Proxy Statements. Never miss a financial update by signing up for alerts. XERISOL™ & XERIJECT™ READY-TO-USE, LIQUID-STABLE The solution is dried into a powder and reconstituted in an aprotic, polar (FDA-approved) liquid. The liquid prevents aggregation and fibrillation of the drug substance. 3. When a ready-to-use XeriSol formulation is injected into a patient, it is effectively placed back into a water environment where it CORPORATE GOVERNANCE Corporate Governance Guidelines. Code of Conduct. Xeris has an "Ethics and Compliance Hotline" for reports of potential Code violations, or for any questions/concerns: PARTNERING WITH XERIS PHARMACEUTICALS Partnering. Xeris is interested in establishing a portfolio of strategic collaborations with key partners who wish to codevelop products using Xeris’ platform technologies (XeriSol ™ and XeriJect ™ ). Currently, Xeris is working on a number of formulation feasibility programs in the areas of peptides, vaccines, and monoclonal antibodies. XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, and pumps. Uncover what tomorrow brings. ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. PRESS RELEASES FROM XERIS PHARMACEUTICALS Xeris Pharmaceuticals to Webcast Upcoming Presentations. 5/6/2021. Xeris Pharmaceuticals to Announce First Quarter 2021 Financial Results and Host Conference Call and Webcast on May 13, 2021. 5/6/2021. Xeris Pharmaceuticals Announces Extension of the Interest-Only Period of Its Debt Facility With Oxford Finance and Silicon Valley Bank. 4/29/2021.XERIS PRODUCTS
XERIS INVESTOR EVENTS AND PRESENTATIONSXERIS INVESTOR RELATIONSXERIS PHARMAXERIS PHARMACEUTICALS INC DISCUSSIONXEROX INVESTORXEROX INVESTOR RELATIONS SEC FILINGSXERIS PHARMACEUTICALS Xeris-Strongbridge Conference Call. Supporting Materials. To listen to our webcast under 'events and presentations', click here. Dial-in Replay Number : (800) 585-8367. Dial-in Replay Number: (416) 621-4642.Passcode : 6359699.
XERIS PIPELINE
Xeris has a robust pipeline across its ready-to-use portfolios. Through a series of grant-supported collaborations, Xeris is exploring additional uses for ready-to-use glucagon in mild-to-moderate hypoglycemia. Additionally, Xeris is evaluating stable, liquid glucagon as a combination therapy inside a physiological, closed-loop,bi-hormonal
XERIS PHARMACEUTICALS ANNOUNCES POSITIVE FINDINGS FROM THE Rebound hypoglycemia observed in the placebo arm with oral glucose tablet use; No rebound hypoglycemia observed in the RTU glucagon treatment arm CHICAGO --(BUSINESS WIRE)--May 22, 2020-- Xeris Pharmaceuticals, Inc. (Nasdaq: XERS), a specialtyXERIS LEADERSHIP
Paul R. Edick. Chairman & Chief Executive Officer. Mr. Edick joined Xeris Pharmaceuticals as Chairman and Chief Executive Officer in January 2017. Prior to Xeris, Mr. Edick was Founding Partner of 3G Advisors, a consultancy to the pharmaceutical, healthcare, and healthcare investor communities. From 2010 to 2014. Paul R. Edick. XERIS PHARMACEUTICALS, INC.ABOUT XERISRESEARCH & DEVELOPMENTNEWS ROOMINVESTORSCAREERSCONTACT US While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, and pumps. Uncover what tomorrow brings. ABOUT XERIS PHARMACEUTICALS, INC. Meet Xeris Pharmaceuticals. Xeris is a specialty pharmaceutical company leveraging novel formulation technology platforms to develop and commercialize ready-to-use, liquid-stable injectables. Come seewhat we're about.
XERIS CAREER OPPORTUNITIES Careers. Our team is dedicated to: The development of patient-focused, ready-to-use injectable drugs that address critical, unmet medical needs. Lowering the overall cost of treatments. The improvement of both the patient experience and quality of life. Based upon a foundation of strong platform technology, we intend to buildsustainable
XERIS INVESTOR RELATIONS See Xeris' financial and stock information, events & presentations, corporate governance, and investor FAQs. PRESS RELEASES FROM XERIS PHARMACEUTICALS Xeris Pharmaceuticals to Webcast Upcoming Presentations. 5/6/2021. Xeris Pharmaceuticals to Announce First Quarter 2021 Financial Results and Host Conference Call and Webcast on May 13, 2021. 5/6/2021. Xeris Pharmaceuticals Announces Extension of the Interest-Only Period of Its Debt Facility With Oxford Finance and Silicon Valley Bank. 4/29/2021.XERIS PRODUCTS
XERIS INVESTOR EVENTS AND PRESENTATIONSXERIS INVESTOR RELATIONSXERIS PHARMAXERIS PHARMACEUTICALS INC DISCUSSIONXEROX INVESTORXEROX INVESTOR RELATIONS SEC FILINGSXERIS PHARMACEUTICALS Xeris-Strongbridge Conference Call. Supporting Materials. To listen to our webcast under 'events and presentations', click here. Dial-in Replay Number : (800) 585-8367. Dial-in Replay Number: (416) 621-4642.Passcode : 6359699.
XERIS PIPELINE
Xeris has a robust pipeline across its ready-to-use portfolios. Through a series of grant-supported collaborations, Xeris is exploring additional uses for ready-to-use glucagon in mild-to-moderate hypoglycemia. Additionally, Xeris is evaluating stable, liquid glucagon as a combination therapy inside a physiological, closed-loop,bi-hormonal
XERIS PHARMACEUTICALS ANNOUNCES POSITIVE FINDINGS FROM THE Rebound hypoglycemia observed in the placebo arm with oral glucose tablet use; No rebound hypoglycemia observed in the RTU glucagon treatment arm CHICAGO --(BUSINESS WIRE)--May 22, 2020-- Xeris Pharmaceuticals, Inc. (Nasdaq: XERS), a specialtyXERIS LEADERSHIP
Paul R. Edick. Chairman & Chief Executive Officer. Mr. Edick joined Xeris Pharmaceuticals as Chairman and Chief Executive Officer in January 2017. Prior to Xeris, Mr. Edick was Founding Partner of 3G Advisors, a consultancy to the pharmaceutical, healthcare, and healthcare investor communities. From 2010 to 2014. Paul R. Edick. NEWS ARTICLES, PRESS RELEASES, AND EVENTS FROM XERIS See the latest news about the progress and achievements of Xeris' platforms, financial and partnership communications, and event info for conferences and walks. CONTACT US - XERIS PHARMACEUTICALS, INC. Contact Us. Please contact the Xeris Contact Center at 1-877-XERIS-37 to submit a medical/product information question and/or to report an adverse event, and/or product complaint. By entering your email address, you agree to receive emails from Xeris (including promotionaloffers and
CORPORATE GOVERNANCE Corporate Governance Guidelines. Code of Conduct. Xeris has an "Ethics and Compliance Hotline" for reports of potential Code violations, or for any questions/concerns: PARTNERING WITH XERIS PHARMACEUTICALS Partnering. Xeris is interested in establishing a portfolio of strategic collaborations with key partners who wish to codevelop products using Xeris’ platform technologies (XeriSol ™ and XeriJect ™ ). Currently, Xeris is working on a number of formulation feasibility programs in the areas of peptides, vaccines, and monoclonal antibodies.SEC FILINGS
SEC Filings. Group: CHOOSE FROM LIST 3,4,5 Annual Filings Current Reports Mergers & Acquisitions Other Proxy Filings Quarterly Filings Registration Statements. Filing Year: - ANY - 2021 2020. Filing date. Form. Description. Filing Group. XERIS FINANCIAL INFORMATION Financials. Find relevant financial information about Xeris through our annual reports and proxy statements. Annual Reports. Proxy Statements. Select Section Annual Reports Proxy Statements. Never miss a financial update by signing up for alerts. SEC FILING | XERIS PHARMACEUTICALS, INC. PRICING TERM SHEET. June 25, 2020. Xeris Pharmaceuticals, Inc. Concurrent Offerings of. $75,000,000 Aggregate Principal Amount of. 5.00% Convertible Senior Notes due 2025 (the “Convertible Notes Offering”) and. 7,400,000 Shares of Common Stock, par value $0.001 per share (the “Common Stock Offering”) The information in thispricing term
LOW BLOOD SUGAR TREATMENT WITH INSULIN PRAMLINTIDE Pramlintide is the drug that resembles amylin, a human hormone that slows the absorption of carbohydrates through its effects on stomach emptying. Pramlintide administration mimics the human body's natural interaction between insulin and amylin to regulate normal blood sugar levels with meals. Xeris is currently developing a ready-to-use XERIS PHARMACEUTICALS ANNOUNCES POSITIVE TOPLINE RESULTS All subjects successfully self-administered a mini dose of RTU glucagon Rebound hypoglycemia observed more frequently in the placebo arm and with oral glucose tablet use Subcutaneous RTU glucagon is safe and well-tolerated CHICAGO --(BUSINESS WIRE)--Dec. 10, 2019-- XerisPharmaceuticals
SEC FILING | XERIS PHARMACEUTICALS, INC. the registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with section 8(a) of the securities act of 1933, as amended, or until the registration statement shall become* About Xeris
* Meet Xeris Pharmaceuticals* Products
* Therapeutic Areas
* Medical Affairs
* Leadership
* Research & Development* Pipeline
* Technology
* Partnering
* Expanded Access Policy* News Room
* Press Releases
* Events
* Investors
* Events & Presentations* SEC Filings
* Stock Information
* Financials
* Corporate Governance* Investor FAQs
* Careers
* Contact Us
*
Stock information
NASDAQ: XERS
$4.05
Jun 04, 2021 04:00 PM EST NEWS ROOM Xeris Pharmaceuticals, Inc. to Acquire Strongbridge Biopharma plc in Stock and CVR Transaction, Creating an Innovative Leader in Endocrinology and Rare Diseases Upcoming events Partnerwith us Meet our
leadership
INNOVATIVE MEDICAL SOLUTIONS FOR LONG-STANDING CHALLENGES Our unique technology addresses the issues that arise from water-based delivery systems with a novel, proprietary, non-aqueous formulation. This innovative approach has been implemented in GVOKE® PRE-FILLED SYRINGE (GLUCAGON INJECTION) and GVOKE HYPOPEN®. Learn more about our GVOKE® offerings SOLUTIONS FOR MANY THERAPEUTIC AREAS We're currently developing innovative, ready-to-use solutions for various therapeutic categories to remove many associated treatment burdens and ultimately improve patients' lives.See how we can help
PROGRESS FOR SEVERE HYPOGLYCEMIA AND EPILEPSY ARE JUST THE START While clinical trials are currently underway to investigate how Xeris' formulation technology can be applied for hypoglycemia and epilepsy management, Xeris' novel formulation also has the potential for use in single-use auto-injectors, prefilled syringes, ready-to-use vials, andpumps.
Uncover what tomorrow brings A DIFFERENT APPROACH At Xeris, we're always looking for ways to really impact patients' lives. That's why we offer a novel formulation technology to bring ready-to-use, liquid-stable injectables to patients whose lives are often burdened by the “time of use” preparation routine. Explore what we have to offer You have chosen a link to an external site. By clicking "ok", a new window will open. Do you wish to continue?OK No Thanks!
Not opening? Check to see you have pop-ups enabled or copy link belowin a new window.
We use cookies to help personalize your experience. For more information on how we use cookies and similar technologies, and how you can change your settings, please see our Privacy Policy.
Accept Cookies
BACK
TO TOP instagram
PLEASE SEE FULL PRESCRIBING INFORMATION* Contact Us |
* Terms of Use |
* Privacy Policy |
* Privacy Shield Policy | * Financial Conflict of Interest Policy|
* Sitemap
Gvoke®, Gvoke HypoPen®, Xerisol™, Xeriject™, Xeris Pharmaceuticals®, and their associated logos are trademarks of Xeris Pharmaceuticals, Inc. All other trademarks are the property of theirrespective owners.
Copyright ©2020 Xeris Pharmaceuticals, Inc. All rights reserved. US-CORP-19-00006v10 04/21*
*
*
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0