Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://canucan.com
Are you over 18 and want to see adult content?

A complete backup of https://namdeb.com
Are you over 18 and want to see adult content?

A complete backup of https://mfa-ks.net
Are you over 18 and want to see adult content?
A complete backup of https://bellevuebotanical.org
Are you over 18 and want to see adult content?
A complete backup of https://klavakot.ru
Are you over 18 and want to see adult content?

A complete backup of https://yalls.org
Are you over 18 and want to see adult content?
A complete backup of https://aestheticmagazinetoronto.com
Are you over 18 and want to see adult content?
A complete backup of https://piggy-bank.co.uk
Are you over 18 and want to see adult content?

A complete backup of https://projetocomprova.com.br
Are you over 18 and want to see adult content?

A complete backup of https://verdesmeraldaolive.com
Are you over 18 and want to see adult content?

A complete backup of https://ilsitodifirenze.it
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of https://delba.be
Are you over 18 and want to see adult content?
A complete backup of https://ccsingenieros.com
Are you over 18 and want to see adult content?
A complete backup of https://nearlynatural.com
Are you over 18 and want to see adult content?
A complete backup of https://amirhoseinabolfazli.ir
Are you over 18 and want to see adult content?
A complete backup of https://banhammer.us
Are you over 18 and want to see adult content?
A complete backup of https://shockya.com
Are you over 18 and want to see adult content?

A complete backup of https://6play.fr
Are you over 18 and want to see adult content?
A complete backup of https://wrightsmedia.com
Are you over 18 and want to see adult content?
A complete backup of https://nonsolofitness.it
Are you over 18 and want to see adult content?

A complete backup of https://oekolog.at
Are you over 18 and want to see adult content?
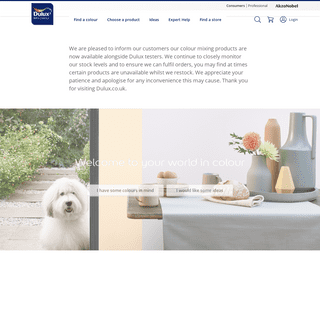
A complete backup of https://dulux.co.uk
Are you over 18 and want to see adult content?
Text
ENGAGEMENT
Capture outcome assessments remotely. Deploy diaries, ePROs, and more rapidly. Learn More. We know now that when patients are able to access a clinical trial online, using telemedicine and remote consent, most clinical trials will enroll three times faster. That’s anCAREERS AT MEDABLE
VP / Sr Director of Application Platform. Palo Alto, CA. VP of Consumer Experience Engineering. Palo Alto, CA. VP of Enterprise Engineering (Remote) Palo Alto, CA. QA Validation Specialist. Palo Alto, CA. Sr. Instructional Designer - REMOTE.OUR LEADERSHIP TEAM
Our Company. Medable's mission is to get effective therapies to patients faster. We provide an end-to-end, cloud-based platform with a flexible suite of tools that allows patients, healthcare providers, clinical research organizations and pharmaceutical sponsors to work together as a team in clinical trials.MEDABLE
It all starts with the right protocol. In this webinar, Medable and WCG Avoca will share results from the 2020 Avoca State of the Industry Survey and real clinical trial case studies from Medable. In doing so, attendees will understand the common challenges faced by study teams, and how weaving DCT strategies into their protocols can helpMEDABLE
Today, the multiple disparate touch points required for clinical research are burdensome for patients, sites and sponsors. Patient engagement, recruitment, and surveillance for trials are crucial to unlocking these challenges and expanding research access.MEDABLE BLOG
Follow Medable's blog with thought-leading insights on transforming the patient experience through decentralized and virtual digitaltrials.
MEDABLE
Please enter your org name to sign in. SelectMEDABLE
The right to object to the sale of personal data. The right to access your own personal information collected by Medable (also called a data portability request). The right to equal service and price, even for consumers who exercise their privacy rights. NO ONE SHOULD WALK A CANCER JOURNEY ALONE/LESSONS FROM MY No One Should Walk a Cancer Journey Alone/Lessons from My Father. Oct 7, 2020 4:39:35 PM. In 2020, over 276,000 American women and 2,600 men will be newly diagnosed with breast cancer. For these individuals, Breast Cancer Awareness month is especially poignant. Those hearing the words ‘you have cancer’ quickly realize the importance ofMEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt at MEDABLE - THE DECENTRALIZED CLINICAL TRIAL PLATFORMSOLUTIONSRESOURCESCOMPANYENROLLMENT AND SCREENINGPATIENTENGAGEMENT
Capture outcome assessments remotely. Deploy diaries, ePROs, and more rapidly. Learn More. We know now that when patients are able to access a clinical trial online, using telemedicine and remote consent, most clinical trials will enroll three times faster. That’s anCAREERS AT MEDABLE
VP / Sr Director of Application Platform. Palo Alto, CA. VP of Consumer Experience Engineering. Palo Alto, CA. VP of Enterprise Engineering (Remote) Palo Alto, CA. QA Validation Specialist. Palo Alto, CA. Sr. Instructional Designer - REMOTE.OUR LEADERSHIP TEAM
Our Company. Medable's mission is to get effective therapies to patients faster. We provide an end-to-end, cloud-based platform with a flexible suite of tools that allows patients, healthcare providers, clinical research organizations and pharmaceutical sponsors to work together as a team in clinical trials.MEDABLE
It all starts with the right protocol. In this webinar, Medable and WCG Avoca will share results from the 2020 Avoca State of the Industry Survey and real clinical trial case studies from Medable. In doing so, attendees will understand the common challenges faced by study teams, and how weaving DCT strategies into their protocols can helpMEDABLE
Today, the multiple disparate touch points required for clinical research are burdensome for patients, sites and sponsors. Patient engagement, recruitment, and surveillance for trials are crucial to unlocking these challenges and expanding research access.MEDABLE BLOG
Follow Medable's blog with thought-leading insights on transforming the patient experience through decentralized and virtual digitaltrials.
MEDABLE
Please enter your org name to sign in. SelectMEDABLE
The right to object to the sale of personal data. The right to access your own personal information collected by Medable (also called a data portability request). The right to equal service and price, even for consumers who exercise their privacy rights. NO ONE SHOULD WALK A CANCER JOURNEY ALONE/LESSONS FROM MY No One Should Walk a Cancer Journey Alone/Lessons from My Father. Oct 7, 2020 4:39:35 PM. In 2020, over 276,000 American women and 2,600 men will be newly diagnosed with breast cancer. For these individuals, Breast Cancer Awareness month is especially poignant. Those hearing the words ‘you have cancer’ quickly realize the importance ofMEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt atMEDABLE
It all starts with the right protocol. In this webinar, Medable and WCG Avoca will share results from the 2020 Avoca State of the Industry Survey and real clinical trial case studies from Medable. In doing so, attendees will understand the common challenges faced by study teams, and how weaving DCT strategies into their protocols can helpGARTNER REPORT
Gartner Report. “We maintain that d-clinical cloud solutions promote more effective solution integration, increased data transparency and reduced cost, and can decrease trial cycle time.”. - Gartner, in their August 2020 report. At Medable, we’ve long believed in the power of digital cloud solutions. In fact, as a pioneer of andprovider
MEDABLE
Today, the multiple disparate touch points required for clinical research are burdensome for patients, sites and sponsors. Patient engagement, recruitment, and surveillance for trials are crucial to unlocking these challenges and expanding research access.MEDABLE
Medable Inc (“Medable”, “we” or “us”) provides various products and services, to be used in clinical research studies or clinical trials. This ACCESS (American COVID-19 Collaborative Enabling Seamless Science or “ACCESS”) App was created during the epidemiological emergency created by the SARS-CoV-2 virus as anobservational
MEDABLE
Please enter your org name to sign in. Select DIGITAL PATIENT SCREENING Find the right patients by integrating with a variety to digital recruitment tools. Digitalizing the process, we were able to remotely screen 11,000 patients in record time, reducing eligible patient enrollment from two years to just one year. Site reduction of 75% from100 sites
CONTACT US - NEED HELP? WE'RE HERE WHEN YOU NEED US We are here to answer your questions Medable HQ T: (877) 820-6259 525 University Ave, Suite A70 Palo Alto, CA 94301 If you are interested in investing in Medable please contact us at: ir@medable.com If you are a Customer and need support please email Support@medable.comMEDABLE
Medable Inc (“Medable”, “we” or “us”) provides various products and services, to be used in clinical settings. Medable offers this Televisit Aapp to facilitate the visit between a healthcare provider and patient in a HIPPA-secure manner.in accordance with the requirements of the Health Insurance Portability and AccountabilityAct of
WHY I JOINED MEDABLE This type of revolution is why I joined Medable. When interviewing with the CEO Dr. Michelle Longmire and her co-founder Tim Smith, it was clear – we shared the goal of doing the same for clinical trials. Decentralizing operations, opening up freedom for patients and sites, creating digital therapies that could be delivered anywhere the MEDABLE NAMED “LIFE SCIENCES INNOVATION CHAMPION” IN THE Written by Dan Horsey. Dan joins the Marketing team at Medable. Previously, Dan was a Content Manager at Signant Health and a Marketing Manager at IQVIA. He got his start in healthcare marketing in 2014, working with Pharmacy Benefits Managers in the managed Medicare and Medicaid spaces, and has been writing in healthcare capacities for over 10 years. MEDABLE - THE DECENTRALIZED CLINICAL TRIAL PLATFORMSOLUTIONSRESOURCESCOMPANYENROLLMENT AND SCREENINGPATIENTENGAGEMENT
Capture outcome assessments remotely. Deploy diaries, ePROs, and more rapidly. Learn More. We know now that when patients are able to access a clinical trial online, using telemedicine and remote consent, most clinical trials will enroll three times faster. That’s anCAREERS AT MEDABLE
VP / Sr Director of Application Platform. Palo Alto, CA. VP of Consumer Experience Engineering. Palo Alto, CA. VP of Enterprise Engineering (Remote) Palo Alto, CA. QA Validation Specialist. Palo Alto, CA. Sr. Instructional Designer - REMOTE.OUR LEADERSHIP TEAM
Our Company. Medable's mission is to get effective therapies to patients faster. We provide an end-to-end, cloud-based platform with a flexible suite of tools that allows patients, healthcare providers, clinical research organizations and pharmaceutical sponsors to work together as a team in clinical trials.MEDABLE
It all starts with the right protocol. In this webinar, Medable and WCG Avoca will share results from the 2020 Avoca State of the Industry Survey and real clinical trial case studies from Medable. In doing so, attendees will understand the common challenges faced by study teams, and how weaving DCT strategies into their protocols can helpMEDABLE
Today, the multiple disparate touch points required for clinical research are burdensome for patients, sites and sponsors. Patient engagement, recruitment, and surveillance for trials are crucial to unlocking these challenges and expanding research access.MEDABLE BLOG
Follow Medable's blog with thought-leading insights on transforming the patient experience through decentralized and virtual digitaltrials.
MEDABLE
Please enter your org name to sign in. SelectMEDABLE
The right to object to the sale of personal data. The right to access your own personal information collected by Medable (also called a data portability request). The right to equal service and price, even for consumers who exercise their privacy rights. NO ONE SHOULD WALK A CANCER JOURNEY ALONE/LESSONS FROM MY No One Should Walk a Cancer Journey Alone/Lessons from My Father. Oct 7, 2020 4:39:35 PM. In 2020, over 276,000 American women and 2,600 men will be newly diagnosed with breast cancer. For these individuals, Breast Cancer Awareness month is especially poignant. Those hearing the words ‘you have cancer’ quickly realize the importance ofMEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt at MEDABLE - THE DECENTRALIZED CLINICAL TRIAL PLATFORMSOLUTIONSRESOURCESCOMPANYENROLLMENT AND SCREENINGPATIENTENGAGEMENT
Capture outcome assessments remotely. Deploy diaries, ePROs, and more rapidly. Learn More. We know now that when patients are able to access a clinical trial online, using telemedicine and remote consent, most clinical trials will enroll three times faster. That’s anCAREERS AT MEDABLE
VP / Sr Director of Application Platform. Palo Alto, CA. VP of Consumer Experience Engineering. Palo Alto, CA. VP of Enterprise Engineering (Remote) Palo Alto, CA. QA Validation Specialist. Palo Alto, CA. Sr. Instructional Designer - REMOTE.OUR LEADERSHIP TEAM
Our Company. Medable's mission is to get effective therapies to patients faster. We provide an end-to-end, cloud-based platform with a flexible suite of tools that allows patients, healthcare providers, clinical research organizations and pharmaceutical sponsors to work together as a team in clinical trials.MEDABLE
It all starts with the right protocol. In this webinar, Medable and WCG Avoca will share results from the 2020 Avoca State of the Industry Survey and real clinical trial case studies from Medable. In doing so, attendees will understand the common challenges faced by study teams, and how weaving DCT strategies into their protocols can helpMEDABLE
Today, the multiple disparate touch points required for clinical research are burdensome for patients, sites and sponsors. Patient engagement, recruitment, and surveillance for trials are crucial to unlocking these challenges and expanding research access.MEDABLE BLOG
Follow Medable's blog with thought-leading insights on transforming the patient experience through decentralized and virtual digitaltrials.
MEDABLE
Please enter your org name to sign in. SelectMEDABLE
The right to object to the sale of personal data. The right to access your own personal information collected by Medable (also called a data portability request). The right to equal service and price, even for consumers who exercise their privacy rights. NO ONE SHOULD WALK A CANCER JOURNEY ALONE/LESSONS FROM MY No One Should Walk a Cancer Journey Alone/Lessons from My Father. Oct 7, 2020 4:39:35 PM. In 2020, over 276,000 American women and 2,600 men will be newly diagnosed with breast cancer. For these individuals, Breast Cancer Awareness month is especially poignant. Those hearing the words ‘you have cancer’ quickly realize the importance ofMEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt atMEDABLE
It all starts with the right protocol. In this webinar, Medable and WCG Avoca will share results from the 2020 Avoca State of the Industry Survey and real clinical trial case studies from Medable. In doing so, attendees will understand the common challenges faced by study teams, and how weaving DCT strategies into their protocols can helpGARTNER REPORT
Gartner Report. “We maintain that d-clinical cloud solutions promote more effective solution integration, increased data transparency and reduced cost, and can decrease trial cycle time.”. - Gartner, in their August 2020 report. At Medable, we’ve long believed in the power of digital cloud solutions. In fact, as a pioneer of andprovider
MEDABLE
Today, the multiple disparate touch points required for clinical research are burdensome for patients, sites and sponsors. Patient engagement, recruitment, and surveillance for trials are crucial to unlocking these challenges and expanding research access.MEDABLE
Medable Inc (“Medable”, “we” or “us”) provides various products and services, to be used in clinical research studies or clinical trials. This ACCESS (American COVID-19 Collaborative Enabling Seamless Science or “ACCESS”) App was created during the epidemiological emergency created by the SARS-CoV-2 virus as anobservational
MEDABLE
Please enter your org name to sign in. Select DIGITAL PATIENT SCREENING Find the right patients by integrating with a variety to digital recruitment tools. Digitalizing the process, we were able to remotely screen 11,000 patients in record time, reducing eligible patient enrollment from two years to just one year. Site reduction of 75% from100 sites
CONTACT US - NEED HELP? WE'RE HERE WHEN YOU NEED US We are here to answer your questions Medable HQ T: (877) 820-6259 525 University Ave, Suite A70 Palo Alto, CA 94301 If you are interested in investing in Medable please contact us at: ir@medable.com If you are a Customer and need support please email Support@medable.comMEDABLE
Medable Inc (“Medable”, “we” or “us”) provides various products and services, to be used in clinical settings. Medable offers this Televisit Aapp to facilitate the visit between a healthcare provider and patient in a HIPPA-secure manner.in accordance with the requirements of the Health Insurance Portability and AccountabilityAct of
WHY I JOINED MEDABLE This type of revolution is why I joined Medable. When interviewing with the CEO Dr. Michelle Longmire and her co-founder Tim Smith, it was clear – we shared the goal of doing the same for clinical trials. Decentralizing operations, opening up freedom for patients and sites, creating digital therapies that could be delivered anywhere the MEDABLE NAMED “LIFE SCIENCES INNOVATION CHAMPION” IN THE Written by Dan Horsey. Dan joins the Marketing team at Medable. Previously, Dan was a Content Manager at Signant Health and a Marketing Manager at IQVIA. He got his start in healthcare marketing in 2014, working with Pharmacy Benefits Managers in the managed Medicare and Medicaid spaces, and has been writing in healthcare capacities for over 10 years. MEDABLE - THE DECENTRALIZED CLINICAL TRIAL PLATFORMSOLUTIONSRESOURCESCOMPANYENROLLMENT AND SCREENINGPATIENTENGAGEMENT
Capture outcome assessments remotely. Deploy diaries, ePROs, and more rapidly. Learn More. We know now that when patients are able to access a clinical trial online, using telemedicine and remote consent, most clinical trials will enroll three times faster. That’s anCAREERS AT MEDABLE
VP / Sr Director of Application Platform. Palo Alto, CA. VP of Consumer Experience Engineering. Palo Alto, CA. VP of Enterprise Engineering (Remote) Palo Alto, CA. QA Validation Specialist. Palo Alto, CA. Sr. Instructional Designer - REMOTE.OUR LEADERSHIP TEAM
Our Company. Medable's mission is to get effective therapies to patients faster. We provide an end-to-end, cloud-based platform with a flexible suite of tools that allows patients, healthcare providers, clinical research organizations and pharmaceutical sponsors to work together as a team in clinical trials.MEDABLE
It all starts with the right protocol. In this webinar, Medable and WCG Avoca will share results from the 2020 Avoca State of the Industry Survey and real clinical trial case studies from Medable. In doing so, attendees will understand the common challenges faced by study teams, and how weaving DCT strategies into their protocols can help ACCESS APP - MEDABLE Medable Inc (“Medable”, “we” or “us”) provides various products and services, to be used in clinical research studies or clinical trials. This ACCESS (American COVID-19 Collaborative Enabling Seamless Science or “ACCESS”) App was created during the epidemiological emergency created by the SARS-CoV-2 virus as anobservational
MEDABLE
Please enter your org name to sign in. SelectMEDABLE BLOG
Gillian (Gill) Livock is Head of Commercial & Strategic Partnerships at Medable, responsible for driving collaborations which have united many different stakeholders to focus on bringingeffective therapies to patients faster. COVID 19 has demanded even more innovation, collaboration, and resilience as we identify ways to support patients, healthcare professionals and keep clinical trials WHY I JOINED MEDABLE This type of revolution is why I joined Medable. When interviewing with the CEO Dr. Michelle Longmire and her co-founder Tim Smith, it was clear – we shared the goal of doing the same for clinical trials. Decentralizing operations, opening up freedom for patients and sites, creating digital therapies that could be delivered anywhere the NO ONE SHOULD WALK A CANCER JOURNEY ALONE/LESSONS FROM MY No One Should Walk a Cancer Journey Alone/Lessons from My Father. Oct 7, 2020 4:39:35 PM. In 2020, over 276,000 American women and 2,600 men will be newly diagnosed with breast cancer. For these individuals, Breast Cancer Awareness month is especially poignant. Those hearing the words ‘you have cancer’ quickly realize the importance ofMEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt at MEDABLE - THE DECENTRALIZED CLINICAL TRIAL PLATFORMSOLUTIONSRESOURCESCOMPANYENROLLMENT AND SCREENINGPATIENTENGAGEMENT
Capture outcome assessments remotely. Deploy diaries, ePROs, and more rapidly. Learn More. We know now that when patients are able to access a clinical trial online, using telemedicine and remote consent, most clinical trials will enroll three times faster. That’s anCAREERS AT MEDABLE
VP / Sr Director of Application Platform. Palo Alto, CA. VP of Consumer Experience Engineering. Palo Alto, CA. VP of Enterprise Engineering (Remote) Palo Alto, CA. QA Validation Specialist. Palo Alto, CA. Sr. Instructional Designer - REMOTE.OUR LEADERSHIP TEAM
Our Company. Medable's mission is to get effective therapies to patients faster. We provide an end-to-end, cloud-based platform with a flexible suite of tools that allows patients, healthcare providers, clinical research organizations and pharmaceutical sponsors to work together as a team in clinical trials.MEDABLE
It all starts with the right protocol. In this webinar, Medable and WCG Avoca will share results from the 2020 Avoca State of the Industry Survey and real clinical trial case studies from Medable. In doing so, attendees will understand the common challenges faced by study teams, and how weaving DCT strategies into their protocols can help ACCESS APP - MEDABLE Medable Inc (“Medable”, “we” or “us”) provides various products and services, to be used in clinical research studies or clinical trials. This ACCESS (American COVID-19 Collaborative Enabling Seamless Science or “ACCESS”) App was created during the epidemiological emergency created by the SARS-CoV-2 virus as anobservational
MEDABLE
Please enter your org name to sign in. SelectMEDABLE BLOG
Gillian (Gill) Livock is Head of Commercial & Strategic Partnerships at Medable, responsible for driving collaborations which have united many different stakeholders to focus on bringingeffective therapies to patients faster. COVID 19 has demanded even more innovation, collaboration, and resilience as we identify ways to support patients, healthcare professionals and keep clinical trials WHY I JOINED MEDABLE This type of revolution is why I joined Medable. When interviewing with the CEO Dr. Michelle Longmire and her co-founder Tim Smith, it was clear – we shared the goal of doing the same for clinical trials. Decentralizing operations, opening up freedom for patients and sites, creating digital therapies that could be delivered anywhere the NO ONE SHOULD WALK A CANCER JOURNEY ALONE/LESSONS FROM MY No One Should Walk a Cancer Journey Alone/Lessons from My Father. Oct 7, 2020 4:39:35 PM. In 2020, over 276,000 American women and 2,600 men will be newly diagnosed with breast cancer. For these individuals, Breast Cancer Awareness month is especially poignant. Those hearing the words ‘you have cancer’ quickly realize the importance ofMEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt at TELEVISIT FOR CLINICAL TRIALS Pair TeleVisits with eConsent or eCOA. Reduce patient and site burden. Increase data quality and adherence. A Harvard School of Medicine study, in fact, showed that an in-person doctor visit in the U.S. takes about 121 minutes – and only 20 minutes of that time is spent face-to-face with a doctor. Opportunity Costs of Ambulatory MedicalCare
ACCESS APP - MEDABLE Medable Inc (“Medable”, “we” or “us”) provides various products and services, to be used in clinical research studies or clinical trials. This ACCESS (American COVID-19 Collaborative Enabling Seamless Science or “ACCESS”) App was created during the epidemiological emergency created by the SARS-CoV-2 virus as anobservational
GARTNER REPORT
Gartner Report. “We maintain that d-clinical cloud solutions promote more effective solution integration, increased data transparency and reduced cost, and can decrease trial cycle time.”. - Gartner, in their August 2020 report. At Medable, we’ve long believed in the power of digital cloud solutions. In fact, as a pioneer of andprovider
DIGITAL PATIENT SCREENING Find the right patients by integrating with a variety to digital recruitment tools. Digitalizing the process, we were able to remotely screen 11,000 patients in record time, reducing eligible patient enrollment from two years to just one year. Site reduction of 75% from100 sites
MEDABLE
Medable Inc (“Medable”, “we” or “us”) provides various products and services, to be used in clinical settings. Medable offers this Televisit Aapp to facilitate the visit between a healthcare provider and patient in a HIPPA-secure manner.in accordance with the requirements of the Health Insurance Portability and AccountabilityAct of
CONTACT US - NEED HELP? WE'RE HERE WHEN YOU NEED US Contact Medable. We are here to answer your questions. Medable HQ. T: (877) 820-6259. 525 University Ave, Suite A70. Palo Alto, CA 94301. If you are interested in investing in Medable please contact us at: ir@medable.com. First Name*. Last Name*.MEDABLE BLOG
Follow Medable's blog with thought-leading insights on transforming the patient experience through decentralized and virtual digitaltrials.
RESOURCES - MEDABLE
Resources about Decentralized Clinical Trials. 11/10 SCRS Lessons from the Classroom: What Clinical Research Can Learn from Education’s Transition to Remote Learning WHY I JOINED MEDABLE This type of revolution is why I joined Medable. When interviewing with the CEO Dr. Michelle Longmire and her co-founder Tim Smith, it was clear – we shared the goal of doing the same for clinical trials. Decentralizing operations, opening up freedom for patients and sites, creating digital therapies that could be delivered anywhere the HIGHLIGHTS FROM SCOPE 2021, AND THE CLINICAL TRIAL ROAD AHEAD Highlights From Scope 2021, and the Clinical Trial Road Ahead. Those in the clinical trial industry understand that there are several barometers that can be used to gauge the overall state of the industry. Chief among these are the “big conferences” that occureach year, such as
MEDABLE - THE DECENTRALIZED CLINICAL TRIAL PLATFORMSOLUTIONSRESOURCESCOMPANYENROLLMENT AND SCREENINGPATIENTENGAGEMENT
Medable is powering the future of clinical trials, with our decentralized and remote trial platform.CAREERS AT MEDABLE
Medable is pioneering a new category of life science technologies, and we're looking for glass-breakers to join us.OUR LEADERSHIP TEAM
Michelle Longmire is the Co-Founder and CEO of Medable. As a Stanford-trained physician-scientist, Dr. Longmire identified critical barriers to drug development and founded Medable to pioneer a new category of clinical trial technologiesMEDABLE
Medable is on a mission to get effective therapies to patients faster by transforming clinical drug development with disruptivetechnologies.
MEDABLE BLOG
Follow Medable's blog with thought-leading insights on transforming the patient experience through decentralized and virtual digitaltrials.
INTRODUCTION
Here you'll find comprehensive guides, step-by-step tutorials, and API reference documents to help you get up and going with Medable's products as quickly as possible.MEDABLE
Please enter your org name to sign in. Select WHY I JOINED MEDABLE When medicine changes, the world changes: new treatments, vaccines, and therapies create tectonic shifts in global quality of life. But it’s not easy—the space of possible treatments is vast, and the steps we take to a solution can be uncertain, logistically difficult,and costly.
MEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt at NO ONE SHOULD WALK A CANCER JOURNEY ALONE/LESSONS FROM MYAMERICAN BREAST CANCER WALKAMERICAN CANCER SOCIETY BREAST CANCER WALKBREAST CANCER WALK IN OCTOBERBREAST CANCER WALKSLONG ISLAND BREAST CANCERWALK
In 2020, o ver 276,000 American women and 2,600 men will be newly diagnosed with breast cancer. For these individuals, Breast Cancer Awareness month is especially poignant. Those hearing the words ‘you have cancer’ quickly realize the importance of having a support network as they move through diagnosis, treatment, and survivorship. MEDABLE - THE DECENTRALIZED CLINICAL TRIAL PLATFORMSOLUTIONSRESOURCESCOMPANYENROLLMENT AND SCREENINGPATIENTENGAGEMENT
Medable is powering the future of clinical trials, with our decentralized and remote trial platform.CAREERS AT MEDABLE
Medable is pioneering a new category of life science technologies, and we're looking for glass-breakers to join us.OUR LEADERSHIP TEAM
Michelle Longmire is the Co-Founder and CEO of Medable. As a Stanford-trained physician-scientist, Dr. Longmire identified critical barriers to drug development and founded Medable to pioneer a new category of clinical trial technologiesMEDABLE
Medable is on a mission to get effective therapies to patients faster by transforming clinical drug development with disruptivetechnologies.
MEDABLE BLOG
Follow Medable's blog with thought-leading insights on transforming the patient experience through decentralized and virtual digitaltrials.
INTRODUCTION
Here you'll find comprehensive guides, step-by-step tutorials, and API reference documents to help you get up and going with Medable's products as quickly as possible.MEDABLE
Please enter your org name to sign in. Select WHY I JOINED MEDABLE When medicine changes, the world changes: new treatments, vaccines, and therapies create tectonic shifts in global quality of life. But it’s not easy—the space of possible treatments is vast, and the steps we take to a solution can be uncertain, logistically difficult,and costly.
MEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt at NO ONE SHOULD WALK A CANCER JOURNEY ALONE/LESSONS FROM MYAMERICAN BREAST CANCER WALKAMERICAN CANCER SOCIETY BREAST CANCER WALKBREAST CANCER WALK IN OCTOBERBREAST CANCER WALKSLONG ISLAND BREAST CANCERWALK
In 2020, o ver 276,000 American women and 2,600 men will be newly diagnosed with breast cancer. For these individuals, Breast Cancer Awareness month is especially poignant. Those hearing the words ‘you have cancer’ quickly realize the importance of having a support network as they move through diagnosis, treatment, and survivorship.MEDABLE
Medable is on a mission to get effective therapies to patients faster by transforming clinical drug development with disruptivetechnologies.
ACCESS APP - MEDABLE Medable is on a mission to get effective therapies to patients faster by transforming clinical drug development with disruptivetechnologies.
GARTNER REPORT
At Medable, we’ve long believed in the power of digital cloud solutions. In fact, as a pioneer of and provider of over 100+ decentralized trials, we’ve guided our clients towards lower cost, faster deployment times, and the elimination of data silos. If you’re curious about the onset of “cloud solutions” for clinical development, or have questions about how they can transform TELEVISIT FOR CLINICAL TRIALS A Harvard School of Medicine study, in fact, showed that an in-person doctor visit in the U.S. takes about 121 minutes – and only 20 minutes of that time is spent face-to-face with a doctor.PRIVACY CENTER
Medable is on a mission to get effective therapies to patients faster by transforming clinical drug development with disruptivetechnologies.
INTRODUCTION
Here you'll find comprehensive guides, step-by-step tutorials, and API reference documents to help you get up and going with Medable's products as quickly as possible. CONTACT US - NEED HELP? WE'RE HERE WHEN YOU NEED US We are here to answer your questions Medable HQ T: (877) 820-6259 525 University Ave, Suite A70 Palo Alto, CA 94301 If you are interested in investing in Medable please contact us at: ir@medable.com WHAT IS REMOTE PATIENT MONITORING AND HOW DOES IT WORK Leverage Medable and connected devices to enable remote patient monitoring. Offer your patients a simple way to collect key trial data without interfering in their daily lives.MEDABLE BLOG
Alison (Ali) Holland is Head of Decentralized and Remote trials at Medable and is chartered to design and expand the end-to-end capabilities needed to partially or fully virtualize clinical studies, building on the commitment to reduce clinical trial times by 50%. Ali has over 20 years of experience in a variety of leadership roles, including Feasibility Head and Six Sigma Black Belt at BIAS AND DECENTRALIZED TRIALS: HOW TO AVOID SELECTION BIAS Written by Mary Costello. Mary Costello has spent over thirty years in the healthcare and clinical research fields, working in commercial leadership roles with large organizations like Covance and Thermo Fisher and more recently supporting the growth and expansion of eClinical Solutions and Elligo Health Research.Remove cookies
Webinar:
REDUCE PATIENT BURDEN AND IMPROVE OUTCOMES WITH REAL WORLD DATARegister Today
c
*
Product
Decentralized Trial Platform Digital & DCT ScreeningeConsent
Televisit
eCOA & ePRO
Remote Trial Patient Monitoring BioStat & Data Science*
Company
Leadership Team
Careers
Patient Advisory CouncilLegal
*
Contact
Contact Us
Request a Demo
* Blog
Contact us
Request Demo
DECENTRALIZE RESEARCH WITH FLEXIBILITY Right-size how your protocol is decentralized with our flexible, modular digital and DCT platformRequest a Demo
DIGITAL SCREENING:
RECRUIT PATIENTS AT HOME Enable remote screening at scale, globally Meet your studies enrollment targets faster Develop any screening assessmentLearn More
TELECONSENT:
FLEXIBLE REMOTE INFORMED CONSENT Improve consent readability with responsive layouts on web, mobile, &more
Deliver remote eConsent to patients on par with consumer digitalexperiences
Accommodate local regulations and languages with global flexibilityLearn More
TELEVISIT:
BRING ON-SITE EXPERIENCES HOME Engage patients and remove the burden of time and travel of an on-sitevisit
Improve patient access and engagement Streamline communication with a safe and compliant Trial-Fit Telemedicine solutionLearn More
TELECOA:
CLINICAL OUTCOME ASSESSMENTS ANYWHERE Right size your COA strategy Capture outcome assessments remotely Deploy diaries, ePROs, and more rapidlyLearn More
We know now that when patients are able to access a clinical trial online, using telemedicine and remote consent, most clinical trials will enroll three times faster. That’s an incredible data point that we didn’t know before COVID-19. Dr. Michelle Longmire, CEO of Medable REMOTE PATIENT MONITORING: DCT CONNECTED DEVICES See real-time patient outcome updates Provide context with quality of life data Configure intelligent real-time alertsLearn More
Trusted by
Why Medable?
1M
Over 1 million patients on platform>90%
>90% patient app satisfaction85+
85+ studies across 41+ countries10-14w
Accelerated Deployment of 10-14 weeks Medable is on a mission to get effective therapies to patients faster by transforming clinical drug development with disruptivetechnologies.
PRODUCTS
* DCT Overview
* Digital & DCT Screening* eConsent
* Televisit
* eCOA & ePRO
* Remote Trial Patient Monitoring * BioStat & Data ScienceCOMPANY
* Leadership Team
* Careers
* Patient Advisory Council* Legal
* Privacy
* Blog
CONTACT
* Request a Demo
* Contact Us
Copyright 2021 © Medable PRIVACY PREFERENCE CENTER When you visit any website, it may store or retrieve information on your browser, mostly in the form of cookies. This information might be about you, your preferences or your device and is mostly used to make the site work as you expect it to. The information does not usually directly identify you, but it can give you a more personalized web experience. Because we respect your right to privacy, you can choose not to allow some types of cookies. Click on the different category headings to find out more and change our default settings. However, blocking some types of cookies may impact your experience of the site and the services we are able to offer.More information
Allow All
MANAGE CONSENT PREFERENCES STRICTLY NECESSARY COOKIESAlways Active
These cookies are necessary for the website to function and cannot be switched off in our systems. They are usually only set in response to actions made by you which amount to a request for services, such as setting your privacy preferences, logging in or filling in forms. You can set your browser to block or alert you about these cookies, but some parts of the site will not then work. These cookies do not store any personally identifiable information.Cookies Details
PERFORMANCE COOKIES
Performance Cookies
These cookies allow us to count visits and traffic sources so we can measure and improve the performance of our site. They help us to know which pages are the most and least popular and see how visitors move around the site. All information these cookies collect is aggregated and therefore anonymous. If you do not allow these cookies we will not know when you have visited our site, and will not be able to monitorits performance.
Cookies Details
FUNCTIONAL COOKIES
Functional Cookies
These cookies enable the website to provide enhanced functionality and personalisation. They may be set by us or by third party providers whose services we have added to our pages. If you do not allow these cookies then some or all of these services may not function properly.Cookies Details
TARGETING COOKIES
Targeting Cookies
These cookies may be set through our site by our advertising partners. They may be used by those companies to build a profile of your interests and show you relevant adverts on other sites. They do not store directly personal information, but are based on uniquely identifying your browser and internet device. If you do not allow these cookies, you will experience less targeted advertising.Cookies Details
BACK BUTTON PERFORMANCE COOKIES Vendor Search Search IconFilter Icon
Clear
checkbox label labelApply Cancel
Consent Leg.Interest checkbox label label checkbox label label checkbox label label*
33ACROSS
HOST DESCRIPTION
VIEW COOKIES
*
Name
cookie name
Confirm My Choices
By clicking “Accept All Cookies”, you agree to the storing of cookies on your device to enhance site navigation, analyze site usage, and assist in our marketing efforts. Accept All CookiesCookies SettingsDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0