Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://spotifypremiumapkdl.blogspot.com/
Are you over 18 and want to see adult content?

A complete backup of http://www.alwadifa-maroc.com/
Are you over 18 and want to see adult content?

A complete backup of https://chinaq.me/kr190603c/5.html
Are you over 18 and want to see adult content?
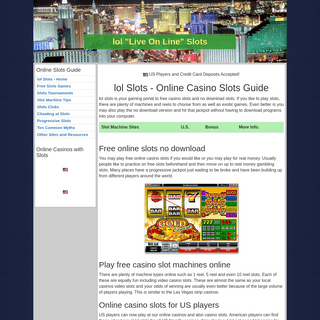
A complete backup of https://www.lolslots.com/
Are you over 18 and want to see adult content?
A complete backup of https://libros.plus/revolucion-en-silicon-valley/
Are you over 18 and want to see adult content?
A complete backup of https://trustscam.nl/zaipoc.com
Are you over 18 and want to see adult content?

A complete backup of https://animekisa.tv/
Are you over 18 and want to see adult content?

A complete backup of https://ballherehere.com/tag/%e0%b9%80%e0%b8%9b%e0%b8%a3%e0%b8%b9/
Are you over 18 and want to see adult content?
A complete backup of http://mpt49.ru.net/v/295954
Are you over 18 and want to see adult content?
Favourite Annotations
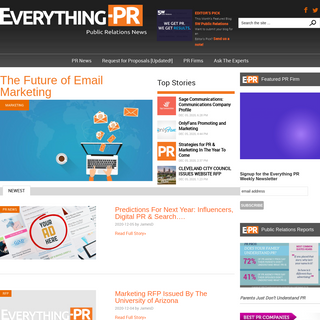
A complete backup of everything-pr.com
Are you over 18 and want to see adult content?
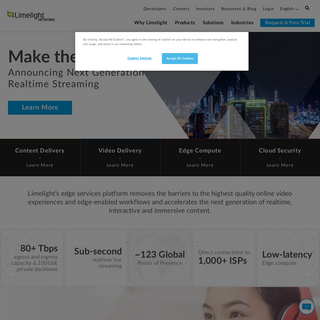
A complete backup of limelightnetworks.com
Are you over 18 and want to see adult content?

A complete backup of popularpittsburgh.com
Are you over 18 and want to see adult content?

A complete backup of magasin-cnac.org
Are you over 18 and want to see adult content?

A complete backup of ideeenbrouwerij.nu
Are you over 18 and want to see adult content?

A complete backup of greatwesternweed.com
Are you over 18 and want to see adult content?
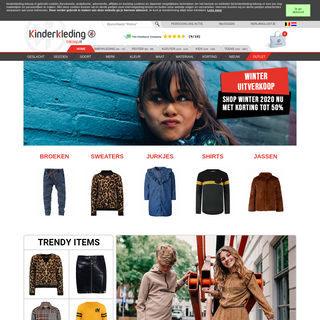
A complete backup of kinderkleding-tekoop.nl
Are you over 18 and want to see adult content?
Text
better able to
EVENTS | ASSOCIATION FOR ACCESSIBLE MEDICINESCONTACT USABOUT USRESOURCESOUR BOARD OF DIRECTORSACCESS! 2019 The Association for Accessible Medicines sponsors a number of events where industry leaders can network, share and learn. Take a looktoday.
ACCESS! 2021 VIRTUAL MEETING AAM’s annual meeting Access! 2021 brings together policymakers, influential speakers and industry leaders to chart the future of generics and biosimilars. We’ve made it easier now than ever before to attend the conference. No matter where you are in the world May 26-27 you will be able to attend. Access! 2021 is a fully interactive ACCESS WEEK IS COMING May 27, 2021 | 3:30 pm – 5:30 pm EDT. AAM Members are invited to attend special programming dedicated to industry communications and digital advocacy as part of Access! 2021 Conference Week. AGENDA | ACCESS! 2021 VIRTUAL CONFERENCE Join industry expert Adam J. Fein, PhD, as he helps you navigate this challenging and dynamic period in U.S. drug channels. During this interactive virtual session, Dr. Fein will explain the key trends reshaping U.S. drug channels, examine the emergence of organized market access channels, highlight business and marketing implications for generic and biosimilar medicines, and discuss CURRENT TRENDS AND BEST PRACTICES IN ANDA LABELING 2. Objectives • Best practices to help improve the quality of labeling submissions • Other tips and clarifications. www.fda.gov POTENTIAL SAVINGS FROM ACCELERATING US APPROVAL OF COMPLEX Potential Savings from Accelerating US Approval of Complex Generics | www.GetMGA.com 6 are available) in Medicare Part D in 2017 was 90.8%, according to the Centers for Medicare ESSENTIAL PERFORMANCE REQUIREMENTS DEMYSTIFIED Essential Performance Requirements Demystified Henri Akouka, P.E. Associate Director, R&D, Teva Pharmaceuticals POST APPROVAL CHANGES: BEST PRACTICES AND 8 Post-Approval Change Regulations •21 CFR 314.70- Supplements and other changes to an approved application. •314.70(a)(1)(i):the applicant must notify FDA about each NAVIGATING Q1/Q2 FOR COMPLEX GENERICS Navigating Q1/Q2 for Complex Generics Generic + Biosimilar Medicines Conference November 4-6, 2019 . Xiaohui (Jeff) Jiang, PhD. Deputy Director. Division of Therapeutic Performance GENERICS & BIOSIMILARS The Association for Accessible Medicines improves access to safe, quality, effective medicine. Better access to medicine is relevant to everybody because, after all, we’re all patients at some point. Health is the foundation for everything in life. Healthy people arebetter able to
EVENTS | ASSOCIATION FOR ACCESSIBLE MEDICINESCONTACT USABOUT USRESOURCESOUR BOARD OF DIRECTORSACCESS! 2019 The Association for Accessible Medicines sponsors a number of events where industry leaders can network, share and learn. Take a looktoday.
ACCESS! 2021 VIRTUAL MEETING AAM’s annual meeting Access! 2021 brings together policymakers, influential speakers and industry leaders to chart the future of generics and biosimilars. We’ve made it easier now than ever before to attend the conference. No matter where you are in the world May 26-27 you will be able to attend. Access! 2021 is a fully interactive ACCESS WEEK IS COMING May 27, 2021 | 3:30 pm – 5:30 pm EDT. AAM Members are invited to attend special programming dedicated to industry communications and digital advocacy as part of Access! 2021 Conference Week. AGENDA | ACCESS! 2021 VIRTUAL CONFERENCE Join industry expert Adam J. Fein, PhD, as he helps you navigate this challenging and dynamic period in U.S. drug channels. During this interactive virtual session, Dr. Fein will explain the key trends reshaping U.S. drug channels, examine the emergence of organized market access channels, highlight business and marketing implications for generic and biosimilar medicines, and discuss CURRENT TRENDS AND BEST PRACTICES IN ANDA LABELING 2. Objectives • Best practices to help improve the quality of labeling submissions • Other tips and clarifications. www.fda.gov POTENTIAL SAVINGS FROM ACCELERATING US APPROVAL OF COMPLEX Potential Savings from Accelerating US Approval of Complex Generics | www.GetMGA.com 6 are available) in Medicare Part D in 2017 was 90.8%, according to the Centers for Medicare ESSENTIAL PERFORMANCE REQUIREMENTS DEMYSTIFIED Essential Performance Requirements Demystified Henri Akouka, P.E. Associate Director, R&D, Teva Pharmaceuticals POST APPROVAL CHANGES: BEST PRACTICES AND 8 Post-Approval Change Regulations •21 CFR 314.70- Supplements and other changes to an approved application. •314.70(a)(1)(i):the applicant must notify FDA about each NAVIGATING Q1/Q2 FOR COMPLEX GENERICS Navigating Q1/Q2 for Complex Generics Generic + Biosimilar Medicines Conference November 4-6, 2019 . Xiaohui (Jeff) Jiang, PhD. Deputy Director. Division of Therapeutic Performance ACQUISITIONS/LICENSING AND MERGERS WHAT ARE THE REGULATORY • New and former owners of application are required to submit notification of the transfer to FDA (see 21 CFR 314.72) • Former owner must state that all rights to the application have POST APPROVAL CHANGES: BEST PRACTICES AND 8 Post-Approval Change Regulations •21 CFR 314.70- Supplements and other changes to an approved application. •314.70(a)(1)(i):the applicant must notify FDA about each DRUG-DEVICE COMBINATION PRODUCTS QUALITY ASSESSMENT Drug-Device Combination Products Quality Assessment Ashley B. Boam, MSBE Director. Office of Policy for Pharmaceutical Quality. CDER/OPQ.GRx+Biosims 2019
CGMP TRENDS, DATA INTEGRITY AND HANDLING AN FDA INSPECTION Title: CGMP Trends, Data Integrity, and Handling an Inspection Mark Schwartz Director Hyman, Phelps & McNamara P.C. Author: Scott I. Goldman Created Date REMS AND RESTRICTED DISTRIBUTION PROGRAMS Alex Brill June 2017 The sustained concern over high drug prices has led policymakers to identify undue barriers to generic entry as a contributing factor. One such barrier is brand drug manufacturers’ misuse of Risk Evaluation and Mitigation Strategy (REMS) and other restricted access programs to block generic competition. This study explains how REMS programs and other strategies PHARMACEUTICAL PATENT ABUSE: TO INFINITY AND BEYOND Pharmaceutical Patent Abuse: To Infinity and Beyond! Our pharmaceutical industry in the United States is predicated on a balance between innovation and access. Brand pharmaceutical companies are rewarded for inventing and developing new treatments and cures that improve the quality of life for everyone. In return for the innovation, current law PRESCRIPTION DRUG & OPIOID EDUCATION PROGRAM 0:00. 0:00 / 1:43. Live. •. In response to this crisis, last year AAM launched the Prescription Drug Safety Network, an evidence-based curriculum last year to help prevent abuse on campus—and it’s free to any college in America. Created with EVERFI, a leader in digital learning and prevention education solutions , the curriculum equipsOUR PRESS RELEASES
Press Releases. Press Release. May 26, 2021 | Rachel Schwartz. Black Women’s Health Imperative Named AAM’s 2021 “Champion of Access”. Press Release. May 24, 2021 | Erica Klinger. AAM Names Erik Komendant Senior Vice President, Government Affairs. Press Release.May 24, 2021 |
AAM RESPONSE TO CA GOVERNOR NEWSOM SIGNING AB 824 AAM Response to CA Governor Newsom Signing AB 824. Share. WASHINGTON, DC (October 8, 2019)--. "As the Federal Trade Commission has found, recent patent litigation settlements have overwhelmingly accelerated the launch of more affordable generic and biosimilar medicines prior to the branded drug's patent expiration date.JOHN DUCKER
John Ducker is President and Chief Executive Officer of Fresenius Kabi USA, and a member of the Managing Board of its parent company, Fresenius Kabi AG. He is responsible for the company’s strategy and commercial operations in the United States and Canada. John joined Fresenius Kabi in 1989 and became a member of the board of Fresenius Kabi AG in 2006 and has been in his current role since 2010. GENERICS & BIOSIMILARS The Association for Accessible Medicines works to ensure more generic and biosimilar medicines are accessible to more people who need them. LISA STEELMAN JOINS AAM AS VICE PRESIDENT, STATE WASHINGTON, DC (August 10, 2020) — Today Lisa Steelman is joining the Association for Accessible Medicines (AAM). As Vice President, State Government Affairs, she serves as a senior strategist and lobbyist in partnership with the Senior Vice President, Government Affairs, Chris Bowlin, and other members of the State Government Affairs team to represent AAM and its member CURRENT TRENDS AND BEST PRACTICES IN ANDA LABELING 2. Objectives • Best practices to help improve the quality of labeling submissions • Other tips and clarifications. www.fda.gov POTENTIAL SAVINGS FROM ACCELERATING US APPROVAL OF COMPLEX Potential Savings from Accelerating US Approval of Complex Generics | www.GetMGA.com 6 are available) in Medicare Part D in 2017 was 90.8%, according to the Centers for Medicare DRUG-DEVICE COMBINATION PRODUCTS QUALITY ASSESSMENT Drug-Device Combination Products Quality Assessment Ashley B. Boam, MSBE Director. Office of Policy for Pharmaceutical Quality. CDER/OPQ.GRx+Biosims 2019
ACQUISITIONS/LICENSING AND MERGERS WHAT ARE THE REGULATORY Acquisitions/Licensing and Mergers What Are the Regulatory Requirements & Strategies to Consider? Scott D. Tomsky VP, Regulatory Affairs, Generics, North America,JOHN DUCKER
John Ducker is President and Chief Executive Officer of Fresenius Kabi USA, and a member of the Managing Board of its parent company, Fresenius Kabi AG. He is responsible for the company’s strategy and commercial operations in the United States and Canada. John joined Fresenius Kabi in 1989 and became a member of the board of Fresenius Kabi AG in 2006 and has been in his current role since 2010. ESSENTIAL PERFORMANCE REQUIREMENTS DEMYSTIFIED Essential Performance Requirements Demystified Henri Akouka, P.E. Associate Director, R&D, Teva Pharmaceuticals CGMP TRENDS, DATA INTEGRITY AND HANDLING AN FDA INSPECTION Title: CGMP Trends, Data Integrity, and Handling an Inspection Mark Schwartz Director Hyman, Phelps & McNamara P.C. Author: Scott I. Goldman Created Date LEGAL ISSUES IN GDUFA, FDARA, AND ADMINISTRATION PROPOSALS 16 FDARA Section 808 (FDC Act 505(j)) •180-day exclusivity period for CGT’s— –(I) if the application is for a drug that is the same as a CGT for which any first approved applicant has commenced commercial marketing, the application shall be made GENERICS & BIOSIMILARS The Association for Accessible Medicines works to ensure more generic and biosimilar medicines are accessible to more people who need them. LISA STEELMAN JOINS AAM AS VICE PRESIDENT, STATE WASHINGTON, DC (August 10, 2020) — Today Lisa Steelman is joining the Association for Accessible Medicines (AAM). As Vice President, State Government Affairs, she serves as a senior strategist and lobbyist in partnership with the Senior Vice President, Government Affairs, Chris Bowlin, and other members of the State Government Affairs team to represent AAM and its member CURRENT TRENDS AND BEST PRACTICES IN ANDA LABELING 2. Objectives • Best practices to help improve the quality of labeling submissions • Other tips and clarifications. www.fda.gov POTENTIAL SAVINGS FROM ACCELERATING US APPROVAL OF COMPLEX Potential Savings from Accelerating US Approval of Complex Generics | www.GetMGA.com 6 are available) in Medicare Part D in 2017 was 90.8%, according to the Centers for Medicare DRUG-DEVICE COMBINATION PRODUCTS QUALITY ASSESSMENT Drug-Device Combination Products Quality Assessment Ashley B. Boam, MSBE Director. Office of Policy for Pharmaceutical Quality. CDER/OPQ.GRx+Biosims 2019
ACQUISITIONS/LICENSING AND MERGERS WHAT ARE THE REGULATORY Acquisitions/Licensing and Mergers What Are the Regulatory Requirements & Strategies to Consider? Scott D. Tomsky VP, Regulatory Affairs, Generics, North America,JOHN DUCKER
John Ducker is President and Chief Executive Officer of Fresenius Kabi USA, and a member of the Managing Board of its parent company, Fresenius Kabi AG. He is responsible for the company’s strategy and commercial operations in the United States and Canada. John joined Fresenius Kabi in 1989 and became a member of the board of Fresenius Kabi AG in 2006 and has been in his current role since 2010. ESSENTIAL PERFORMANCE REQUIREMENTS DEMYSTIFIED Essential Performance Requirements Demystified Henri Akouka, P.E. Associate Director, R&D, Teva Pharmaceuticals CGMP TRENDS, DATA INTEGRITY AND HANDLING AN FDA INSPECTION Title: CGMP Trends, Data Integrity, and Handling an Inspection Mark Schwartz Director Hyman, Phelps & McNamara P.C. Author: Scott I. Goldman Created Date LEGAL ISSUES IN GDUFA, FDARA, AND ADMINISTRATION PROPOSALS 16 FDARA Section 808 (FDC Act 505(j)) •180-day exclusivity period for CGT’s— –(I) if the application is for a drug that is the same as a CGT for which any first approved applicant has commenced commercial marketing, the application shall be made GENERICS & BIOSIMILARS The Association for Accessible Medicines works to ensure more generic and biosimilar medicines are accessible to more people who need them. AAM STATEMENT ON BIDEN-HARRIS ADMINISTRATION’S 100-DAY WASHINGTON DC (June 8, 2021) – The Association for Accessible Medicines (AAM) supports the Biden-Harris administration’s 100-day review of the pharmaceutical and active pharmaceutical ingredient (API) supply chains. The Biden-Harris administration has produced a thoughtful plan to strengthen the security of the U.S. pharmaceutical supply chain that recognizes the importance of engaging ACCESS WEEK IS COMING May 27, 2021 | 3:30 pm – 5:30 pm EDT. AAM Members are invited to attend special programming dedicated to industry communications and digital advocacy as part of Access! 2021 Conference Week. A RESPECTFUL STYLE: WIHP’S FIRESIDE CHAT WITH Powered by AAM, Women in Health Policy (WiHP) now has more than 700 members in our LinkedIn community. Online and (when possible) in person, we get together to share experiences and perspectives and support each other as we navigate our careers. ACCESS! 2021 VIRTUAL MEETING We’ve made it easier now than ever before to attend the conference. No matter where you are in the world May 26-27 you will be able to attend. Access! 2021 is a fully interactive conference with the ability to chat with other conference attendees, speakers andexhibitors.
AGENDA | ACCESS! 2021 VIRTUAL CONFERENCE Join industry expert Adam J. Fein, PhD, as he helps you navigate this challenging and dynamic period in U.S. drug channels. During this interactive virtual session, Dr. Fein will explain the key trends reshaping U.S. drug channels, examine the emergence of organized market access channels, highlight business and marketing implications for generic and biosimilar medicines, and discuss AAM STATEMENT ON RESIGNATION OF CHIP DAVIS WASHINGTON, DC (February 10, 2020)- The Association for Accessible Medicines (AAM), the trade association representing the manufacturers of generic and biosimilar medicines, announced today that Chester “Chip” Davis, Jr. is leaving the organization.. Jeffrey Francer, AAM Senior Vice President and General Counsel, will serve as interim CEO while the board determines a permanent replacement. TELL CONGRESS TO OPPOSE THE BLOCKING ACT The Lower Health Care Costs Act of 2019 is a big giveaway to Big Pharma as it protects their high-priced monopolies. If you are concerned about the ever-rising price of prescription medicines, contact your Senator now and urge them to BLOCK 205. EMERGING TECHNOLOGY PROGRAM Emerging Technology Program Sau (Larry) Lee, Director, Office of Testing and Research, OPQ GRx + Biosims 2019. US FDA Center for Drug Evaluation and Research PRE-ASSIGNMENT PORTAL 7. WHO IS USING THE PORTAL? The Portal is the now the preferred method for submitting pre- assignment requests for NDAs, BLAs, ANDAs, INDs and MFs to request a pre- assignment number. GENERICS & BIOSIMILARS The Association for Accessible Medicines improves access to safe, quality, effective medicine. Better access to medicine is relevant to everybody because, after all, we’re all patients at some point. Health is the foundation for everything in life. Healthy people arebetter able to
EVENTS | ASSOCIATION FOR ACCESSIBLE MEDICINESCONTACT USABOUT USRESOURCESOUR BOARD OF DIRECTORSACCESS! 2019 The Association for Accessible Medicines sponsors a number of events where industry leaders can network, share and learn. Take a looktoday.
LISA STEELMAN JOINS AAM AS VICE PRESIDENT, STATE WASHINGTON, DC (August 10, 2020) — Today Lisa Steelman is joining the Association for Accessible Medicines (AAM). As Vice President, State Government Affairs, she serves as a senior strategist and lobbyist in partnership with the Senior Vice President, Government Affairs, Chris Bowlin, and other members of the State Government Affairs team to represent AAM and its member CURRENT TRENDS AND BEST PRACTICES IN ANDA LABELING 2. Objectives • Best practices to help improve the quality of labeling submissions • Other tips and clarifications. www.fda.gov SKYROCKETING DRUG PRICES: WHAT'S DRIVING UP COSTS A recent report shows the average annual price of specialty drugs has tripled over the last ten years from nearly $18,000 to more than $52,000 today.1. With brand-name drugs now accounting for 77 percent of all spending on prescription drugs, patients are experiencing higher pharmacy costs, higher premiums and higher deductibles as aresult of
AAM RESPONSE TO CA GOVERNOR NEWSOM SIGNING AB 824 AAM Response to CA Governor Newsom Signing AB 824. Share. WASHINGTON, DC (October 8, 2019)--. "As the Federal Trade Commission has found, recent patent litigation settlements have overwhelmingly accelerated the launch of more affordable generic and biosimilar medicines prior to the branded drug's patent expiration date. DRUG-DEVICE COMBINATION PRODUCTS QUALITY ASSESSMENT Drug-Device Combination Products Quality Assessment Ashley B. Boam, MSBE Director. Office of Policy for Pharmaceutical Quality. CDER/OPQ.GRx+Biosims 2019
ACQUISITIONS/LICENSING AND MERGERS WHAT ARE THE REGULATORY Acquisitions/Licensing and Mergers What Are the Regulatory Requirements & Strategies to Consider? Scott D. Tomsky VP, Regulatory Affairs, Generics, North America, ESSENTIAL PERFORMANCE REQUIREMENTS DEMYSTIFIEDESSENTIAL PERFORMANCE60601
Essential Performance Requirements Demystified Henri Akouka, P.E. Associate Director, R&D, Teva Pharmaceuticals LEGAL ISSUES IN GDUFA, FDARA, AND ADMINISTRATION PROPOSALS 16 FDARA Section 808 (FDC Act 505(j)) •180-day exclusivity period for CGT’s— –(I) if the application is for a drug that is the same as a CGT for which any first approved applicant has commenced commercial marketing, the application shall be made GENERICS & BIOSIMILARS The Association for Accessible Medicines improves access to safe, quality, effective medicine. Better access to medicine is relevant to everybody because, after all, we’re all patients at some point. Health is the foundation for everything in life. Healthy people arebetter able to
EVENTS | ASSOCIATION FOR ACCESSIBLE MEDICINESCONTACT USABOUT USRESOURCESOUR BOARD OF DIRECTORSACCESS! 2019 The Association for Accessible Medicines sponsors a number of events where industry leaders can network, share and learn. Take a looktoday.
LISA STEELMAN JOINS AAM AS VICE PRESIDENT, STATE WASHINGTON, DC (August 10, 2020) — Today Lisa Steelman is joining the Association for Accessible Medicines (AAM). As Vice President, State Government Affairs, she serves as a senior strategist and lobbyist in partnership with the Senior Vice President, Government Affairs, Chris Bowlin, and other members of the State Government Affairs team to represent AAM and its member CURRENT TRENDS AND BEST PRACTICES IN ANDA LABELING 2. Objectives • Best practices to help improve the quality of labeling submissions • Other tips and clarifications. www.fda.gov SKYROCKETING DRUG PRICES: WHAT'S DRIVING UP COSTS A recent report shows the average annual price of specialty drugs has tripled over the last ten years from nearly $18,000 to more than $52,000 today.1. With brand-name drugs now accounting for 77 percent of all spending on prescription drugs, patients are experiencing higher pharmacy costs, higher premiums and higher deductibles as aresult of
AAM RESPONSE TO CA GOVERNOR NEWSOM SIGNING AB 824 AAM Response to CA Governor Newsom Signing AB 824. Share. WASHINGTON, DC (October 8, 2019)--. "As the Federal Trade Commission has found, recent patent litigation settlements have overwhelmingly accelerated the launch of more affordable generic and biosimilar medicines prior to the branded drug's patent expiration date. DRUG-DEVICE COMBINATION PRODUCTS QUALITY ASSESSMENT Drug-Device Combination Products Quality Assessment Ashley B. Boam, MSBE Director. Office of Policy for Pharmaceutical Quality. CDER/OPQ.GRx+Biosims 2019
ACQUISITIONS/LICENSING AND MERGERS WHAT ARE THE REGULATORY Acquisitions/Licensing and Mergers What Are the Regulatory Requirements & Strategies to Consider? Scott D. Tomsky VP, Regulatory Affairs, Generics, North America, ESSENTIAL PERFORMANCE REQUIREMENTS DEMYSTIFIEDESSENTIAL PERFORMANCE60601
Essential Performance Requirements Demystified Henri Akouka, P.E. Associate Director, R&D, Teva Pharmaceuticals LEGAL ISSUES IN GDUFA, FDARA, AND ADMINISTRATION PROPOSALS 16 FDARA Section 808 (FDC Act 505(j)) •180-day exclusivity period for CGT’s— –(I) if the application is for a drug that is the same as a CGT for which any first approved applicant has commenced commercial marketing, the application shall be made PRESCRIPTION DRUG & OPIOID EDUCATION PROGRAM 0:00. 0:00 / 1:43. Live. •. In response to this crisis, last year AAM launched the Prescription Drug Safety Network, an evidence-based curriculum last year to help prevent abuse on campus—and it’s free to any college in America. Created with EVERFI, a leader in digital learning and prevention education solutions , the curriculum equipsOUR PRESS RELEASES
Press Releases. Press Release. May 26, 2021 | Rachel Schwartz. Black Women’s Health Imperative Named AAM’s 2021 “Champion of Access”. Press Release. May 24, 2021 | Erica Klinger. AAM Names Erik Komendant Senior Vice President, Government Affairs. Press Release.May 24, 2021 |
ADVOCACY | ASSOCIATION FOR ACCESSIBLE MEDICINES Speak Up for Access Health care is an issue we all care about, and all of us—and those we love—will need medicine one day. AAM encourages voters and patients like you to advocate at the federal and state level for accessible prescription drugs. Your voice and your storiesmake a
LISA STEELMAN JOINS AAM AS VICE PRESIDENT, STATE WASHINGTON, DC (August 10, 2020) — Today Lisa Steelman is joining the Association for Accessible Medicines (AAM). As Vice President, State Government Affairs, she serves as a senior strategist and lobbyist in partnership with the Senior Vice President, Government Affairs, Chris Bowlin, and other members of the State Government Affairs team to represent AAM and its member AAM STATEMENT ON RESIGNATION OF CHIP DAVIS WASHINGTON, DC (February 10, 2020)- The Association for Accessible Medicines (AAM), the trade association representing the manufacturers of generic and biosimilar medicines, announced today that Chester “Chip” Davis, Jr. is leaving the organization.. Jeffrey Francer, AAM Senior Vice President and General Counsel, will serve as interim CEO while the board determines a permanent replacement. OUR BOARD OF DIRECTORS Learn more about the Association for Accessible Medicines. Click here to view the AAM Board of Directors. AGENDA | ACCESS! 2021 VIRTUAL CONFERENCE Join industry expert Adam J. Fein, PhD, as he helps you navigate this challenging and dynamic period in U.S. drug channels. During this interactive virtual session, Dr. Fein will explain the key trends reshaping U.S. drug channels, examine the emergence of organized market access channels, highlight business and marketing implications for generic and biosimilar medicines, and discussCHRISTINE SIMMON
Christine Simmon joined the Association for Accessible Medicines (AAM) in 2012 as the Senior Vice President of Policy & Strategic Alliances. She is Executive Director of AAM’s Biosimilars Council.. In her current role, Christine is responsible for leading policy development and issues management for AAM, directing the Biosimilars Council, and building relationships with strategicVINCE MANCINELLI II
Vince Mancinelli is the President of Rhodes Pharmaceuticals L.P. and has over 30 years of leadership experience in the pharmaceutical industry. Previously, he was a co-founder and Chief Operating Officer of GenPak Solutions, a specialty pharmaceutical packaging company. He held various senior executive roles at Mylan, including Head of North American Operations and Executive Vice-President and ESSENTIAL PERFORMANCE REQUIREMENTS DEMYSTIFIED Essential Performance Requirements Demystified Henri Akouka, P.E. Associate Director, R&D, Teva PharmaceuticalsClose
* Toggle submenu (Advocacy)Advocacy* Issue Overview
* Prescription for Savings * Sustainable Supply Chain* Biosimilars
* IP & Patent Reform * Medicaid Generics Penalty* State Legislation
* Trade
* Toggle submenu (Events)Events* Access! 2021
* GRx+Biosims 2021
* Our Impact
* Stories
* Toggle submenu (About Us)About Us* Generic Quality
* About the Association* Our Team
* Our Members
* Meet Our Board
* Social Commitment
* Generic Medicines
* Biosimilars
* Careers
* Contact Us
* Toggle submenu (Resources)Resources* Search Resources
* 2020 Access and Savings Report * Supply Chain Blueprint * Industry Response to COVID-19* Press Releases
* Blog
* State Lawmaker Toolkit* Amicus Briefs
* Join
* Take Action
Skip to main navigationClose
MAIN NAVIGATION
* Advocacy
* Issue Overview
* Prescription for Savings * Sustainable Supply Chain* Biosimilars
* IP & Patent Reform * Medicaid Generics Penalty* State Legislation
* Trade
* Events
* Our Impact
* Stories
* About Us
* Generic Quality
* About the Association* Our Team
* Our Members
* Meet Our Board
* Social Commitment
* Generic Medicines
* Biosimilars
* Careers
* Contact Us
* Resources
* Search Resources
* 2020 Access and Savings Report * Supply Chain Blueprint * Industry Response to COVID-19* Press Releases
* Blog
* State Lawmaker Toolkit* Amicus Briefs
* Join
* Take Action
Menu
ICYMI - View memorable moments and quotes from #Access2021Learn More
Close
*
Congress Can Help to Secure Our Meds Generic and biosimilar medicines are essential to treating millions of patients. Together, we can take steps to secure patient access no matter what challenges may arise.Learn More
*
While We Await Vaccine Progress, We Can Trust in Generic Drugs Read AAM CEO Dan Leonard's article on the essential role generic drugs play during the COVID-19, while we await a vaccine.Read Blog
*
Health Care Savings, State by State The average state saved $6.1 billion from the use of generic drugs in 2019. How much did your state save?Read Blog
*
Industry Response to COVID-19 The coronavirus (COVID-19) pandemic is disrupting everyday life around the world and imposing unprecedented stress on our health care system. Here are some responses and resources from the U.S. generics and biosimilars industry.Learn More
* 1
* 2
* 3
* 4
* Previous
* Next
WORKING TO LEAVE A POSITIVE IMPACT ON... Industry We promote competition in the pharmaceutical industry by lowering barriers to entry for manufacturers of generic and biosimilar medicines and combatting anti-competitive abuses by some brand drug companies who put profits before patients. LEARN MORE People We measure our success as an industry by the lives we impact. We work to protect the low cost of generic and biosimilar medicines and to drive down the cost of developing new ones, expanding access to as many people as possible. LEARN MORE Policy We are a trusted partner and established authority within the pharmaceutical field. We provide accurate, timely data to educate policymakers and promote general public knowledge regarding the positive impact of affordable generic and biosimilar medicines.LEARN MORE
ABOUT US
The Association for Accessible Medicines improves access to safe, quality, effective medicine. Better access to medicine is relevant to everybody because, after all, we’re all patients at some point. Health is the foundation for everything in life. Healthy people are better able to reach their full potential. Generics and biosimilars help more people in more places live healthier and longer.Who We Are
RECENT UPDATES
View All
Amicus Briefs
Blog
Fact Sheet
Presentation
Press Release
Reports
Tweets
Videos
Blog
JUN 3, 2021 | Erica Klinger Access! Recap: Day OnePress Release
MAY 26, 2021 | Rachel Schwartz Black Women’s Health Imperative Named AAM’s 2021 “Champion ofAccess”
Blog
MAY 26, 2021 | Erica Klinger Akaimi Is Leading a Creative LifestyleBlog
MAY 25, 2021 | Jewel Smith Proudly Serving American Heroes: AAM Associate Member Golden StateMedical Supply
Report
MAY 24, 2021 | Erica Klinger AAM Voices of AccessBlog
MAY 24, 2021 | Erica Klinger Rich Has the Wind at His Back* Load More
Generics are a proven prescription for a healthier society. Generic and biosimilar medicines make it easier and more affordable for patients to live healthier lives. Learn More ASSOCIATION FOR ACCESSIBLE MEDICINES 601 New Jersey Ave NW, Suite 850Washington DC 20001
202.249.7100
info@accessiblemeds.orgMAIN NAVIGATION
* Advocacy
* Events
* Our Impact
* Stories
* About Us
* Resources
* Join
* Take Action
� 2020 Association for Accessible Medicines (AAM), All RightsReserved
Privacy Policy | Terms of Use| Contact
SIGN-UP FOR UPDATES
Receive relevant industry news, event information and the latest resources on biosimilars and generic medicines.Subscribe
STAY CONNECTED
For the latest updates, follow us on social media.__
__
__
__
__
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0