Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://www.plurk.com/p/1lvgdd#!
Are you over 18 and want to see adult content?

A complete backup of https://lmsstudentresources.weebly.com/
Are you over 18 and want to see adult content?

A complete backup of https://www.himemix.com/2020/12/no946_sachika01/
Are you over 18 and want to see adult content?
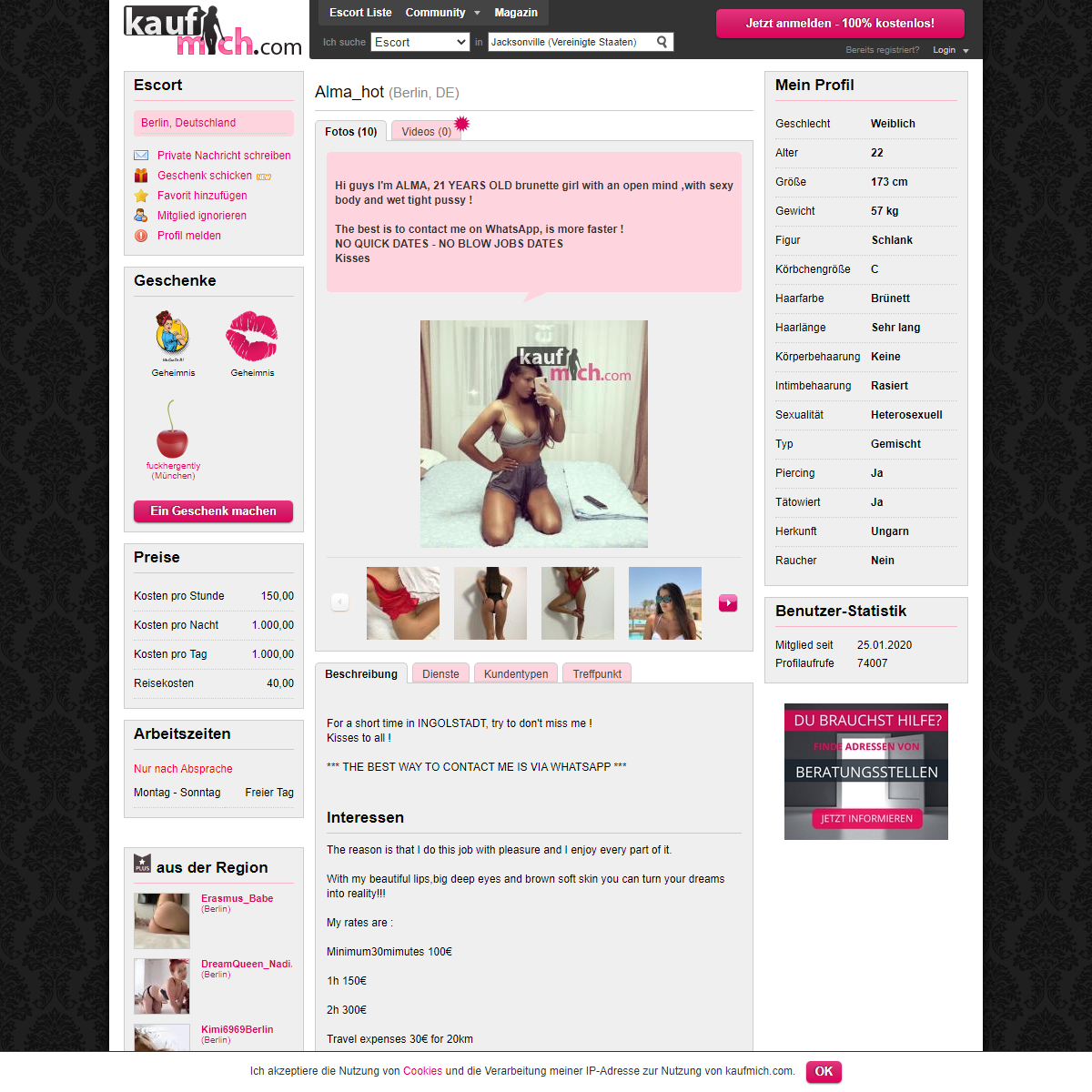
A complete backup of https://www.kaufmich.com/Alma_hot
Are you over 18 and want to see adult content?

A complete backup of https://anaktaeng.blogspot.com/2011/09/chrisye.html
Are you over 18 and want to see adult content?

A complete backup of https://www.lighthousepubjc.com/
Are you over 18 and want to see adult content?

A complete backup of https://website.informer.com/inox-weber.de
Are you over 18 and want to see adult content?
Favourite Annotations
A complete backup of liveswinburneeduau-my.sharepoint.com
Are you over 18 and want to see adult content?
A complete backup of lafootballmagazine.com
Are you over 18 and want to see adult content?
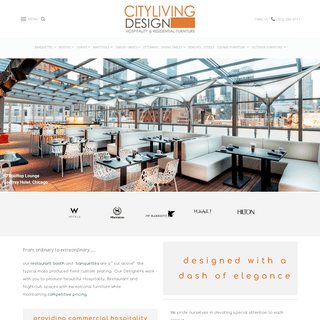
A complete backup of citylivingdesign.com
Are you over 18 and want to see adult content?
A complete backup of online-ucak-bileti.com
Are you over 18 and want to see adult content?
A complete backup of santiagonzalez.wordpress.com
Are you over 18 and want to see adult content?

A complete backup of c-force-streetviewer.com
Are you over 18 and want to see adult content?
Text
FOUNDATION MEDICINE
Foundation Medicine is a world-leading molecular insights company, connecting physicians, patients and biopharma partners to the latest insights in cancer genomics. Learn more.FOUNDATIONONE CDX
FoundationOne®CDx is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors and is for prescription use only.The test analyzes 324 genes as well as genomic signatures including microsatellite instability (MSI) and tumor mutational burden (TMB) and is a companion diagnostic to identify patients who may benefit from treatment with LICENSES | FOUNDATION MEDICINE All Foundation Medicine comprehensive genomic profiling tests are CAP Accredited, CLIA Certified, and New York State Department of Health (NYSDOH) Approved. View our permits and accreditations. FOUNDATIONONE LIQUID CDX FoundationOne®Liquid CDx is for prescription use only and is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors.The test analyzes 324 genes utilizing circulating cell-free DNA and is FDA-approved to report short variants in 311 genes and as a companion diagnostic to identify patients who may benefit from treatment with BILLING AND REIMBURSEMENT The Foundation Medicine FoundationAccess™ program supports providers and their patients through the coverage and billing process. For each comprehensive genomic profiling test ordered, we complete a benefits investigation and reach out to patients whom we expect may have out ofpocket costs.
2021 ASCO ANNUAL MEETING We’re excited to share our work in advancing precision medicine in oncology at this year’s American Society of Clinical Oncology (ASCO) Annual Meeting. Learn more.ADMIN | IAM
Welcome Back. Sign into your account. Email Address. PasswordPRITI HEGDE, PHD
Priti Hegde, Ph.D., joined Foundation Medicine as the Chief Scientific Officer in August 2019. Dr. Hegde oversees clinical product development, cancer genomics, and early-stage research, as well as regulatory and quality assurance, to accelerate advancement of the Company’s leading comprehensive genomic profiling portfolio. FOUNDATION MEDICINE LAUNCHES PRECISION MEDICINE EXCHANGE CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Foundation Medicine, Inc. (NASDAQ:FMI) today announced the launch of its precision medicine partner program, Precision Medicine Exchange Consortium™ (PMEC), to facilitate data exchange, advance research, and support education and applications of precision medicine in oncology and molecularpathology.
FOUNDATION MEDICINE ANNOUNCES KEY DEVELOPMENTS IN Company Receives CE Mark Approval for FoundationOne ®, Adds New Distribution Partner and Appoints Experienced Executive to Lead International Expansion. CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Foundation Medicine, Inc. (NASDAQ: FMI) today announced that it has obtained a CE mark for FoundationOne ®.The CE mark allows FoundationOne to be placed on the market in the EuropeanFOUNDATION MEDICINE
Foundation Medicine is a world-leading molecular insights company, connecting physicians, patients and biopharma partners to the latest insights in cancer genomics. Learn more.FOUNDATIONONE CDX
FoundationOne®CDx is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors and is for prescription use only.The test analyzes 324 genes as well as genomic signatures including microsatellite instability (MSI) and tumor mutational burden (TMB) and is a companion diagnostic to identify patients who may benefit from treatment with LICENSES | FOUNDATION MEDICINE All Foundation Medicine comprehensive genomic profiling tests are CAP Accredited, CLIA Certified, and New York State Department of Health (NYSDOH) Approved. View our permits and accreditations. FOUNDATIONONE LIQUID CDX FoundationOne®Liquid CDx is for prescription use only and is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors.The test analyzes 324 genes utilizing circulating cell-free DNA and is FDA-approved to report short variants in 311 genes and as a companion diagnostic to identify patients who may benefit from treatment with BILLING AND REIMBURSEMENT The Foundation Medicine FoundationAccess™ program supports providers and their patients through the coverage and billing process. For each comprehensive genomic profiling test ordered, we complete a benefits investigation and reach out to patients whom we expect may have out ofpocket costs.
2021 ASCO ANNUAL MEETING We’re excited to share our work in advancing precision medicine in oncology at this year’s American Society of Clinical Oncology (ASCO) Annual Meeting. Learn more.ADMIN | IAM
Welcome Back. Sign into your account. Email Address. PasswordPRITI HEGDE, PHD
Priti Hegde, Ph.D., joined Foundation Medicine as the Chief Scientific Officer in August 2019. Dr. Hegde oversees clinical product development, cancer genomics, and early-stage research, as well as regulatory and quality assurance, to accelerate advancement of the Company’s leading comprehensive genomic profiling portfolio. FOUNDATION MEDICINE LAUNCHES PRECISION MEDICINE EXCHANGE CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Foundation Medicine, Inc. (NASDAQ:FMI) today announced the launch of its precision medicine partner program, Precision Medicine Exchange Consortium™ (PMEC), to facilitate data exchange, advance research, and support education and applications of precision medicine in oncology and molecularpathology.
FOUNDATION MEDICINE ANNOUNCES KEY DEVELOPMENTS IN Company Receives CE Mark Approval for FoundationOne ®, Adds New Distribution Partner and Appoints Experienced Executive to Lead International Expansion. CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Foundation Medicine, Inc. (NASDAQ: FMI) today announced that it has obtained a CE mark for FoundationOne ®.The CE mark allows FoundationOne to be placed on the market in the European BILLING AND REIMBURSEMENT The Foundation Medicine FoundationAccess™ program supports providers and their patients through the coverage and billing process. For each comprehensive genomic profiling test ordered, we complete a benefits investigation and reach out to patients whom we expect may have out ofpocket costs.
FOUNDATIONONE LIQUID CDX FoundationOne®Liquid CDx is for prescription use only and is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors.The test analyzes 324 genes utilizing circulating cell-free DNA and is FDA-approved to report short variants in 311 genes and as a companion diagnostic to identify patients who may benefit from treatment withORDER A TEST
Foundation Medicine tests must be ordered by a licensed physician. For faster order processing, login or sign up for a Foundation Medicine online account. Orders can also be submitted via email or via fax to +1 (617) 418-2290 using our Test Requisition Form.. If sending a solid tumor specimen, Foundation Medicine can complete the specimen retrieval process if requested on the order formFOUNDATIONONE HEME
FoundationOne Heme is a comprehensive genomic profiling (CGP) test combining DNA and RNA sequencing for patients with hematologic malignancies, sarcomas or solid tumors where RNA sequencing is desired. FoundationOne Heme detects known, novel, and complex fusion events as well as other common genomic alterations (substitutions,indels, and CNVs).
PATIENT FAQS
The testing offered by Foundation Medicine is a type of genomic testing called comprehensive genomic profiling (CGP). It looks for mutations in over 300 genes in the tumor with one sample. The mutations in the tumor are what may impact tumor growth, spread, and response to therapy. Foundation Medicine testing may identify bothkinds of
2021 ASCO ANNUAL MEETING FoundationOne Liquid CDx. FoundationOne®Liquid CDx, the broadest FDA-approved liquid biopsy, is now the only blood-based comprehensive genomic profiling companion diagnostic (CDx) for Lynparza® (olaparib)in prostate cancer.
IHC TESTING
PD-L1 IHC Testing at Foundation Medicine. The results of PD-L1 IHC testing combined with CGP results, including data on tumor mutational burden (TMB) and microsatellite instability (MSI), may help you make more informed treatment decisions regarding use of immunotherapies and enrollment in appropriate clinical trials for your patients FOUNDATION MEDICINE LAUNCHES FOUNDATIONONE®TRACKER CTDNA Personalized technology aims to address both early- and advanced-stage cancer monitoring applications. CAMBRIDGE, Mass. & AUSTIN, Texas--(BUSINESS WIRE)-- Foundation Medicine, Inc. and Natera, Inc. (NASDAQ: NTRA), today announced the launch of the research use version of FoundationOne®Tracker, Foundation Medicine’s personalized circulating tumor DNA (ctDNA) monitoring assay. CPMO ROTATION PROGRAM 2021 The Opportunity. The selected candidate will join the CPMO on a six month rotation and will work closely with a CPMO mentor to manage projects, as assigned, to COVERAGE, BILLING, AND FINANCIAL ASSISTANCE FOR FOUNDATION Coverage, Billing, and Financial Assistance for Foundation Medicine Tests 86% of patients have a financial responsibility of $100 or less for Foundation Medicine testing.1, 3–7 Insurance CoverageFOUNDATION MEDICINE
Foundation Medicine is a world-leading molecular insights company, connecting physicians, patients and biopharma partners to the latest insights in cancer genomics. Learn more.FOUNDATIONONE CDX
FoundationOne®CDx is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors and is for prescription use only.The test analyzes 324 genes as well as genomic signatures including microsatellite instability (MSI) and tumor mutational burden (TMB) and is a companion diagnostic to identify patients who may benefit from treatment with FOUNDATIONONE LIQUID CDX FoundationOne®Liquid CDx is for prescription use only and is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors.The test analyzes 324 genes utilizing circulating cell-free DNA and is FDA-approved to report short variants in 311 genes and as a companion diagnostic to identify patients who may benefit from treatment with BILLING AND REIMBURSEMENT The Foundation Medicine FoundationAccess™ program supports providers and their patients through the coverage and billing process. For each comprehensive genomic profiling test ordered, we complete a benefits investigation and reach out to patients whom we expect may have out ofpocket costs.
PATIENT FAQS
The testing offered by Foundation Medicine is a type of genomic testing called comprehensive genomic profiling (CGP). It looks for mutations in over 300 genes in the tumor with one sample. The mutations in the tumor are what may impact tumor growth, spread, and response to therapy. Foundation Medicine testing may identify bothkinds of
ADMIN | IAM
Welcome Back. Sign into your account. Email Address. Password FOUNDATION MEDICINE LAUNCHES PRECISION MEDICINE EXCHANGE CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Foundation Medicine, Inc. (NASDAQ:FMI) today announced the launch of its precision medicine partner program, Precision Medicine Exchange Consortium™ (PMEC), to facilitate data exchange, advance research, and support education and applications of precision medicine in oncology and molecularpathology.
PRITI HEGDE, PHD
Priti Hegde, Ph.D., joined Foundation Medicine as the Chief Scientific Officer in August 2019. Dr. Hegde oversees clinical product development, cancer genomics, and early-stage research, as well as regulatory and quality assurance, to accelerate advancement of the Company’s leading comprehensive genomic profiling portfolio. FOUNDATION MEDICINE ANNOUNCES KEY DEVELOPMENTS IN Company Receives CE Mark Approval for FoundationOne ®, Adds New Distribution Partner and Appoints Experienced Executive to Lead International Expansion. CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Foundation Medicine, Inc. (NASDAQ: FMI) today announced that it has obtained a CE mark for FoundationOne ®.The CE mark allows FoundationOne to be placed on the market in the European COVERAGE, BILLING, AND FINANCIAL ASSISTANCE FOR FOUNDATION Coverage, Billing, and Financial Assistance for Foundation Medicine Tests 86% of patients have a financial responsibility of $100 or less for Foundation Medicine testing.1, 3–7 Insurance CoverageFOUNDATION MEDICINE
Foundation Medicine is a world-leading molecular insights company, connecting physicians, patients and biopharma partners to the latest insights in cancer genomics. Learn more.FOUNDATIONONE CDX
FoundationOne®CDx is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors and is for prescription use only.The test analyzes 324 genes as well as genomic signatures including microsatellite instability (MSI) and tumor mutational burden (TMB) and is a companion diagnostic to identify patients who may benefit from treatment with FOUNDATIONONE LIQUID CDX FoundationOne®Liquid CDx is for prescription use only and is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors.The test analyzes 324 genes utilizing circulating cell-free DNA and is FDA-approved to report short variants in 311 genes and as a companion diagnostic to identify patients who may benefit from treatment with BILLING AND REIMBURSEMENT The Foundation Medicine FoundationAccess™ program supports providers and their patients through the coverage and billing process. For each comprehensive genomic profiling test ordered, we complete a benefits investigation and reach out to patients whom we expect may have out ofpocket costs.
PATIENT FAQS
The testing offered by Foundation Medicine is a type of genomic testing called comprehensive genomic profiling (CGP). It looks for mutations in over 300 genes in the tumor with one sample. The mutations in the tumor are what may impact tumor growth, spread, and response to therapy. Foundation Medicine testing may identify bothkinds of
ADMIN | IAM
Welcome Back. Sign into your account. Email Address. Password FOUNDATION MEDICINE LAUNCHES PRECISION MEDICINE EXCHANGE CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Foundation Medicine, Inc. (NASDAQ:FMI) today announced the launch of its precision medicine partner program, Precision Medicine Exchange Consortium™ (PMEC), to facilitate data exchange, advance research, and support education and applications of precision medicine in oncology and molecularpathology.
PRITI HEGDE, PHD
Priti Hegde, Ph.D., joined Foundation Medicine as the Chief Scientific Officer in August 2019. Dr. Hegde oversees clinical product development, cancer genomics, and early-stage research, as well as regulatory and quality assurance, to accelerate advancement of the Company’s leading comprehensive genomic profiling portfolio. FOUNDATION MEDICINE ANNOUNCES KEY DEVELOPMENTS IN Company Receives CE Mark Approval for FoundationOne ®, Adds New Distribution Partner and Appoints Experienced Executive to Lead International Expansion. CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Foundation Medicine, Inc. (NASDAQ: FMI) today announced that it has obtained a CE mark for FoundationOne ®.The CE mark allows FoundationOne to be placed on the market in the European COVERAGE, BILLING, AND FINANCIAL ASSISTANCE FOR FOUNDATION Coverage, Billing, and Financial Assistance for Foundation Medicine Tests 86% of patients have a financial responsibility of $100 or less for Foundation Medicine testing.1, 3–7 Insurance Coverage FOUNDATIONONE LIQUID CDX FoundationOne®Liquid CDx is for prescription use only and is a qualitative next-generation sequencing based in vitro diagnostic test for advanced cancer patients with solid tumors.The test analyzes 324 genes utilizing circulating cell-free DNA and is FDA-approved to report short variants in 311 genes and as a companion diagnostic to identify patients who may benefit from treatment withFOUNDATIONONE HEME
FoundationOne Heme is a comprehensive genomic profiling (CGP) test combining DNA and RNA sequencing for patients with hematologic malignancies, sarcomas or solid tumors where RNA sequencing is desired. FoundationOne Heme detects known, novel, and complex fusion events as well as other common genomic alterations (substitutions,indels, and CNVs).
LICENSES | FOUNDATION MEDICINE All Foundation Medicine comprehensive genomic profiling tests are CAP Accredited, CLIA Certified, and New York State Department of Health (NYSDOH) Approved. View our permits and accreditations.ORDER A TEST
Foundation Medicine tests must be ordered by a licensed physician. For faster order processing, login or sign up for a Foundation Medicine online account. Orders can also be submitted via email or via fax to +1 (617) 418-2290 using our Test Requisition Form.. If sending a solid tumor specimen, Foundation Medicine can complete the specimen retrieval process if requested on the order formPATIENT FAQS
The testing offered by Foundation Medicine is a type of genomic testing called comprehensive genomic profiling (CGP). It looks for mutations in over 300 genes in the tumor with one sample. The mutations in the tumor are what may impact tumor growth, spread, and response to therapy. Foundation Medicine testing may identify bothkinds of
IHC TESTING
The results of PD-L1 IHC testing combined with CGP results, including data on tumor mutational burden (TMB) and microsatellite instability (MSI), may help you make more informed treatment decisions regarding use of immunotherapies and enrollment in appropriate clinical trialsfor your patients.
2021 ASCO ANNUAL MEETING FoundationOne Liquid CDx. FoundationOne®Liquid CDx, the broadest FDA-approved liquid biopsy, is now the only blood-based comprehensive genomic profiling companion diagnostic (CDx) for Lynparza® (olaparib)in prostate cancer.
IMMUNOTHERAPY BIOMARKERS Programmed Death Ligand-1 (PD-L1) PD-L1 is a protein biomarker that is strongly associated with immune system suppression. 18 Positive PD-L1 immunohistochemistry (IHC) can indicate that a patient will be more likely to respond to immunotherapy. However, the variability of this biomarker highlights the need for additional tools to predict which FOUNDATION MEDICINE LAUNCHES FOUNDATIONONE®TRACKER CTDNA Personalized technology aims to address both early- and advanced-stage cancer monitoring applications. CAMBRIDGE, Mass. & AUSTIN, Texas--(BUSINESS WIRE)-- Foundation Medicine, Inc. and Natera, Inc. (NASDAQ: NTRA), today announced the launch of the research use version of FoundationOne®Tracker, Foundation Medicine’s personalized circulating tumor DNA (ctDNA) monitoring assay. COVERAGE, BILLING, AND FINANCIAL ASSISTANCE FOR FOUNDATION Coverage, Billing, and Financial Assistance for Foundation Medicine Tests 86% of patients have a financial responsibility of $100 or less for Foundation Medicine testing.1, 3–7 Insurance Coverage + Skip to main content Log In Create an account * Products & ServicesBack
Explore
* About Our Products & Services* Our Tests
Back
Our Tests
* Compare Our Tests
* FoundationOne CDx
* FoundationOne Liquid CDx * FoundationOne Heme* IHC Testing
* Provider Services
Back
Provider Services
* Overview
* Online Ordering & Report Integrations * Clinical Research & Trial Matching* Decision Support
* Biopharma Partner ServicesBack
Biopharma Partner Services* Overview
* Genomic Data Solutions * Clinical Trial Solutions * Companion Diagnostic Development & Commercialization * Actionable Results Latest CDx Indication FoundationOne®Liquid CDx, the broadest FDA-approved liquid biopsy, is now the only blood-based comprehensive genomic profiling companion diagnostic (CDx) for Lynparza® (olaparib) in prostate cancer.* Resource Center
Back
Explore
* View All Resources* For Providers
Back
Provider FAQ
* What is Comprehensive Genomic Profiling? * Which genes are tested? * How do I order a Foundation Medicine test? * View all Provider FAQsProvider Resources
* Why Comprehensive Genomic Profiling? * Immunotherapy Biomarkers * Billing and Financial Assistance * View all Provider Resources * For Biopharma PartnersBack
Biopharma Partner FAQ * How can genomic data generated by Foundation Medicine tests be used for biomarker discovery? * How can working with Foundation Medicine help facilitate drugdevelopment?
* How do I contact someone in the biopharma group to discuss aproject?
* View all Biopharma Partner FAQsBiopharma Resources
* Biopharma Partner Services * Biopharma Brochure * View all Biopharma Resources* For Patients
Back
Patient FAQs
* Do all test results lead to actionable treatment options? * Does insurance cover my test? * Do the tests apply to all types of cancer? * View all Patient FAQsPatient Resources
* Patient Brochure
* Doctor Discussion Guide * Financial Assistance Application * View all Patient Resources Latest CDx Indication FoundationOne®Liquid CDx, the broadest FDA-approved liquid biopsy, is now the only blood-based comprehensive genomic profiling companion diagnostic (CDx) for Lynparza® (olaparib) in prostate cancer.* News & Events
Back
Explore
* News & Events
* Press Releases
* Foundation Forward Blog* Events
* Media Library
Useful Information
* About Us
* Leadership Team
* Browse Publications* About Us
Back
About Us
* Our Story
* Our History
* Leadership Team
* Contact Us
Useful Information
* Testing Portfolio
* Why Comprehensive Genomic Profiling? * Browse Publications* Careers
* For Patients
* Order a Test
NOW FDA-APPROVED: FOUNDATIONONE®LIQUID CDX With the broadest gene panel of any FDA-approved blood-based test, FoundationOne Liquid CDx analyzes over 300 genes and has Medicare coverage for qualifying patients across all solid tumors.*Learn More Dismiss
DIRECTLY IMPACTING PATIENT CARE TODAY _and Shaping the Future of Patient Care Tomorrow_ We are dedicated to a transformation in cancer care driven by the belief that the more we know about cancer’s molecular underpinnings, the better we can help physicians make the best treatment decisions for their patients. Through richer science, deeper insights, and stronger partnerships—we’re redefining the way cancer is treated. ADVANCING CANCER CARE TOGETHER Every advancement we make today has the potential to help the next patient diagnosed tomorrow. In just ten years, we’ve made incredible progress—breakthroughs that have helped shift the treatment paradigm to directly impact patient care. Foundation Medicine, a leading decision insights company, is helping physicians connect their patients to the latest cancer treatment options—making precision medicine a reality for thousands.FOR PROVIDERS
Foundation Medicine’s proven portfolio of tests and services offers the quality and commitment you need to help guide treatment strategies for all of your advanced cancer patients.PROVIDER RESOURCES
*
FoundationOne®CDx
*
Liquid Biopsy: From an Improbable Idea, New Potential Emerges*
Provider FAQs
FOR BIOPHARMA PARTNERS We provide actionable insights into the oncogenomics of both rare and common cancers, fueling discovery through commercialization by using a robust database and deep experience with FDA approvals. BIOPHARMA PARTNER RESOURCES*
An End-To-End Approach To PrecisionMedicine
*
Biopharma Brochure
*
Biopharma Partner FAQsFOR PATIENTS
Every cancer is unique. That’s why we are committed to providing specific genomic insights into your cancer to help your doctor guide a personalized care plan.PATIENT RESOURCES
*
Information For Patients*
Patient Brochure
*
Patient FAQs
MORE THAN NUMBERS
Our unique knowledge base, FoundationCore® , is one of the world's largest cancer genomic databases, with more than 400,000 genomic profiles. It is designed to evaluate the genomic landscape across cancer types to better understand tumor biology, molecular biomarkers, and which treatments might work for which patients. This all helps researchers and biopharma companies develop new therapies, design better clinical trials, and advance precision medicine.>400k
Patients Tested
>50
Biopharma Partners
>500
Peer-reviewed Publications VALUABLE INSIGHTS, ACTIONABLE OPTIONS Our tests help to identify the genomic alterations driving a patient's cancer and match them with relevant targeted therapies, immunotherapies, and clinical trial options. We also providedecision support
services, financial assistance , and technology solutionsto help support
all aspects of patient care. Learn more about our products and servicesPATIENT STORY
Treating cancer means more than just dealing with the disease; it also means treating the whole person. Christine Bray, ovarian cancer survivor, shares her journey to remission after obtaining a Foundation Medicine comprehensive genomic profiling test. We Continue to Drive for Future Breakthroughs in Patient CareLATEST CDX TEST
FoundationOne®Liquid CDx is the broadest FDA-approved blood-based comprehensive genomic profiling test, providing guideline-recommended genomic results from a simple blood draw. Learn more about FoundationOne®Liquid CDxADDITIONAL NOTES
*Medicare and Medicare Advantage members have coverage of FoundationOne Liquid CDx in accordance with the Centers for Medicare and Medicaid Services (CMS) national coverage determination(NCD) criteria.
IMPORTANT SAFETY INFORMATIONSELECT
FoundationOne CDx FoundationOne Liquid CDx FoundationOne CDx * FoundationOne CDx * FoundationOne Liquid CDx * FoundationOne CDx * FoundationOne Liquid CDxFOUNDATIONONE CDX
FoundationOne®CDx is a qualitative next-generation sequencing based _in vitro _diagnostic test for advanced cancer patients with solid tumors and is for prescription use only. The test analyzes 324 genes as well as genomic signatures including microsatellite instability (MSI) and tumor mutational burden (TMB) and is a companion diagnostic to identify patients who may benefit from treatment with specific therapies in accordance with the approved therapeutic product labeling. Additional genomic findings may be reported and are not prescriptive or conclusive for labeled use of any specific therapeutic product. Use of the test does not guarantee a patient will be matched to a treatment. A negative result does not rule out the presence of an alteration. Some patients may require a biopsy. For the complete label, including companion diagnostic indications and important risk information, please visit www.F1CDxLabel.com.
FOUNDATIONONE LIQUID CDX FoundationOne®Liquid CDx is for prescription use only and is a qualitative next-generation sequencing based _in vitro _diagnostic test for advanced cancer patients with solid tumors. The test analyzes 324 genes utilizing circulating cell-free DNA and is FDA-approved to report short variants in 311 genes and as a companion diagnostic to identify patients who may benefit from treatment with specific therapies (listed in Table 1 of the Intended Use) in accordance with the approved therapeutic product labeling. Additional genomic findings may be reported and are not prescriptive or conclusive for labeled use of any specific therapeutic product. Use of the test does not guarantee a patient will be matched to a treatment. A negative result does not rule out the presence of an alteration. Patients who are negative for companion diagnostic mutations should be reflexed to tumor tissue testing and mutation status confirmed using an FDA-approved tumor tissue test, if feasible. For the complete label, including companion diagnostic indications and complete risk information, please visit www.F1LCDxLabel.com.
* Products & Services* Our Tests
* Provider Services
* Biopharma Partner Services* News & Events
* Press Releases
* Blog
* Events
* About Us
* Our Story
* Leadership Team
* Careers
* Contact Us
* Login
* Order a Test
� 2020 FOUNDATION MEDICINE, INC.* Legal & Privacy
* California Compliance* Licenses
*
*
*
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0