Are you over 18 and want to see adult content?
More Annotations
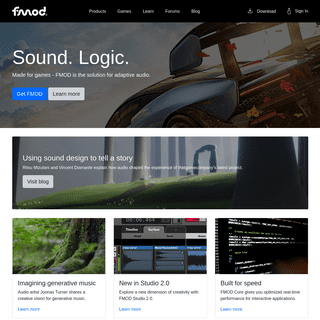
A complete backup of congratulationsto.com
Are you over 18 and want to see adult content?

A complete backup of mysocialsecurity.be
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of sekolahsaham.com
Are you over 18 and want to see adult content?

A complete backup of bazarsahambourse.com
Are you over 18 and want to see adult content?
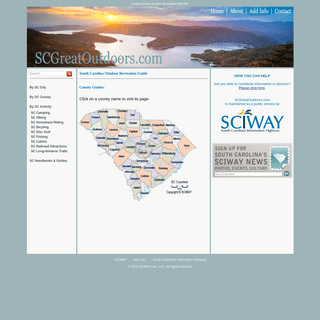
A complete backup of scgreatoutdoors.com
Are you over 18 and want to see adult content?

A complete backup of padremanyanet.edu.co
Are you over 18 and want to see adult content?
Text
ONCORE - LOGIN
Advarra Educational Resources. Q&A from Remote Monitoring: Study Compliance In A Changing World. Improving Study Activation Time for Gene Therapy Research. Pitfalls of Chemistry, Manufacturing, and Controls in Investigational New Drug Preparation. Regulatory Fine Points: What Research Sites Need to Do for Part 11 Compliance.ALLEGRO LOGIN
Allegro Login. Supported browsers: The latest stable versions of Chrome, Edge, Firefox & Safari. WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here. CRUSHFTP WEBINTERFACE Remember Me. Login. I forgot my password.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tips CRUSHFTP WEBINTERFACE :: RESET PASSWORD CrushFTP WebInterface :: Reset Password. English Bulgarian Chinese (中國) Czech (Čeština) Danish (Danske) Dutch (Nederlands) French (Français) German (Deutsch) Hungarian (Magyar) Italian (Italiano) Japanese (日本語) Korean Polish (Polskie) Romanian Russian Slovak (Slovenský) Spanish (Español) Username or Email : Password :Password
ONCORE - LOGIN
Advarra Educational Resources. Q&A from Remote Monitoring: Study Compliance In A Changing World. Improving Study Activation Time for Gene Therapy Research. Pitfalls of Chemistry, Manufacturing, and Controls in Investigational New Drug Preparation. Regulatory Fine Points: What Research Sites Need to Do for Part 11 Compliance.ALLEGRO LOGIN
Allegro Login. Supported browsers: The latest stable versions of Chrome, Edge, Firefox & Safari. WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here. CRUSHFTP WEBINTERFACE Remember Me. Login. I forgot my password.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tips CRUSHFTP WEBINTERFACE :: RESET PASSWORD CrushFTP WebInterface :: Reset Password. English Bulgarian Chinese (中國) Czech (Čeština) Danish (Danske) Dutch (Nederlands) French (Français) German (Deutsch) Hungarian (Magyar) Italian (Italiano) Japanese (日本語) Korean Polish (Polskie) Romanian Russian Slovak (Slovenský) Spanish (Español) Username or Email : Password :Password
WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tips CRUSHFTP WEBINTERFACE :: RESET PASSWORD CrushFTP WebInterface :: Reset Password. English Bulgarian Chinese (中國) Czech (Čeština) Danish (Danske) Dutch (Nederlands) French (Français) German (Deutsch) Hungarian (Magyar) Italian (Italiano) Japanese (日本語) Korean Polish (Polskie) Romanian Russian Slovak (Slovenský) Spanish (Español) Username or Email : Password :Password
ALLEGRO LOGIN
Allegro Login. Supported browsers: The latest stable versions of Chrome, Edge, Firefox & Safari.ONCORE - LOGIN
Advarra Educational Resources. Q&A from Remote Monitoring: Study Compliance In A Changing World. Improving Study Activation Time for Gene Therapy Research. Pitfalls of Chemistry, Manufacturing, and Controls in Investigational New Drug Preparation. Regulatory Fine Points: What Research Sites Need to Do for Part 11 Compliance. CRUSHFTP WEBINTERFACE Remember Me. Login. I forgot my password. WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tips CRUSHFTP WEBINTERFACE :: RESET PASSWORD CrushFTP WebInterface :: Reset Password. English Bulgarian Chinese (中國) Czech (Čeština) Danish (Danske) Dutch (Nederlands) French (Français) German (Deutsch) Hungarian (Magyar) Italian (Italiano) Japanese (日本語) Korean Polish (Polskie) Romanian Russian Slovak (Slovenský) Spanish (Español) Username or Email : Password :Password
ALLEGRO LOGIN
Allegro Login. Supported browsers: The latest stable versions of Chrome, Edge, Firefox & Safari.ONCORE - LOGIN
Advarra Educational Resources. Q&A from Remote Monitoring: Study Compliance In A Changing World. Improving Study Activation Time for Gene Therapy Research. Pitfalls of Chemistry, Manufacturing, and Controls in Investigational New Drug Preparation. Regulatory Fine Points: What Research Sites Need to Do for Part 11 Compliance. CRUSHFTP WEBINTERFACE Remember Me. Login. I forgot my password. WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tips CRUSHFTP WEBINTERFACE :: RESET PASSWORD CrushFTP WebInterface :: Reset Password. English Bulgarian Chinese (中國) Czech (Čeština) Danish (Danske) Dutch (Nederlands) French (Français) German (Deutsch) Hungarian (Magyar) Italian (Italiano) Japanese (日本語) Korean Polish (Polskie) Romanian Russian Slovak (Slovenský) Spanish (Español) Username or Email : Password :Password
WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tips CRUSHFTP WEBINTERFACE :: RESET PASSWORD CrushFTP WebInterface :: Reset Password. English Bulgarian Chinese (中國) Czech (Čeština) Danish (Danske) Dutch (Nederlands) French (Français) German (Deutsch) Hungarian (Magyar) Italian (Italiano) Japanese (日本語) Korean Polish (Polskie) Romanian Russian Slovak (Slovenský) Spanish (Español) Username or Email : Password :Password
ALLEGRO LOGIN
Allegro Login. Supported browsers: The latest stable versions of Chrome, Edge, Firefox & Safari.ONCORE - LOGIN
Advarra Educational Resources. Q&A from Remote Monitoring: Study Compliance In A Changing World. Improving Study Activation Time for Gene Therapy Research. Pitfalls of Chemistry, Manufacturing, and Controls in Investigational New Drug Preparation. Regulatory Fine Points: What Research Sites Need to Do for Part 11 Compliance. CRUSHFTP WEBINTERFACE Remember Me. Login. I forgot my password. WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tipsALLEGRO LOGIN
Allegro Login. Supported browsers: The latest stable versions of Chrome, Edge, Firefox & Safari.ONCORE - LOGIN
Advarra Educational Resources. Q&A from Remote Monitoring: Study Compliance In A Changing World. Improving Study Activation Time for Gene Therapy Research. Pitfalls of Chemistry, Manufacturing, and Controls in Investigational New Drug Preparation. Regulatory Fine Points: What Research Sites Need to Do for Part 11 Compliance. CRUSHFTP WEBINTERFACE Remember Me. Login. I forgot my password. WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tips WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here.GETTING STARTED
Welcome to Allegro Community, a secure and confidential online environment to network with other research sites and professionals, share common best practices and procedures, and collaborate with other Allegro users for tips CRUSHFTP WEBINTERFACE :: RESET PASSWORD CrushFTP WebInterface :: Reset Password. English Bulgarian Chinese (中國) Czech (Čeština) Danish (Danske) Dutch (Nederlands) French (Français) German (Deutsch) Hungarian (Magyar) Italian (Italiano) Japanese (日本語) Korean Polish (Polskie) Romanian Russian Slovak (Slovenský) Spanish (Español) Username or Email : Password :Password
ALLEGRO LOGINALLEGRO BANKCADENCE ALLEGRO Allegro Login. Supported browsers: The latest stable versions of Chrome, Edge, Firefox & Safari.ONCORE - LOGIN
Advarra Educational Resources. Q&A from Remote Monitoring: Study Compliance In A Changing World. Improving Study Activation Time for Gene Therapy Research. Pitfalls of Chemistry, Manufacturing, and Controls in Investigational New Drug Preparation. Regulatory Fine Points: What Research Sites Need to Do for Part 11 Compliance. CRUSHFTP WEBINTERFACEFORTE RESEARCH APPSALLEGRO FORTE RESEARCHFORTE RESEARCH SYSTEMS INCFORTE CLINICAL RESEARCH WEBINARSFORTE RESEARCH MADISON WIFORTE MADISON WI Remember Me. Login. I forgot my password. WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here. ALLEGRO LOGINALLEGRO BANKCADENCE ALLEGRO Allegro Login. Supported browsers: The latest stable versions of Chrome, Edge, Firefox & Safari.ONCORE - LOGIN
Advarra Educational Resources. Q&A from Remote Monitoring: Study Compliance In A Changing World. Improving Study Activation Time for Gene Therapy Research. Pitfalls of Chemistry, Manufacturing, and Controls in Investigational New Drug Preparation. Regulatory Fine Points: What Research Sites Need to Do for Part 11 Compliance. CRUSHFTP WEBINTERFACEFORTE RESEARCH APPSALLEGRO FORTE RESEARCHFORTE RESEARCH SYSTEMS INCFORTE CLINICAL RESEARCH WEBINARSFORTE RESEARCH MADISON WIFORTE MADISON WI Remember Me. Login. I forgot my password. WWW.FORTERESEARCH.COM Moved Permanently. The document has moved here. CRUSHFTP WEBINTERFACE :: RESET PASSWORD CrushFTP WebInterface :: Reset Password. English Bulgarian Chinese (中國) Czech (Čeština) Danish (Danske) Dutch (Nederlands) French (Français) German (Deutsch) Hungarian (Magyar) Italian (Italiano) Japanese (日本語) Korean Polish (Polskie) Romanian Russian Slovak (Slovenský) Spanish (Español) Username or Email : Password :Password
Skip to content
* Search
* Contact
Advarra | Forte Advarra | ForteMenu
* Solutions
* Customers
* Resources
* About Us
* Search
* Contact Us
TRANSFORM CLINICAL RESEARCH WITH A SITE-CENTRIC APPROACH Experience breakthrough efficiencies in the key areas of Clinical Trial Management, Clinical Data Management, and ResearchAdministration
Solutions
x
SOLUTIONS
Our solutions work together to streamline your clinical researchoperations
View all Solutions
SERVICES
Increase your productivity with our shared services including protocol calendars, budget setup and moreView all Services
CLINICAL TRIAL MANAGEMENT Enterprise Research System Business Intelligence eRegulatory Management Automated Participant Payments CLINICAL DATA MANAGEMENT Electronic Data Capture RESEARCH ADMINISTRATION Research Evaluation and Reporting Business Intelligencex
ABOUT
Our mission is to advance human health by empowering compliant, efficient, and collaborative research * Our StoryFounded with the desire to redefine how clinical trialsare conducted
* Our CultureCore values are more than just words on the wall * Our TeamTogether we find innovative ways to help our customers * Our AdvisorsMeet our Customer Advisory Board * CareersExplore open positions * MediaRead recent Forte newsx
RESOURCES
article Articles
Stay informed with quick reads about the clinical research industry.ebook eBooks
Read helpful deep dives into industry trends and best practices. Case Study Case Studies Learn how customers are transforming clinical research with ourtechnologies.
infographic Infographics Visualize key metrics and stories in the clinical research industry. worksheet Worksheets Download interactive sheets to help streamline your researchoperations.
webinar Webinars
Hear from industry leaders about the latest topics in clinical research — view live or on-demand.View all Resources
Read Featured Series webinar Upcoming Webinar HOW SITES AND SPONSORS ARE COLLABORATING TO MOVE ESOURCE FORWARD Tuesday, April 21, 202012:00 pm CST
Register Today
TOP RESEARCH CENTERS USE FORTE SOLUTIONS TO UNLEASH THEIR RESEARCHPOTENTIAL
Our customers include NCI-designated cancer centers, Clinical and Translational Science Award (CTSA) recipients and leading healthsystems.
Learn what it’s like to work with usASK ADVARRA LIVE
Join us April 16, 2020 at 12 p.m. CST to discuss the real-world impact of COVID-19 on researchRegister now
RECOMMENDED READING
Learn more about industry best practices, keep up with recent product updates and catch the latest news from Forte.INDUSTRY PICKS
* WORKSHEET: Project Management in Clinical Trials: Weekly Work Plan * EBOOK: ACRP Technology Competency and Adoption Survey ResultsReport
* INFOGRAPHIC: Patient Recruitment and Enrollment in Clinical Trials * WEBINAR: The Importance of Data Cleaning and Quality in ClinicalResearch
POPULAR ARTICLES
* Overcome These 5 Common Clinical Trial Challenges Using a CTMS * Breaking Down the CCSG: An Overview of the Cancer Center SupportGrant
* Continuing Education Unit (CEU) Basics for Clinical Research * 3 Essential Elements of Change Management Within Clinical ResearchOperations
IN THE NEWS
* Advarra Offers Free Research Compliance Resources During COVID-19Crisis
* Advarra Announces Intent to Acquire Forte, Market-Leading Provider of Clinical Technology Solutions * Forte Continues Growth and Support of the Nation’s Leading Cancer Research Institutions * Duke’s Big-Bang CTMS Deployment For Clinical Research – OnCoreCTMS
OUR COMPANY CULTURE
We are a group of purpose-driven individuals united by our core values – and everything we do is for our customers. Learn more about our culture TECHNOLOGY DEVELOPED THROUGH COLLABORATION We develop clinical research software to help cancer centers, academic medical centers and health systems streamline their operations.Sign up for a demo
COLLABORATION IS AT THE HEART OF EVERYTHING WE DO Our customers join a community of leading research institutions to collaborate on industry-wide efficiencies and shape the solutions thatsupport them.
Learn what it’s like to work with usSOLUTIONS
* OnCore
* EDC
* eReg
* EVAL
* Insights
* Payments
* Allegro
SERVICES
* Budget Setup Service* Calendars
* Certification
* EDC Study Design
* Optimization
* Staff AugmentationRESOURCES
* Articles
* eBooks
* Webinars
* Worksheets
* Infographics
* Case Studies
INFORMATION
* Customers
* Privacy
* Legal
* Security
* ISO Certification
COMPANY
* About Forte
* Our Culture
* Our Team
* CareersHiring
* Media
* Contact Us
Twitter Facebook
Advarra | Forte
triangle-up
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0