Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://usamilitarymedals.com
Are you over 18 and want to see adult content?
A complete backup of https://trumbullandporterhotel.com
Are you over 18 and want to see adult content?

A complete backup of https://journeyofhearts.org
Are you over 18 and want to see adult content?

A complete backup of https://tyshenka-voiskovoy-specrezerv.ru
Are you over 18 and want to see adult content?
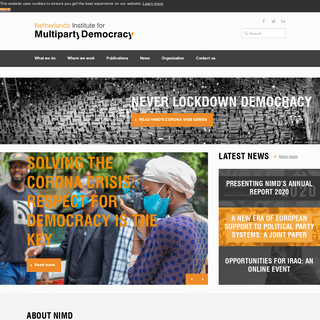
A complete backup of https://nimd.org
Are you over 18 and want to see adult content?

A complete backup of https://bandarqidn.com
Are you over 18 and want to see adult content?

A complete backup of https://developerminnat.xyz
Are you over 18 and want to see adult content?
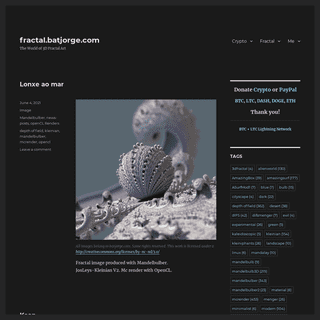
A complete backup of https://batjorge.com
Are you over 18 and want to see adult content?

A complete backup of https://athletesinaction.org
Are you over 18 and want to see adult content?
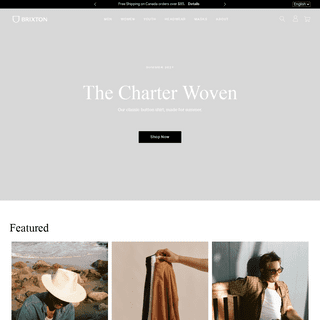
A complete backup of https://brixton.com
Are you over 18 and want to see adult content?

A complete backup of https://bowlofdelicious.com
Are you over 18 and want to see adult content?
A complete backup of https://google.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of www.www.zenra.net
Are you over 18 and want to see adult content?

A complete backup of www.www.nanabook.com
Are you over 18 and want to see adult content?
A complete backup of www.dvdgayonline.com
Are you over 18 and want to see adult content?
A complete backup of www.stickamgirls.net
Are you over 18 and want to see adult content?
A complete backup of www.aristoshemales.com
Are you over 18 and want to see adult content?

A complete backup of www.planetsuzy.org
Are you over 18 and want to see adult content?
A complete backup of noodlemagazine.com
Are you over 18 and want to see adult content?
Text
UPDATE MAGAZINE
Update EXTRA. Update EXTRA is an occasional electronic-only feature of Update magazine designed to provide interviews, reporting, and analysis on emerging issues of broad interest to our community. WEBINARS - FOOD AND DRUG LAW INSTITUTE (FDLI)IL LAW INSTITUTEINTERNATIONAL LAW INSTITUTE FDLI invites you to submit a proposal recommending a topic for an upcoming webinar. Ideas related to all FDA-regulated industries are welcome! Webinar Topics Can Address: Ideas, Trends, and Current Hot Topics in Food and Drug Law. Ideas for Professional Development. Topics for Beginner/Intermediate/Advanced Level Regulatory or LegalPractitioners.
READY OR NOT, HERE IT COMES! THE DRUG SUPPLY CHAIN Part II: DSCSA Enforcement On February 7, 2019, FDA issued a Warning Letter to McKesson Corporation, a wholesale drug distributor, following a Form FDA 483 issued in connection with a 2018 inspection of the firm’s corporate headquarters and a distribution facility.This marks the first Warning Letter issued by the Agency citing violations of DSCSA provisions currently in effect. THE LEGALIZATION OF HEMP SCIENTIFIC EXCHANGE: GREY AREAS AND BEST PRACTICESSCIENTIFIC EXCHANGE DEFINITIONSCIENTIFIC EXCHANGE VERSUS PROMOTIONSCIENCE EXCHANGE COMPANYSCIENCE EXCHANGE INCSCIENTIFIC MINDS Scientific Exchange: Grey Areas and Best Practices Susan Cantrell, Chief Executive Officer, Academy of Managed Care Pharmacy Kellie Combs, Partner, Ropes & Gray LLP Mark Gaydos, VP, NA General Medicines & Established Products/US Advertising & Promotion, Global Regulatory Affairs, Sanofi Moderated by Ellen Schumacher, Executive Director, Commercial Regulatory Affairs, Bristol- INTRODUCTION TO FDA REVIEW AND APPROVAL OF BIOLOGICAL PRODUCTS 3 Background on Biologics What is a biologic? –Simple definition: A medicine derived from living organisms –The FDA definition: A virus, therapeutic serum, toxin, antitoxin, vaccine, TOWARD A GLOBAL SOLUTION ON VACCINE LIABILITY AND 1 Toward a Global Solution on Vaccine Liability and Compensation JOHN D. WINTER, CASSYE COLE, AND JONAH WACHOLDER* ABSTRACT In the past fiveyears,
HOME - FOOD AND DRUG LAW INSTITUTE (FDLI)VIRTUAL LEARNINGPROGRAMSRESOURCESGET INVOLVEDCAREERABOUT The Food and Drug Law Institute (FDLI), founded in 1949, is a nonprofit membership organization that offers education, training, publications, and professional engagement opportunities in the 2021 FDLI ANNUAL CONFERENCE AGENDA Always informative and entertaining, this perennially popular session promises insight into the most significant current litigation and a look at cases to keep an eye on in the coming year. Annual Conference attendees receive the companion e-publication, Top Food and Drug Law Cases 2020, and Cases to Watch, 2021. CAREER - FOOD AND DRUG LAW INSTITUTE (FDLI)IL LAW INSTITUTEINTERNATIONAL LAW INSTITUTENATIONAL DRUG COURT INSTITUTETN LAW INSTITUTELAW ENFORCEMENT INSTITUTE For young professionals, the New to Food and Drug Law Group provides an additional avenue for learning and connecting. Our Introductory Courses offer a foundation for success as well as a valuable review for those with more experience. Students will benefit from these programs, too. The Career Center offers a chance to explore new opportunities at all levels.UPDATE MAGAZINE
Update EXTRA. Update EXTRA is an occasional electronic-only feature of Update magazine designed to provide interviews, reporting, and analysis on emerging issues of broad interest to our community. WEBINARS - FOOD AND DRUG LAW INSTITUTE (FDLI)IL LAW INSTITUTEINTERNATIONAL LAW INSTITUTE FDLI invites you to submit a proposal recommending a topic for an upcoming webinar. Ideas related to all FDA-regulated industries are welcome! Webinar Topics Can Address: Ideas, Trends, and Current Hot Topics in Food and Drug Law. Ideas for Professional Development. Topics for Beginner/Intermediate/Advanced Level Regulatory or LegalPractitioners.
READY OR NOT, HERE IT COMES! THE DRUG SUPPLY CHAIN Part II: DSCSA Enforcement On February 7, 2019, FDA issued a Warning Letter to McKesson Corporation, a wholesale drug distributor, following a Form FDA 483 issued in connection with a 2018 inspection of the firm’s corporate headquarters and a distribution facility.This marks the first Warning Letter issued by the Agency citing violations of DSCSA provisions currently in effect. THE LEGALIZATION OF HEMP SCIENTIFIC EXCHANGE: GREY AREAS AND BEST PRACTICESSCIENTIFIC EXCHANGE DEFINITIONSCIENTIFIC EXCHANGE VERSUS PROMOTIONSCIENCE EXCHANGE COMPANYSCIENCE EXCHANGE INCSCIENTIFIC MINDS Scientific Exchange: Grey Areas and Best Practices Susan Cantrell, Chief Executive Officer, Academy of Managed Care Pharmacy Kellie Combs, Partner, Ropes & Gray LLP Mark Gaydos, VP, NA General Medicines & Established Products/US Advertising & Promotion, Global Regulatory Affairs, Sanofi Moderated by Ellen Schumacher, Executive Director, Commercial Regulatory Affairs, Bristol- INTRODUCTION TO FDA REVIEW AND APPROVAL OF BIOLOGICAL PRODUCTS 3 Background on Biologics What is a biologic? –Simple definition: A medicine derived from living organisms –The FDA definition: A virus, therapeutic serum, toxin, antitoxin, vaccine, TOWARD A GLOBAL SOLUTION ON VACCINE LIABILITY AND 1 Toward a Global Solution on Vaccine Liability and Compensation JOHN D. WINTER, CASSYE COLE, AND JONAH WACHOLDER* ABSTRACT In the past fiveyears,
CAREER - FOOD AND DRUG LAW INSTITUTE (FDLI) For young professionals, the New to Food and Drug Law Group provides an additional avenue for learning and connecting. Our Introductory Courses offer a foundation for success as well as a valuable review for those with more experience. Students will benefit from these programs, too. The Career Center offers a chance to explore new opportunities at all levels. WEBINARS - FOOD AND DRUG LAW INSTITUTE (FDLI) FDLI invites you to submit a proposal recommending a topic for an upcoming webinar. Ideas related to all FDA-regulated industries are welcome! Webinar Topics Can Address: Ideas, Trends, and Current Hot Topics in Food and Drug Law. Ideas for Professional Development. Topics for Beginner/Intermediate/Advanced Level Regulatory or LegalPractitioners.
TOBACCO AND NICOTINE PRODUCTS REGULATION AND POLICY Join a diverse group of stakeholders – public health advocates, researchers, manufacturers, lawyers, consumer interest groups, entrepreneurs, governmental agencies, and others – for this three-day conference on effectively regulating the broad spectrum of tobacco and nicotine products in the US. AUTHOR HAPPY HOUR: EVOLVING FOOD AND DIETARY SUPPLEMENT Join FDLI at the first “Author Happy Hour” for the Summer 2021 issue of Update magazine to discuss the article, “Food and Supplement Class Action Suits That Rely on Alleged Regulatory Violations.”Theodora McCormick of Epstein Becker Green will lead a discussion on the evolution of food and dietary supplement class action suits from those historically based on allegations of deceptive FOOD AND DRUG LAW JOURNAL The Food and Drug Law Journal is a peer-reviewed quarterly devoted to the analysis of legislation, regulations, court decisions, and public policies affecting industries regulated by the U.S. Food and Drug Administration (FDA) and related agencies and authorities, including the development, manufacture, marketing, and use of drugs, medical devices, biologics, food, dietary supplements FDLI ANNUAL CONFERENCE: EXPLORING ADVANCED TOPICS IN FOOD ERIKA F. LIETZAN is the William H. Pittman Professor of Law & Timothy J. Heinsz Professor of Law at the University of Missouri School of Law, where she focuses her teaching and scholarship on food and drug law, intellectual property, and administrative law.She’s currently working on an article relating to pharmaceutical trademarks and an article on medical device patents. ADVERTISING AND PROMOTION FOR MEDICAL PRODUCTS CONFERENCE With this in mind, we are excited to announce that the 2021 Advertising and Promotion for Medical Products Conference will be a virtual event. Please know we are actively exploring options to ensure an engaging and enjoyable experience with ample opportunities to connect with your peers. Be sure to check the course website regularlyfor
FOOD ADVERTISING, LABELING, AND LITIGATION CONFERENCE Food Advertising, Labeling, and Litigation Conference - Food and Drug Law Institute (FDLI) Email Updates. Become a Member. 800-956-6293. in fo@fdli.org. Log In. HIGHLIGHTS AND INSIGHTS FROM THE GENERAL SNUS MODIFIED Highlights and Insights from the General Snus Modified Risk Tobacco Product Decision Documents . By James M. Solyst. INTRODUCTION. The October 22, 2019 Food and Drug Administration (FDA) decision to authorize the marketing of the Swedish Match USA line of General snus products as Modified Risk Tobacco Products (MRTPs) was a much-needed statement of support for tobacco harm reduction COMPLIANCE CENTRAL WITH FDA CDER COMPLIANCE DIRECTOR Compliance Central with FDA Center Compliance Directors: Part I Donald Ashley, Director, Office of Compliance, Center for Drug Evaluation and Research, FDA Erin Keith, Associate Director for Compliance and Quality, Center for Devices and Radiological Health, FDA Melissa Mendoza, Deputy Director, Office of Compliance and Biologics Quality,Center for
Skip to content
* __Email Updates
* __Become a Member
* __800-956-6293
* __info@fdli.org
* __Log In
* __Email Updates
* __Become a Member
* __800-956-6293
* __info@fdli.org
* __Log In
* Programs __
* All Programs
* Introductory Courses* Webinars
* Past Conferences
* CLE and Policies
* Resources __
* Books
* Update Magazine
* Top Food and Drug Cases * Food and Drug Law Journal * Food and Drug Policy Forum * Sign Up for FDLI SmartBrief* Get Involved __
* Member Benefits
* Membership Categories* Join
* Committees
* Submit Your Ideas
* Speak
* Author
* Sponsor
* New to Food and Drug Law* Career __
* Career Center
* Students
* New to Food and Drug Law * Austern Writing Competition* About __
* Who We Are
* Board of Directors* Committees
* Staff
* Leadership Awards
* Service to FDLI Award*
Search for:
* Programs
* All Programs
* Introductory Courses* Webinars
* Past Conferences
* CLE and Policies
* Resources
* Books
* Update Magazine
* Top Food and Drug Cases * Food and Drug Law Journal * Food and Drug Policy Forum * Sign Up for FDLI SmartBrief* Get Involved
* Member Benefits
* Membership Categories* Join
* Committees
* Submit Your Ideas
* Speak
* Author
* Sponsor
* New to Food and Drug Law* Career
* Career Center
* Students
* New to Food and Drug Law * Austern Writing Competition* About
* Who We Are
* Board of Directors* Committees
* Staff
* Leadership Awards
* Service to FDLI Award*
CLASS ACTION OF FDA-REGULATED PRODUCTS: RECENT UPDATES AND FUTURE FORECASTS AUGUST 21, 2019 | 2:00-3:30 PM EDT | LIVE WEBINARRegister Now
*
ENFORCEMENT, LITIGATION, AND COMPLIANCE CONFERENCE DECEMBER 11-12, 2019 | WASHINGTON, DC CALL FOR PROPOSALS CLOSES AUGUST 8Register Now
*
GOING VIRAL: SAFEGUARDING PUBLIC HEALTH IN THE MODERN ERA _FOOD AND DRUG LAW JOURNAL_ 2019 SYMPOSIUM NOVEMBER 2019 | WASHINGTON, DC CALL FOR ABSTRACTS ENDS AUGUST 9Learn More
*
* �
Homeadmin
2019-08-05T08:51:30-04:00 _A MEMBERSHIP ORGANIZATION ENGAGING THE FOOD AND DRUG LAW COMMUNITY_FDLI NEWS
* Register for the FOOD ADVERTISING, LABELING, AND LITIGATIONCONFERENCE
by August 29 and SAVE! * View the newly-released agenda for ADVERTISING AND PROMOTION FOR MEDICAL PRODUCTS CONFERENCEMEMBERS
* Member Insight Valued: CALL FOR PROPOSALS OPEN FOR 2019 ENFORCEMENT, LITIGATION, AND COMPLIANCE CONFERENCE * Complimentary-for-Members: FINDING REGULATORY ZEN FOR CANNABIS AND HEMP PRODUCTS AFTER THE 2018 FARM BILLGET INVOLVED
* AUGUST 8: Call for proposals for the ENFORCEMENT, LITIGATION, AND COMPLIANCE CONFERENCEcloses
* AUGUST 9: Call for abstracts for the _FOOD AND DRUG LAW JOURNAL_2019 SYMPOSIUM
closes
* Stay in the Know: SUBSCRIBE TO FDLI’S SMARTBRIEF NEWSLETTER UPCOMING CONFERENCES*
*
*
admin 2019-08-09T13:30:55-04:00 FOOD ADVERTISING, LABELING, AND LITIGATION CONFERENCE*
*
*
admin 2019-08-08T12:14:12-04:00 ADVERTISING AND PROMOTION FOR MEDICAL PRODUCTS CONFERENCE*
*
*
Kiana Walker
2019-08-09T11:51:44-04:00 TOBACCO AND NICOTINE PRODUCTS REGULATION AND POLICY CONFERENCEMore Events
INTRODUCTORY COURSES FDLI offers intensive training courses covering the basics of FDA lawand regulation.
More Courses
*
*
*
admin 2019-08-09T12:33:34-04:00 INTRODUCTION TO FOOD LAW AND REGULATION*
*
*
admin 2019-07-31T09:51:09-04:00 INTRODUCTION TO BIOLOGICS AND BIOSIMILARS LAW AND REGULATION*
*
*
admin 2019-08-07T14:39:08-04:00 INTRODUCTION TO ADVERTISING AND PROMOTION FOR MEDICAL PRODUCTS_UPDATE_ MAGAZINE
This publication provides concise articles on cutting-edge food anddrug law issues.
Full Issues
*
*
*
hayley.carpenter@fdli.org 2019-07-18T12:51:23-04:00 NEW DIETARY INGREDIENTS (NDI) AND INNOVATION IN DIETARY SUPPLEMENTS: A CALL FOR NEW COMPLIANCE AND ENFORCEMENT STRATEGIESWEBINARS
FDLI webinars provide timely education in a convenient format and offer interactive features.More Webinars
*
*
*
LauraHaak
2019-08-01T16:23:19-04:00 INTRODUCTION TO THE REGULATION OF CANNABIS-DERIVED PRODUCTS LATEST TWEETS FROM @FOODANDDRUGLAW (SEE ALL)
LATEST TWEETS FROM @FDACOMMISSIONER (SEE ALL)
FOOD AND DRUG LAW INSTITUTE 1155 15th Street NW, Suite 910 Washington, DC 20005CONTACT US
Terms of Use | Privacy PolicyTwitter Facebook
Your Email
Please leave this field empty.×
CLOSE
Go to Top
×
Close
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0