Are you over 18 and want to see adult content?
More Annotations
A complete backup of https://lol-world.ru
Are you over 18 and want to see adult content?
A complete backup of https://myschools.nyc
Are you over 18 and want to see adult content?
A complete backup of https://mulhouse.fr
Are you over 18 and want to see adult content?
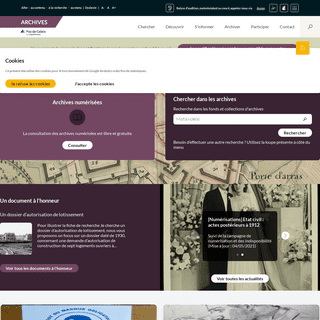
A complete backup of https://archivespasdecalais.fr
Are you over 18 and want to see adult content?
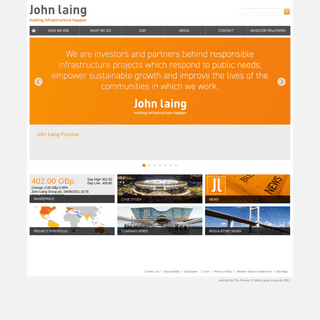
A complete backup of https://laing.com
Are you over 18 and want to see adult content?
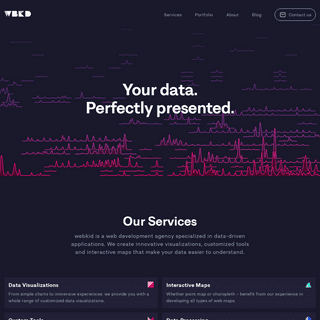
A complete backup of https://webkid.io
Are you over 18 and want to see adult content?
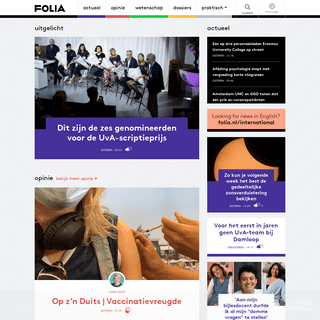
A complete backup of https://folia.nl
Are you over 18 and want to see adult content?
A complete backup of https://showmeyourmumu.com
Are you over 18 and want to see adult content?
A complete backup of https://lararnastidning.se
Are you over 18 and want to see adult content?
A complete backup of https://yottagames.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of www.www.millionairematch.com
Are you over 18 and want to see adult content?

A complete backup of fluffychicks.net
Are you over 18 and want to see adult content?

A complete backup of www.tastyblacks.com
Are you over 18 and want to see adult content?

A complete backup of www.www.enature.tv
Are you over 18 and want to see adult content?

A complete backup of www.www.abcoeur.com
Are you over 18 and want to see adult content?

A complete backup of beautifulagony.com
Are you over 18 and want to see adult content?

A complete backup of www.www.imagefap.com
Are you over 18 and want to see adult content?

A complete backup of www.www.yournnpic.com
Are you over 18 and want to see adult content?
Text
stewardship.
RESPIRATORY PANEL WITH SARS-COV-2 DETECTION The BioFire RP2.1 Panel is a multiplex PCR test that targets 22 viruses and bacteria—including SARS-CoV-2, influenza, RSV, and other common causes of respiratory infections—in about 45 minutes. While SARS-CoV-2 remains top of mind for healthcare providers and RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. FILMARRAY® PANELS—INFECTIOUS DISEASE DIAGNOSTICS The BioFire Panels test for viruses, bacteria, parasites, yeast, and antimicrobial resistance genes. Whether you’re trying to select appropriate therapy for a septic patient or determine exactly which respiratory pathogen is making a young child sick, the BioFire ® FilmArray ® Systems can return answers fast. Click on the icons belowto learn more.
GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by THE BIOFIRE® FILMARRAY® PNEUMONIA PANEL The BioFire PN Panel identifies 33 clinically relevant targets from sputum (including endotracheal aspirate) and bronchoalveolar lavage (including mini-BAL) samples. For 15 of the bacteria, the BioFire PN Panel provides semi-quantitative results, which may help determine whether an organism is a BIOFIRE GASTROINTESTINAL PANEL TESTING Company Name Company Address BioFire® Gastrointestinal (GI) Panel Testing Page 2 of 17 FLM1-MKT-0071-03. Principle of the Procedure The BioFire GI Panel pouch is a closed system disposable that houses all the chemistry required PROTOCOL FOR LABORATORY VERIFICATION OF PERFORMANCE OF THE 1 | P a g e Technical Note BioFire Diagnostics, LLC www.biofiredx.com BFR0000-6592-01 T QS-339B-02 TECHNICAL ::: NOTE Protocol forLaboratory
FILMARRAY® RESPIRATORY PANEL TESTING The FilmArray RP is a multiplexed nucleic acid test intended for use with the FilmArray Instrument for the simultaneous qualitative detection and identification of multiple respiratory viral and bacterial nucleic acids in nasopharyngeal swabs (NPS) SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire System is the new standard for syndromic infectious disease diagnostics. Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. The right test, the first time can impact all areas of patient care. Healthcare providers can see faster diagnoses and improved antibioticstewardship.
RESPIRATORY PANEL WITH SARS-COV-2 DETECTION The BioFire RP2.1 Panel is a multiplex PCR test that targets 22 viruses and bacteria—including SARS-CoV-2, influenza, RSV, and other common causes of respiratory infections—in about 45 minutes. While SARS-CoV-2 remains top of mind for healthcare providers and RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. FILMARRAY® PANELS—INFECTIOUS DISEASE DIAGNOSTICS The BioFire Panels test for viruses, bacteria, parasites, yeast, and antimicrobial resistance genes. Whether you’re trying to select appropriate therapy for a septic patient or determine exactly which respiratory pathogen is making a young child sick, the BioFire ® FilmArray ® Systems can return answers fast. Click on the icons belowto learn more.
GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by THE BIOFIRE® FILMARRAY® PNEUMONIA PANEL The BioFire PN Panel identifies 33 clinically relevant targets from sputum (including endotracheal aspirate) and bronchoalveolar lavage (including mini-BAL) samples. For 15 of the bacteria, the BioFire PN Panel provides semi-quantitative results, which may help determine whether an organism is a BIOFIRE GASTROINTESTINAL PANEL TESTING Company Name Company Address BioFire® Gastrointestinal (GI) Panel Testing Page 2 of 17 FLM1-MKT-0071-03. Principle of the Procedure The BioFire GI Panel pouch is a closed system disposable that houses all the chemistry required PROTOCOL FOR LABORATORY VERIFICATION OF PERFORMANCE OF THE 1 | P a g e Technical Note BioFire Diagnostics, LLC www.biofiredx.com BFR0000-6592-01 T QS-339B-02 TECHNICAL ::: NOTE Protocol forLaboratory
FILMARRAY® RESPIRATORY PANEL TESTING The FilmArray RP is a multiplexed nucleic acid test intended for use with the FilmArray Instrument for the simultaneous qualitative detection and identification of multiple respiratory viral and bacterial nucleic acids in nasopharyngeal swabs (NPS) BIOFIRE COVID-19 TESTING SOLUTIONS BioFire® Respiratory 2.1 (RP2.1) Panel. Now the first FDA De Novo authorized test for COVID-19, the BioFire RP2.1 Panel detects 22 respiratory pathogens, including SARS-CoV-2, to help clinicians quickly rule in and rule out common causes of respiratory illness in about 45 minutes. The BioFire RP2.1 Panel runs on the BioFire ®FilmArray
RESPIRATORY PANEL WITH SARS-COV-2 DETECTION The BioFire RP2.1 Panel is a multiplex PCR test that targets 22 viruses and bacteria—including SARS-CoV-2, influenza, RSV, and other common causes of respiratory infections—in about 45 minutes. While SARS-CoV-2 remains top of mind for healthcare providers and BIOFIRE® FILMARRAY® PRODUCT SUPPORT DOCUMENTS BioFire ® FilmArray ® Instrument – EC Declaration of Conformity; BioFire ® FilmArray ® 2.0 Instrument – EC Declaration of Conformity; BioFire ® FilmArray ® Torch – EC Declaration of Conformity; BioFire ® Respiratory 2.1 plus (RP2.1plus) Panel – EC Declaration of Conformity (OUS)*; BioFire ® FilmArray ® Respiratory 2 (RP2) Panel – EC Declaration of Conformity BIOFIRE DIAGNOSTICS CAREER OPPORTUNITIES BioFire Diagnostics has combined its career opportunities with those of its parent company, bioMérieux. This provides you with even more job opportunities in the fields of manufacturing, engineering, software development, sales, marketing, IT/IS, accounting, and more—all in one place. Click the “Search Careers” button belowto visit the
VOTE BIOFIRE RESPIRATORY 2.1 PANEL FOR BEST CLINICAL Laboratory scientists across the world are being invited to have their say in the 2021 Scientists’ Choice Awards ®.. Now in their 14th year, the annual Scientists’ Choice Awards celebrate the most innovative and effective technologies of the past year and provide scientists worldwide with the opportunity to acknowledge the instruments, assays, tools, or consumables that have made the most THE BIOFIRE® FILMARRAY® PNEUMONIA PANEL The BioFire PN Panel identifies 33 clinically relevant targets from sputum (including endotracheal aspirate) and bronchoalveolar lavage (including mini-BAL) samples. For 15 of the bacteria, the BioFire PN Panel provides semi-quantitative results, which may help determine whether an organism is a BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for VIDEO LIBRARY: WORKSHOPS & WEBINARS From Timely Diagnosis to Improved Patient Outcomes: Respiratory Tract Infection and Meningitis. This webinar is intended for pediatricians, emergency medicine physicians, and infectious disease specialists to increase their knowledge regarding available rapid molecular panel tests for patients with suspected acute respiratory tract infectionsor meningitis.
LEGAL NOTICES
If you don’t see a particular BioFire product listed below, please feel free to contact Jill Powlick at (801) 262-3592 ext.1423 or USPatents@biomerieux.com or Legal.Diagnostics@biofiredx.com for further information on its intellectual property status. Product Names(Part Numbers)
INTENDED USE
423738 Page 3 of 55 BioFire ® Respiratory Panel 2.1 (RP2.1) BFR0000-8303-01 May 2020 PRINCIPLE OF THE PROCEDURE The BioFire ® RP2.1 pouch is a closed system disposable that stores all the necessary reagents for sample SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire System is the new standard for syndromic infectious disease diagnostics. Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. The right test, the first time can impact all areas of patient care. Healthcare providers can see faster diagnoses and improved antibioticstewardship.
RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by THE BIOFIRE® FILMARRAY® PNEUMONIA PANEL The BioFire PN Panel identifies 33 clinically relevant targets from sputum (including endotracheal aspirate) and bronchoalveolar lavage (including mini-BAL) samples. For 15 of the bacteria, the BioFire PN Panel provides semi-quantitative results, which may help determine whether an organism is a BIOFIRE FILMARRAY MENINGITIS/ENCEPHALITIS PANEL (ME Add the BioFire ME Panel as a first line test and get answers in a clinically-actionable time frame. Simple: 2 minutes of hands-on time. Easy: No precise measuring or pipetting required. Fast: Turnaround time of about one hour. Comprehensive: 14 bacterial, viral, and fungaltargets.
SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire System is the new standard for syndromic infectious disease diagnostics. Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. The right test, the first time can impact all areas of patient care. Healthcare providers can see faster diagnoses and improved antibioticstewardship.
RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by THE BIOFIRE® FILMARRAY® PNEUMONIA PANEL The BioFire PN Panel identifies 33 clinically relevant targets from sputum (including endotracheal aspirate) and bronchoalveolar lavage (including mini-BAL) samples. For 15 of the bacteria, the BioFire PN Panel provides semi-quantitative results, which may help determine whether an organism is a BIOFIRE FILMARRAY MENINGITIS/ENCEPHALITIS PANEL (ME Add the BioFire ME Panel as a first line test and get answers in a clinically-actionable time frame. Simple: 2 minutes of hands-on time. Easy: No precise measuring or pipetting required. Fast: Turnaround time of about one hour. Comprehensive: 14 bacterial, viral, and fungaltargets.
BIOFIRE® SYNDROMIC TRENDS: EPIDEMIOLOGY TOOL BioFire Syndromic Trends (Trend) is a software feature from BioFire that provides local and regional pathogen circulation trends on demand. Users can view pathogen trends for their lab as well as regional and national pathogen trends, all of which are created from de-identified data that has been aggregated with other participatinginstitutions
COST SAVINGS BENEFITS: BIOFIRE FILMARRAY SYSTEM In fact, septicemia is the most expensive condition treated in US hospitals, accounting for over $23 million in healthcare costs every year.12 Depending on severity, a single case of sepsis costs an average of $7,970 but can cost as much as $44,027.13. The BioFire ® Blood Culture Identification 2 Panel identifies 43 organisms inpositive blood
PROTOCOL FOR LABORATORY VERIFICATION OF PERFORMANCE OF THE 1 | P a g e Technical Note BioFire Diagnostics, LLC www.biofiredx.com BFR0000-6592-01 T QS-339B-02 TECHNICAL ::: NOTE Protocol forLaboratory
RESPIRATORY PANEL WITH SARS-COV-2 DETECTION The BioFire RP2.1 Panel is a multiplex PCR test that targets 22 viruses and bacteria—including SARS-CoV-2, influenza, RSV, and other common causes of respiratory infections—in about 45 minutes. While SARS-CoV-2 remains top of mind for healthcare providers and BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
VOTE BIOFIRE RESPIRATORY 2.1 PANEL FOR BEST CLINICAL Laboratory scientists across the world are being invited to have their say in the 2021 Scientists’ Choice Awards ®.. Now in their 14th year, the annual Scientists’ Choice Awards celebrate the most innovative and effective technologies of the past year and provide scientists worldwide with the opportunity to acknowledge the instruments, assays, tools, or consumables that have made the most BIOFIRE DIAGNOSTICS CAREER OPPORTUNITIES BioFire Diagnostics has combined its career opportunities with those of its parent company, bioMérieux. This provides you with even more job opportunities in the fields of manufacturing, engineering, software development, sales, marketing, IT/IS, accounting, and more—all in one place. Click the “Search Careers” button belowto visit the
THE BIOFIRE® FILMARRAY® PNEUMONIA PANEL The BioFire PN Panel identifies 33 clinically relevant targets from sputum (including endotracheal aspirate) and bronchoalveolar lavage (including mini-BAL) samples. For 15 of the bacteria, the BioFire PN Panel provides semi-quantitative results, which may help determine whether an organism is a FILMARRAY & PANEL FREQUENTLY ASKED QUESTIONS Insert a USB flash drive into the BioFire 2.0 System computer USB port. In the Browse Runs tab, search for the desired run file (s). Right click on the run (s) and select Export Anonymous Runs. Save the file to the desktop and then move it to a flash drive (it should be an.adb file).
BIOFIRE® SYNDROMIC TRENDS: EPIDEMIOLOGY TOOL BioFire Syndromic Trends (Trend) is a software feature from BioFire that provides local and regional pathogen circulation trends on demand. Users can view pathogen trends for their lab as well as regional and national pathogen trends, all of which are created from de-identified data that has been aggregated with other participatinginstitutions
BIOFIRE GASTROINTESTINAL PANEL TESTING Company Name Company Address BioFire® Gastrointestinal (GI) Panel Testing Page 2 of 17 FLM1-MKT-0071-03. Principle of the Procedure The BioFire GI Panel pouch is a closed system disposable that houses all the chemistry required FILMARRAY® RESPIRATORY PANEL TESTING The FilmArray RP is a multiplexed nucleic acid test intended for use with the FilmArray Instrument for the simultaneous qualitative detection and identification of multiple respiratory viral and bacterial nucleic acids in nasopharyngeal swabs (NPS) SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire ® FilmArray ® System. The Fastest Way to Better Results. The BioFire System is the new standard for syndromic infectious disease diagnostics.Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by BIOFIRE FILMARRAY MENINGITIS/ENCEPHALITIS PANEL (ME Add the BioFire ME Panel as a first line test and get answers in a clinically-actionable time frame. Simple: 2 minutes of hands-on time. Easy: No precise measuring or pipetting required. Fast: Turnaround time of about one hour. Comprehensive: 14 bacterial, viral, and fungaltargets.
FILMARRAY BLOOD CULTURE IDENTIFICATION (BCID/BCID2) PANEL Early identification and treatment of sepsis are essential to combat one of the leading causes of hospital patient deaths. 1 With 43 targets, the newly expanded and FDA-cleared BioFire ® Blood Culture Identification 2 (BCID2) Panel detects pathogens and antimicrobial resistance genes directly from positive blood cultures. BIOFIRE® SYNDROMIC TRENDS: EPIDEMIOLOGY TOOL BioFire Syndromic Trends (Trend) is a software feature from BioFire that provides local and regional pathogen circulation trends on demand. Users can view pathogen trends for their lab as well as regional and national pathogen trends, all of which are created from de-identified data that has been aggregated with other participatinginstitutions
COST SAVINGS BENEFITS: BIOFIRE FILMARRAY SYSTEM In fact, septicemia is the most expensive condition treated in US hospitals, accounting for over $23 million in healthcare costs every year.12 Depending on severity, a single case of sepsis costs an average of $7,970 but can cost as much as $44,027.13. The BioFire ® Blood Culture Identification 2 Panel identifies 43 organisms inpositive blood
PROTOCOLS FOR LABORATORY VERIFICATION OF PERFORMANCE OF 3 | P a g e FLM1-PRT-0263-01 Technical Note BioFire Diagnostics, LLC www.BioFireDX.com T QS-339B-01 TECHNICAL ::: NOTE A BioFire® �FilmArray System is defined as all BioFire® FilmArray® Instruments or modules that are connected to and controlled by a single computersystem.
SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire ® FilmArray ® System. The Fastest Way to Better Results. The BioFire System is the new standard for syndromic infectious disease diagnostics.Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by BIOFIRE FILMARRAY MENINGITIS/ENCEPHALITIS PANEL (ME Add the BioFire ME Panel as a first line test and get answers in a clinically-actionable time frame. Simple: 2 minutes of hands-on time. Easy: No precise measuring or pipetting required. Fast: Turnaround time of about one hour. Comprehensive: 14 bacterial, viral, and fungaltargets.
FILMARRAY BLOOD CULTURE IDENTIFICATION (BCID/BCID2) PANEL Early identification and treatment of sepsis are essential to combat one of the leading causes of hospital patient deaths. 1 With 43 targets, the newly expanded and FDA-cleared BioFire ® Blood Culture Identification 2 (BCID2) Panel detects pathogens and antimicrobial resistance genes directly from positive blood cultures. BIOFIRE® SYNDROMIC TRENDS: EPIDEMIOLOGY TOOL BioFire Syndromic Trends (Trend) is a software feature from BioFire that provides local and regional pathogen circulation trends on demand. Users can view pathogen trends for their lab as well as regional and national pathogen trends, all of which are created from de-identified data that has been aggregated with other participatinginstitutions
COST SAVINGS BENEFITS: BIOFIRE FILMARRAY SYSTEM In fact, septicemia is the most expensive condition treated in US hospitals, accounting for over $23 million in healthcare costs every year.12 Depending on severity, a single case of sepsis costs an average of $7,970 but can cost as much as $44,027.13. The BioFire ® Blood Culture Identification 2 Panel identifies 43 organisms inpositive blood
PROTOCOLS FOR LABORATORY VERIFICATION OF PERFORMANCE OF 3 | P a g e FLM1-PRT-0263-01 Technical Note BioFire Diagnostics, LLC www.BioFireDX.com T QS-339B-01 TECHNICAL ::: NOTE A BioFire® �FilmArray System is defined as all BioFire® FilmArray® Instruments or modules that are connected to and controlled by a single computersystem.
RESPIRATORY PANEL WITH SARS-COV-2 DETECTION The BioFire RP2.1 Panel is a multiplex PCR test that targets 22 viruses and bacteria—including SARS-CoV-2, influenza, RSV, and other common causes of respiratory infections—in about 45 minutes. While SARS-CoV-2 remains top of mind for healthcare providers and BIOFIRE DIAGNOSTICS CAREER OPPORTUNITIES BioFire Diagnostics has combined its career opportunities with those of its parent company, bioMérieux. This provides you with even more job opportunities in the fields of manufacturing, engineering, software development, sales, marketing, IT/IS, accounting, and more—all in one place. Click the “Search Careers” button belowto visit the
BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for THE BIOFIRE® FILMARRAY® PNEUMONIA PANEL The BioFire PN Panel identifies 33 clinically relevant targets from sputum (including endotracheal aspirate) and bronchoalveolar lavage (including mini-BAL) samples. For 15 of the bacteria, the BioFire PN Panel provides semi-quantitative results, which may help determine whether an organism is a FILMARRAY & PANEL FREQUENTLY ASKED QUESTIONS Insert a USB flash drive into the BioFire 2.0 System computer USB port. In the Browse Runs tab, search for the desired run file (s). Right click on the run (s) and select Export Anonymous Runs. Save the file to the desktop and then move it to a flash drive (it should be an.adb file).
WHAT IS PCR?
What is PCR? PCR stands for polymerase chain reaction. It is a revolutionary chemical process that makes it possible to rapidly and exponentially amplify targeted nucleic acid. According to its inventor, American biochemist Kary Mullis, PCR “lets you pick the piece of DNA you’re interested in and have as much of it as youwant.” 1.
BIOFIRE® SYNDROMIC TRENDS: EPIDEMIOLOGY TOOL BioFire Syndromic Trends (Trend) is a software feature from BioFire that provides local and regional pathogen circulation trends on demand. Users can view pathogen trends for their lab as well as regional and national pathogen trends, all of which are created from de-identified data that has been aggregated with other participatinginstitutions
INTENDED USE
423738 Page 3 of 55 BioFire ® Respiratory Panel 2.1 (RP2.1) BFR0000-8303-01 May 2020 PRINCIPLE OF THE PROCEDURE The BioFire ® RP2.1 pouch is a closed system disposable that stores all the necessary reagents for sample FILMARRAY® RESPIRATORY PANEL TESTING The FilmArray RP is a multiplexed nucleic acid test intended for use with the FilmArray Instrument for the simultaneous qualitative detection and identification of multiple respiratory viral and bacterial nucleic acids in nasopharyngeal swabs (NPS) TORCH MELTING CURVE ANALYSIS WITH FILMARRAY 2.0 SOFTWARE 5 | P a g e Technical Note BioFire Diagnostics, LLC www.BioFireDX.com QS-339B-01 01 TECHNICAL ::: NOTE A-RT-9-NOTE: The operator can cancel the import process before it completes by selecting the Cancel button in the bottom-right corner of the dialog. Any runs that were imported before selecting Cancel will remain in the SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire ® FilmArray ® System. The Fastest Way to Better Results. The BioFire System is the new standard for syndromic infectious disease diagnostics.Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. BIOFIRE COVID-19 TESTING SOLUTIONS BioFire® Respiratory 2.1 (RP2.1) Panel. Now the first FDA De Novo authorized test for COVID-19, the BioFire RP2.1 Panel detects 22 respiratory pathogens, including SARS-CoV-2, to help clinicians quickly rule in and rule out common causes of respiratory illness in about 45 minutes. The BioFire RP2.1 Panel runs on the BioFire ®FilmArray
BIOFIRE DIAGNOSTICS CAREER OPPORTUNITIES BioFire Diagnostics has combined its career opportunities with those of its parent company, bioMérieux. This provides you with even more job opportunities in the fields of manufacturing, engineering, software development, sales, marketing, IT/IS, accounting, and more—all in one place. Click the “Search Careers” button belowto visit the
RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. BIOFIRE FILMARRAY MENINGITIS/ENCEPHALITIS PANEL (ME Add the BioFire ME Panel as a first line test and get answers in a clinically-actionable time frame. Simple: 2 minutes of hands-on time. Easy: No precise measuring or pipetting required. Fast: Turnaround time of about one hour. Comprehensive: 14 bacterial, viral, and fungaltargets.
LEGAL NOTICES
If you don’t see a particular BioFire product listed below, please feel free to contact Jill Powlick at (801) 262-3592 ext.1423 or USPatents@biomerieux.com or Legal.Diagnostics@biofiredx.com for further information on its intellectual property status. Product Names(Part Numbers)
MEET INSPIRING WOMEN LEADERS OF BIOFIRE Meet Some of the Inspiring Women Leaders of BioFire. At BioFire, we’re proud of a culture that supports and grows women in leadership. This isn’t just a platitude for us. Our CFO and interim CEO is a woman, along with our chief commercial officer. This representation flows throughout leadership—BioFire also has manyfemale senior vice
SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire ® FilmArray ® System. The Fastest Way to Better Results. The BioFire System is the new standard for syndromic infectious disease diagnostics.Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. BIOFIRE COVID-19 TESTING SOLUTIONS BioFire® Respiratory 2.1 (RP2.1) Panel. Now the first FDA De Novo authorized test for COVID-19, the BioFire RP2.1 Panel detects 22 respiratory pathogens, including SARS-CoV-2, to help clinicians quickly rule in and rule out common causes of respiratory illness in about 45 minutes. The BioFire RP2.1 Panel runs on the BioFire ®FilmArray
BIOFIRE DIAGNOSTICS CAREER OPPORTUNITIES BioFire Diagnostics has combined its career opportunities with those of its parent company, bioMérieux. This provides you with even more job opportunities in the fields of manufacturing, engineering, software development, sales, marketing, IT/IS, accounting, and more—all in one place. Click the “Search Careers” button belowto visit the
RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. BIOFIRE FILMARRAY MENINGITIS/ENCEPHALITIS PANEL (ME Add the BioFire ME Panel as a first line test and get answers in a clinically-actionable time frame. Simple: 2 minutes of hands-on time. Easy: No precise measuring or pipetting required. Fast: Turnaround time of about one hour. Comprehensive: 14 bacterial, viral, and fungaltargets.
LEGAL NOTICES
If you don’t see a particular BioFire product listed below, please feel free to contact Jill Powlick at (801) 262-3592 ext.1423 or USPatents@biomerieux.com or Legal.Diagnostics@biofiredx.com for further information on its intellectual property status. Product Names(Part Numbers)
MEET INSPIRING WOMEN LEADERS OF BIOFIRE Meet Some of the Inspiring Women Leaders of BioFire. At BioFire, we’re proud of a culture that supports and grows women in leadership. This isn’t just a platitude for us. Our CFO and interim CEO is a woman, along with our chief commercial officer. This representation flows throughout leadership—BioFire also has manyfemale senior vice
BIOFIRE COVID-19 TESTING SOLUTIONS BioFire® Respiratory 2.1 (RP2.1) Panel. Now the first FDA De Novo authorized test for COVID-19, the BioFire RP2.1 Panel detects 22 respiratory pathogens, including SARS-CoV-2, to help clinicians quickly rule in and rule out common causes of respiratory illness in about 45 minutes. The BioFire RP2.1 Panel runs on the BioFire ®FilmArray
SETTING NEW STANDARDS IN MOLECULAR DIAGNOSTICS Setting New Standards in Molecular Diagnostics. BioFire’s molecular solutions feature unmatched usability and speed, setting a new standard in molecular diagnostic platforms. The BioFire ® FilmArray � System and comprehensive panels empower all members of the care team to make better diagnostic decisions using the syndromic approach. SYNDROMICTRENDS—NOW WITH SARS-COV-2 DATA New Features, New Interface. The public-facing SyndromicTrends.com website now includes detection-rate information for the BioFire ® Respiratory 2.1 (RP2.1) Panel, including data for SARS-CoV-2 (the pathogen that causes COVID-19). Now visitors can view a stacked area chart of detection-rate data for each of the 22 pathogens detected by the BioFire RP2.1 Panel. BIOFIRE® FILMARRAY® PRODUCT SUPPORT DOCUMENTS BioFire ® FilmArray ® Instrument – EC Declaration of Conformity; BioFire ® FilmArray ® 2.0 Instrument – EC Declaration of Conformity; BioFire ® FilmArray ® Torch – EC Declaration of Conformity; BioFire ® Respiratory 2.1 plus (RP2.1plus) Panel – EC Declaration of Conformity (OUS)*; BioFire ® FilmArray ® Respiratory 2 (RP2) Panel – EC Declaration of Conformity ID MOLECULAR DIAGNOSTIC TESTING FOR LABS The BioFire FilmArray System: Streamlining Lab Processes with Syndromic Testing. By using a molecular syndromic approach and a broad test menu, the FilmArray can better detect and identify the infectious agent with just one easy test. Because of its ease-of-use, the same standard of care can be provided 24/7, removing the need to delay or send out certain tests. TECHNICAL SUPPORT & RESOURCES BioFire Support Assisting You with World Class Support. Excellence in customer support is a value deeply ingrained in the culture of BioFire. We want to provide our customers with the information needed to use our products with confidence and get the most rapid andaccurate results.
CUSTOMER SUPPORT FILMARRAY® PRODUCTS BioFire is dedicated to providing world class customer support 24 hours a day, 7 days a week, 365 days a year. For assistance please contact our customer technical support team at: support@biofiredx.com. 801-736-6354 option 5. 1-800-735-6544.LEGAL NOTICES
If you don’t see a particular BioFire product listed below, please feel free to contact Jill Powlick at (801) 262-3592 ext.1423 or USPatents@biomerieux.com or Legal.Diagnostics@biofiredx.com for further information on its intellectual property status. Product Names(Part Numbers)
BIOFIRE GASTROINTESTINAL PANEL TESTING Company Name Company Address BioFire® Gastrointestinal (GI) Panel Testing Page 2 of 17 FLM1-MKT-0071-03. Principle of the Procedure The BioFire GI Panel pouch is a closed system disposable that houses all the chemistry required MEET INSPIRING WOMEN LEADERS OF BIOFIRE Meet Some of the Inspiring Women Leaders of BioFire. At BioFire, we’re proud of a culture that supports and grows women in leadership. This isn’t just a platitude for us. Our CFO and interim CEO is a woman, along with our chief commercial officer. This representation flows throughout leadership—BioFire also has manyfemale senior vice
SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire ® FilmArray ® System. The Fastest Way to Better Results. The BioFire System is the new standard for syndromic infectious disease diagnostics.Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. BIOFIRE COVID-19 TESTING SOLUTIONS BioFire® Respiratory 2.1 (RP2.1) Panel. Now the first FDA De Novo authorized test for COVID-19, the BioFire RP2.1 Panel detects 22 respiratory pathogens, including SARS-CoV-2, to help clinicians quickly rule in and rule out common causes of respiratory illness in about 45 minutes. The BioFire RP2.1 Panel runs on the BioFire ®FilmArray
BIOFIRE DIAGNOSTICS CAREER OPPORTUNITIES BioFire Diagnostics has combined its career opportunities with those of its parent company, bioMérieux. This provides you with even more job opportunities in the fields of manufacturing, engineering, software development, sales, marketing, IT/IS, accounting, and more—all in one place. Click the “Search Careers” button belowto visit the
RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. BIOFIRE FILMARRAY MENINGITIS/ENCEPHALITIS PANEL (ME Add the BioFire ME Panel as a first line test and get answers in a clinically-actionable time frame. Simple: 2 minutes of hands-on time. Easy: No precise measuring or pipetting required. Fast: Turnaround time of about one hour. Comprehensive: 14 bacterial, viral, and fungaltargets.
LEGAL NOTICES
If you don’t see a particular BioFire product listed below, please feel free to contact Jill Powlick at (801) 262-3592 ext.1423 or USPatents@biomerieux.com or Legal.Diagnostics@biofiredx.com for further information on its intellectual property status. Product Names(Part Numbers)
MEET INSPIRING WOMEN LEADERS OF BIOFIRE Meet Some of the Inspiring Women Leaders of BioFire. At BioFire, we’re proud of a culture that supports and grows women in leadership. This isn’t just a platitude for us. Our CFO and interim CEO is a woman, along with our chief commercial officer. This representation flows throughout leadership—BioFire also has manyfemale senior vice
SYNDROMIC INFECTIOUS DISEASE DIAGNOSTICS The BioFire ® FilmArray ® System. The Fastest Way to Better Results. The BioFire System is the new standard for syndromic infectious disease diagnostics.Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. BIOFIRE COVID-19 TESTING SOLUTIONS BioFire® Respiratory 2.1 (RP2.1) Panel. Now the first FDA De Novo authorized test for COVID-19, the BioFire RP2.1 Panel detects 22 respiratory pathogens, including SARS-CoV-2, to help clinicians quickly rule in and rule out common causes of respiratory illness in about 45 minutes. The BioFire RP2.1 Panel runs on the BioFire ®FilmArray
BIOFIRE DIAGNOSTICS CAREER OPPORTUNITIES BioFire Diagnostics has combined its career opportunities with those of its parent company, bioMérieux. This provides you with even more job opportunities in the fields of manufacturing, engineering, software development, sales, marketing, IT/IS, accounting, and more—all in one place. Click the “Search Careers” button belowto visit the
RESPIRATORY PATHOGEN PANEL TEST Respiratory 2.1. (RP2.1) Panel. SARS-CoV-2 is a top concern for patients and clinicians, but several respiratory pathogens can cause nearly indistinguishable symptoms. The FDA De Novo authorized BioFire RP2.1 Panel is a frontline test to help clinicians quickly diagnose respiratory infections, including COVID-19, influenza, RSV, and manyothers.
BIOFIRE® FILMARRAY® TORCH & 2.0 The BioFire ® FilmArray ® System Syndromic infectious disease testing is the fastest way to better results. With integrated sample preparation, amplification, detection, and analysis, the BioFire System uses multiplex PCR technology to simultaneously test for GASTROINTESTINAL (GI) PATHOGEN PANEL With overlapping symptoms, distinguishing possible causes of gastroenteritis can be challenging for any clinician. To further complicate diagnosis, traditional testing methods are slow, labor intensive, and fail to reveal the etiology of a patient’s gastrointestinal symptoms. 1 Fortunately, syndromic testing from the BioFire GI Panel eliminates limitations from conventional methods by BIOFIRE® BONE & JOINT INFECTION (BJI) PANEL The BioFire BJI Panel tests for a comprehensive grouping of gram-positive and gram-negative bacteria, yeast, and antimicrobial resistance genes most often associated with bone and joint infections. It takes just one syndromic test, one small sample of synovial fluid, and about an hour to get results on 39 clinically relevant targets. BIOFIRE FILMARRAY MENINGITIS/ENCEPHALITIS PANEL (ME Add the BioFire ME Panel as a first line test and get answers in a clinically-actionable time frame. Simple: 2 minutes of hands-on time. Easy: No precise measuring or pipetting required. Fast: Turnaround time of about one hour. Comprehensive: 14 bacterial, viral, and fungaltargets.
LEGAL NOTICES
If you don’t see a particular BioFire product listed below, please feel free to contact Jill Powlick at (801) 262-3592 ext.1423 or USPatents@biomerieux.com or Legal.Diagnostics@biofiredx.com for further information on its intellectual property status. Product Names(Part Numbers)
MEET INSPIRING WOMEN LEADERS OF BIOFIRE Meet Some of the Inspiring Women Leaders of BioFire. At BioFire, we’re proud of a culture that supports and grows women in leadership. This isn’t just a platitude for us. Our CFO and interim CEO is a woman, along with our chief commercial officer. This representation flows throughout leadership—BioFire also has manyfemale senior vice
BIOFIRE COVID-19 TESTING SOLUTIONS BioFire® Respiratory 2.1 (RP2.1) Panel. Now the first FDA De Novo authorized test for COVID-19, the BioFire RP2.1 Panel detects 22 respiratory pathogens, including SARS-CoV-2, to help clinicians quickly rule in and rule out common causes of respiratory illness in about 45 minutes. The BioFire RP2.1 Panel runs on the BioFire ®FilmArray
SETTING NEW STANDARDS IN MOLECULAR DIAGNOSTICS Setting New Standards in Molecular Diagnostics. BioFire’s molecular solutions feature unmatched usability and speed, setting a new standard in molecular diagnostic platforms. The BioFire ® FilmArray � System and comprehensive panels empower all members of the care team to make better diagnostic decisions using the syndromic approach. SYNDROMICTRENDS—NOW WITH SARS-COV-2 DATA New Features, New Interface. The public-facing SyndromicTrends.com website now includes detection-rate information for the BioFire ® Respiratory 2.1 (RP2.1) Panel, including data for SARS-CoV-2 (the pathogen that causes COVID-19). Now visitors can view a stacked area chart of detection-rate data for each of the 22 pathogens detected by the BioFire RP2.1 Panel. BIOFIRE® FILMARRAY® PRODUCT SUPPORT DOCUMENTS BioFire ® FilmArray ® Instrument – EC Declaration of Conformity; BioFire ® FilmArray ® 2.0 Instrument – EC Declaration of Conformity; BioFire ® FilmArray ® Torch – EC Declaration of Conformity; BioFire ® Respiratory 2.1 plus (RP2.1plus) Panel – EC Declaration of Conformity (OUS)*; BioFire ® FilmArray ® Respiratory 2 (RP2) Panel – EC Declaration of Conformity ID MOLECULAR DIAGNOSTIC TESTING FOR LABS The BioFire FilmArray System: Streamlining Lab Processes with Syndromic Testing. By using a molecular syndromic approach and a broad test menu, the FilmArray can better detect and identify the infectious agent with just one easy test. Because of its ease-of-use, the same standard of care can be provided 24/7, removing the need to delay or send out certain tests. TECHNICAL SUPPORT & RESOURCES BioFire Support Assisting You with World Class Support. Excellence in customer support is a value deeply ingrained in the culture of BioFire. We want to provide our customers with the information needed to use our products with confidence and get the most rapid andaccurate results.
CUSTOMER SUPPORT FILMARRAY® PRODUCTS BioFire is dedicated to providing world class customer support 24 hours a day, 7 days a week, 365 days a year. For assistance please contact our customer technical support team at: support@biofiredx.com. 801-736-6354 option 5. 1-800-735-6544.LEGAL NOTICES
If you don’t see a particular BioFire product listed below, please feel free to contact Jill Powlick at (801) 262-3592 ext.1423 or USPatents@biomerieux.com or Legal.Diagnostics@biofiredx.com for further information on its intellectual property status. Product Names(Part Numbers)
BIOFIRE GASTROINTESTINAL PANEL TESTING Company Name Company Address BioFire® Gastrointestinal (GI) Panel Testing Page 2 of 17 FLM1-MKT-0071-03. Principle of the Procedure The BioFire GI Panel pouch is a closed system disposable that houses all the chemistry required MEET INSPIRING WOMEN LEADERS OF BIOFIRE Meet Some of the Inspiring Women Leaders of BioFire. At BioFire, we’re proud of a culture that supports and grows women in leadership. This isn’t just a platitude for us. Our CFO and interim CEO is a woman, along with our chief commercial officer. This representation flows throughout leadership—BioFire also has manyfemale senior vice
* MENU Menu
* HOME
* PRODUCTS
* FilmArray
* LIS Connectivity
* BioFire® Syndromic Trends* Panels
* Respiratory
* Respiratory EZ
* Blood Culture ID
* Gastrointestinal
* Meningitis/Encephalitis* Pneumonia
* Bone and Joint Infection* Research
* Investigator-Initiated Study Program* Testimonials
* Get A Quote
* SOLUTIONS
* BioFire COVID-19 Solutions * The Syndromic Approach* Point of Care
* For Healthcare Providers* For Labs
* For Customers
* COMPANY
* Mission & Values
* Careers
* Contact
* Blog
* Privacy Policy
* MEDIA
* Conferences & Events* Press Releases
* Press Kit
* SUPPORT
* Video Library
* FAQ
* Continuing Education* Return Forms
* Documents
* MENU Menu
covid-19
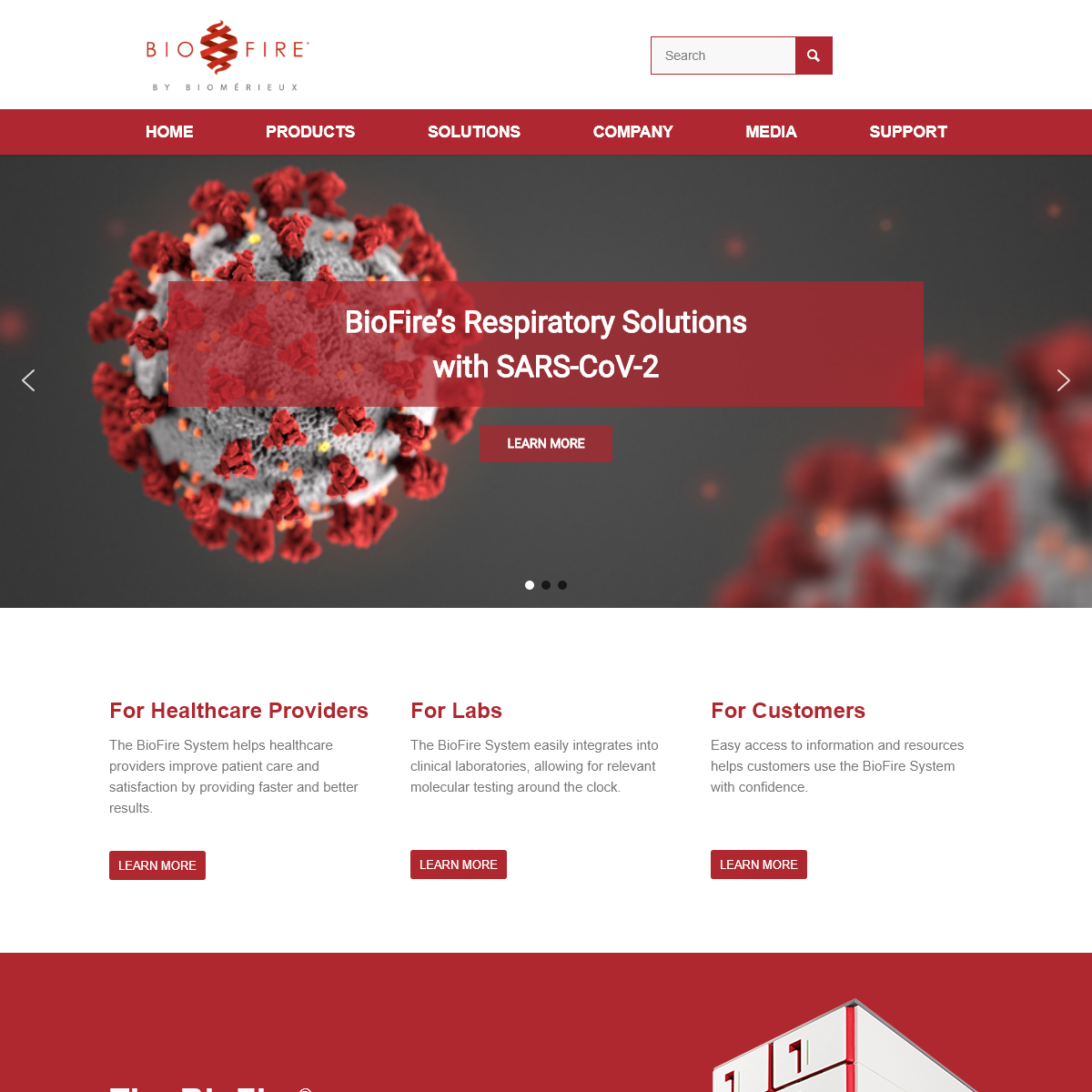
BioFire’s Respiratory Solutionswith SARS-CoV-2
LEARN MORE
Customer Login
CUSTOMER LOGIN
Click here to login and review your order status.LOGIN HERE
SyndromicTesting
Syndromic Testing:
Syndromic Testing:
The Right Test, The First Time. The Right Test, The First Time.LEARN MORE
LEARN MORE
FOR HEALTHCARE PROVIDERS The BioFire System helps healthcare providers improve patient care and satisfaction by providing faster and better results.LEARN MORE
FOR LABS
The BioFire System easily integrates into clinical laboratories, allowing for relevant molecular testing around the clock.LEARN MORE
FOR CUSTOMERS
Easy access to information and resources helps customers use the BioFire System with confidence.LEARN MORE
THE BIOFIRE®
FILMARRAY® SYSTEM
THE FASTEST WAY TO BETTER RESULTS The BioFire System is the new standard for syndromic infectious disease diagnostics . Simple, fast, and comprehensive, the BioFire System delivers accurate results in about one hour. The right test, the first time can impact all areas of patient care. Healthcare providers can see faster diagnoses and improved antibiotic stewardship. Labs can see improved efficiency and reduced costs and downstream testing.LEARN MORE
The BioFire® FilmArray® Panels The FDA-cleared BioFire System panels test for viruses, bacteria, parasites, yeast, and antimicrobial resistance genes. Whether you’re trying to determine optimal therapy for a septic patient or pinpoint which respiratory pathogen is making a young child sick, the BioFire System can provide definitive answers—fast.READ MORE
Link to: The BioFire® Respiratory 2.1 (RP2.1) PanelRESPIRATORY
Link to: The BioFire® FilmArray® Blood Culture Identification (BCID)Panel
BLOOD CULTURE ID
Link to: The BioFire® FilmArray® Gastrointestinal (GI) PanelGASTROINTESTINAL
Link to: The BioFire® FilmArray® Meningitis/Encephalitis (ME) PanelMeningitis/
Encephalitis
Link to: The BioFire® FilmArray® Pneumonia PanelPNEUMONIA
BIOFIRE DIAGNOSTICS
LEADING THE WAY
With more than 25 years of molecular experience, BioFire Diagnostics, LLC sets the standard for molecular diagnostics through its pioneering advances in syndromic infectious disease testing. As bioMérieux’s Global Center of Excellence for Molecular Diagnostics, no other company has FDA-cleared and CE-IVD marked assays for more pathogens. BioFire Diagnostics has produced the easiest, fastest, and most comprehensive multiplex PCR testing available: the BioFire System.LEARN MORE
REQUEST A BIOFIRE SYSTEM DEMO Are you ready to order the BioFire System or want to request a free demo? Click below to take the next step towards faster results. Our team is ready to answer all of your questions.REQUEST A DEMO
* Products
* Solutions
* Company
* Media
* Support
* Get A Quote
* Blog
� Copyright BioFire Diagnostics 2020. All Rights Reserved | PrivacyPolicy | Legal
* Youtube
* Products
* Solutions
* Company
* Media
* Support
* Get A Quote
* Blog
Scroll to top
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0