Are you over 18 and want to see adult content?
More Annotations
The Perfect Steak Co. Official Website
Are you over 18 and want to see adult content?
A complete backup of lexus-mapupdates.eu
Are you over 18 and want to see adult content?

Bagdoom.com | Fashion, watches, electronics | Online shopping BD
Are you over 18 and want to see adult content?

The Conversation: In-depth analysis, research, news and ideas from leading academics and researchers.
Are you over 18 and want to see adult content?

Незнайка — ЕГЭ, ОГЭ, ВПР 2019 и Итоговое сочинение
Are you over 18 and want to see adult content?
iTaxi.pl - Aplikacja taxi online - Pobierz i zamów taksówkę przez Internet
Are you over 18 and want to see adult content?

Broker Looker – the chain between brokers and industrial links
Are you over 18 and want to see adult content?

A complete backup of eisimplementinc.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of wahl-regional.de
Are you over 18 and want to see adult content?
A complete backup of mystampready.com
Are you over 18 and want to see adult content?

A complete backup of digital58.com.ve
Are you over 18 and want to see adult content?
A complete backup of orfoepiyalugeti.blogspot.com
Are you over 18 and want to see adult content?
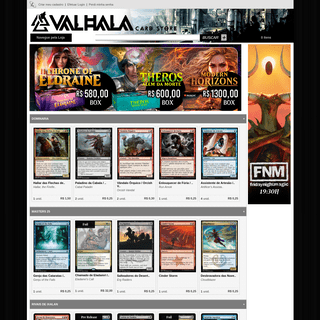
A complete backup of valhalastore.com.br
Are you over 18 and want to see adult content?
Text
LABELING LOGISTICS
Reed Tech is a true partner – an extension of our internal resources. Their software solutions are enabling us to successfully meet UDI compliance dates in the US and UK.”. “Reed Tech has experts in Drug Listing and UDI and we can always count on them for their guidance for the best approach to be taken when needed.”. ABOUT US | REGULATORY PROFESSIONALS IN MEDICAL DEVICES, RX The Reed Tech Life Sciences team has an employee sitting on the standards setting board of this organization. Health Level Seven International (HL7) is a not-for-profit, ANSI-accredited standards developing organization dedicated to providing a comprehensive framework and related standards for the exchange, integration, sharing, and retrieval of electronic health information that supports LEXISNEXIS REED TECH Our mission & purpose. At LexisNexis Reed Tech, our mission and purpose is to enable the advancement of humanity by delivering better outcomes to the innovation community. Each and every day, the work our Team does supports the development of new technologies and CAREERS | LEXISNEXIS REED TECH LexisNexis Reed Tech, a division of RELX, is an equal opportunity employer: qualified applicants are considered for and treated during employment without regard to race, color, creed, religion, sex, national origin, citizenship status, disability status, protected veteran status, age, marital status, sexual orientation, gender identity, genetic information, or any other characteristic NAVIGATOR™ FOR MEDICAL DEVICES Navigator™ for Medical Devices is a unique, best-in-class solution designed to help: Find the best predicates to expedite your 510 (k) applications and discover and monitor safety events. Easily identify predicate devices to bring a new device to market through the FDA’s 510 (k) application process. Find the best predicates for your client FDA CLASS I UDI SUBMISSION DEADLINE Class I devices and devices that have not been classified into class I, class II or class III that are required to be labeled with a UDI code, must submit product data to the FDA’s GUDID by September 24, 2022 (moved from September 24, 2020).FDA announced a two-year shift of the enforcement of this compliance date on June 30, 2020. UDI LABELING (UNIQUE DEVICE IDENTIFICATION): BEST Unique Device Identification (UDI) labeling remains a hot topic among medical device manufacturers under pressure to comply with UDI requirements for US FDA GUDID and other emerging health authorities around the globe. EU EUDAMED is underway and China’s NMPA, South Korea and others have published guidelines.UDI is becoming integrated into medical device labeling all over the FDA CHANGES FOR PRIVATE LABEL DISTRIBUTORS AND CONTRACT These categories were established for listings performed by a CMO under its own NDC Labeler Code (read more about FDA’s drug listing requirements for CMOs here).Although each category references the CMO’s relationship with the distributor of the product (PLD), the FDA has recognized that the naming of these categories has resulted in confusion within the industry. NHS EPROCUREMENT REQUIREMENTS, TIMELINES AND STRATEGIES Live. •. Join Gary Saner and John Lorenc of Reed Tech for a discussion of the UK National Health Service’s (NHS) eProcurement requirements and how affected medical device labelers can comply. Read more about NHS eProcurement in this Q & A article. EXECUTIVE LEADERS RADIO SPOTLIGHTS ETHAN EISNER, PRESIDENT Listen Above. In this 10-minute interview, Ethan discusses the early stages of his career and how that informs his current role as the leader of Reed Tech. Listen above and REED TECH | BEST-IN-CLASS INFORMATION-BASED SOLUTIONS ANDCORPORATE RESPONSIBILITYMEDICAL DEVICE COMPLIANCE SOLUTIONSSTRUCTURED PRODUCTLABELING LOGISTICS
Reed Tech is a true partner – an extension of our internal resources. Their software solutions are enabling us to successfully meet UDI compliance dates in the US and UK.”. “Reed Tech has experts in Drug Listing and UDI and we can always count on them for their guidance for the best approach to be taken when needed.”. ABOUT US | REGULATORY PROFESSIONALS IN MEDICAL DEVICES, RX The Reed Tech Life Sciences team has an employee sitting on the standards setting board of this organization. Health Level Seven International (HL7) is a not-for-profit, ANSI-accredited standards developing organization dedicated to providing a comprehensive framework and related standards for the exchange, integration, sharing, and retrieval of electronic health information that supports LEXISNEXIS REED TECH Our mission & purpose. At LexisNexis Reed Tech, our mission and purpose is to enable the advancement of humanity by delivering better outcomes to the innovation community. Each and every day, the work our Team does supports the development of new technologies and CAREERS | LEXISNEXIS REED TECH LexisNexis Reed Tech, a division of RELX, is an equal opportunity employer: qualified applicants are considered for and treated during employment without regard to race, color, creed, religion, sex, national origin, citizenship status, disability status, protected veteran status, age, marital status, sexual orientation, gender identity, genetic information, or any other characteristic NAVIGATOR™ FOR MEDICAL DEVICES Navigator™ for Medical Devices is a unique, best-in-class solution designed to help: Find the best predicates to expedite your 510 (k) applications and discover and monitor safety events. Easily identify predicate devices to bring a new device to market through the FDA’s 510 (k) application process. Find the best predicates for your client FDA CLASS I UDI SUBMISSION DEADLINE Class I devices and devices that have not been classified into class I, class II or class III that are required to be labeled with a UDI code, must submit product data to the FDA’s GUDID by September 24, 2022 (moved from September 24, 2020).FDA announced a two-year shift of the enforcement of this compliance date on June 30, 2020. UDI LABELING (UNIQUE DEVICE IDENTIFICATION): BEST Unique Device Identification (UDI) labeling remains a hot topic among medical device manufacturers under pressure to comply with UDI requirements for US FDA GUDID and other emerging health authorities around the globe. EU EUDAMED is underway and China’s NMPA, South Korea and others have published guidelines.UDI is becoming integrated into medical device labeling all over the FDA CHANGES FOR PRIVATE LABEL DISTRIBUTORS AND CONTRACT These categories were established for listings performed by a CMO under its own NDC Labeler Code (read more about FDA’s drug listing requirements for CMOs here).Although each category references the CMO’s relationship with the distributor of the product (PLD), the FDA has recognized that the naming of these categories has resulted in confusion within the industry. NHS EPROCUREMENT REQUIREMENTS, TIMELINES AND STRATEGIES Live. •. Join Gary Saner and John Lorenc of Reed Tech for a discussion of the UK National Health Service’s (NHS) eProcurement requirements and how affected medical device labelers can comply. Read more about NHS eProcurement in this Q & A article. EXECUTIVE LEADERS RADIO SPOTLIGHTS ETHAN EISNER, PRESIDENT Listen Above. In this 10-minute interview, Ethan discusses the early stages of his career and how that informs his current role as the leader of Reed Tech. Listen above and OVER-THE-COUNTER DRUG PRODUCT MONOGRAPHS: WHAT YOU NEED TO Register here for the next webinar in our Over-the-Counter Drug Monograph series taking place on June 16, 2021 at 2 PM ET- Over-the-Counter Drug Product Monographs: FDA Requirements for Hand Sanitizers and other COVID-19-focused products.. To learn more, please call us at +215-557-3010 or email us at . SINGLESOURCE™ FOR MEDICAL DEVICES SingleSource™ for Medical Devices Features. Central Product Data Management. Store product data in a secure cloud repository. Ensure master data values are current, accurate and comprehensive. Assign roles and permissions for corporate and in-country legal representatives. Simple Data Syndication. UNIQUE DEVICE IDENTIFICATION (UDI) Easily maintain a source data set and provide accurate product data to regulatory authorities and customers. Reed Tech is a leading FDA supplier of Unique Device Identification (UDI) information, submitting close to one-quarter of all NLM Access GUDID UDI records annually. With SingleSource™ for Medical Devices, UDI data is managedthroughout
PRODUCT LOGIN PAGE
LexisNexis, a division of RELX Inc., may contact you in your professional capacity with information about our other products, services and events that we believe may be of interest. ELECTRONIC DRUG REGISTRATION AND LISTING 3 Introduction 4 About FDA’s Electronic Drug Registration and Listing (eDRL) 5 Manufacturers’ Listings and Distributor’s Listings 6 Who is responsible to list a drug product 7 NDC Directory and DailyMed Publication 8 Manufacturer’s Listings Enforcement and Import Issues 9 Drug Listing and National Drug Codes (NDCs) 10 Prerequisites for drug listing submission FDA SPL - STRUCTURED PRODUCT & DRUG LABELING COMPOSITION Reed Tech offers a process to use the Content of Labeling portion of the FDA-accepted SPL Drug Listing file as the source for: Structured Product Labeling (SPL) is a Health Level Seven (HL7) International standard for regulatory guidance documents as a method REED TECH INSIGHTS: WHAT IS UDI-DI AND BUDI-DI? Put simply, BUDI-DI is focused on categorizing medical devices that use the same identifier in the same group. BUDI is the main identifier of a model, while the DI is the specific device unit. However, there is a lot more to BUDI-DI, especially when looking at the differences between BUDI-DI and UDI-DI, and BUDI’s use in the EU.KNOWLEDGE CENTER
Drug & Biologic Product Submissions. This page provides blogs, short videos and recordings focused on industry insights for regulatory requirements and best practices for electronic submission compliance for manufacturers and distributors of drug and biologic products. SAUDI ARABIA (SFDA) UNIQUE DEVICE IDENTIFICATION UPDATE Saudi Food and Drug Authority (SFDA) issued final “Guidance on Requirements for Unique Device Identification (UDI) for Medical Devices” (MDS-G34) (MDS-G34 at HIBCC) in Apri l 2019. NHS EPROCUREMENT REQUIREMENTS, TIMELINES AND STRATEGIES Live. •. Join Gary Saner and John Lorenc of Reed Tech for a discussion of the UK National Health Service’s (NHS) eProcurement requirements and how affected medical device labelers can comply. Read more about NHS eProcurement in this Q & A article. REED TECH | BEST-IN-CLASS INFORMATION-BASED SOLUTIONS ANDCORPORATE RESPONSIBILITYMEDICAL DEVICE COMPLIANCE SOLUTIONSSTRUCTURED PRODUCTLABELING LOGISTICS
Reed Tech is a true partner – an extension of our internal resources. Their software solutions are enabling us to successfully meet UDI compliance dates in the US and UK.”. “Reed Tech has experts in Drug Listing and UDI and we can always count on them for their guidance for the best approach to be taken when needed.”. ABOUT US | REGULATORY PROFESSIONALS IN MEDICAL DEVICES, RX The Reed Tech Life Sciences team has an employee sitting on the standards setting board of this organization. Health Level Seven International (HL7) is a not-for-profit, ANSI-accredited standards developing organization dedicated to providing a comprehensive framework and related standards for the exchange, integration, sharing, and retrieval of electronic health information that supports LEXISNEXIS REED TECH Our mission & purpose. At LexisNexis Reed Tech, our mission and purpose is to enable the advancement of humanity by delivering better outcomes to the innovation community. Each and every day, the work our Team does supports the development of new technologies and CAREERS | LEXISNEXIS REED TECH LexisNexis Reed Tech, a division of RELX, is an equal opportunity employer: qualified applicants are considered for and treated during employment without regard to race, color, creed, religion, sex, national origin, citizenship status, disability status, protected veteran status, age, marital status, sexual orientation, gender identity, genetic information, or any other characteristic NAVIGATOR™ FOR MEDICAL DEVICES Navigator™ for Medical Devices is a unique, best-in-class solution designed to help: Find the best predicates to expedite your 510 (k) applications and discover and monitor safety events. Easily identify predicate devices to bring a new device to market through the FDA’s 510 (k) application process. Find the best predicates for your client SINGLESOURCE™ FOR MEDICAL DEVICES SingleSource™ for Medical Devices Features. Central Product Data Management. Store product data in a secure cloud repository. Ensure master data values are current, accurate and comprehensive. Assign roles and permissions for corporate and in-country legal representatives. Simple Data Syndication. FDA CLASS I UDI SUBMISSION DEADLINE Class I devices and devices that have not been classified into class I, class II or class III that are required to be labeled with a UDI code, must submit product data to the FDA’s GUDID by September 24, 2022 (moved from September 24, 2020).FDA announced a two-year shift of the enforcement of this compliance date on June 30, 2020. UDI LABELING (UNIQUE DEVICE IDENTIFICATION): BEST Unique Device Identification (UDI) labeling remains a hot topic among medical device manufacturers under pressure to comply with UDI requirements for US FDA GUDID and other emerging health authorities around the globe. EU EUDAMED is underway and China’s NMPA, South Korea and others have published guidelines.UDI is becoming integrated into medical device labeling all over the FDA CHANGES FOR PRIVATE LABEL DISTRIBUTORS AND CONTRACT These categories were established for listings performed by a CMO under its own NDC Labeler Code (read more about FDA’s drug listing requirements for CMOs here).Although each category references the CMO’s relationship with the distributor of the product (PLD), the FDA has recognized that the naming of these categories has resulted in confusion within the industry. NHS EPROCUREMENT REQUIREMENTS, TIMELINES AND STRATEGIES Live. •. Join Gary Saner and John Lorenc of Reed Tech for a discussion of the UK National Health Service’s (NHS) eProcurement requirements and how affected medical device labelers can comply. Read more about NHS eProcurement in this Q & A article. REED TECH | BEST-IN-CLASS INFORMATION-BASED SOLUTIONS ANDCORPORATE RESPONSIBILITYMEDICAL DEVICE COMPLIANCE SOLUTIONSSTRUCTURED PRODUCTLABELING LOGISTICS
Reed Tech is a true partner – an extension of our internal resources. Their software solutions are enabling us to successfully meet UDI compliance dates in the US and UK.”. “Reed Tech has experts in Drug Listing and UDI and we can always count on them for their guidance for the best approach to be taken when needed.”. ABOUT US | REGULATORY PROFESSIONALS IN MEDICAL DEVICES, RX The Reed Tech Life Sciences team has an employee sitting on the standards setting board of this organization. Health Level Seven International (HL7) is a not-for-profit, ANSI-accredited standards developing organization dedicated to providing a comprehensive framework and related standards for the exchange, integration, sharing, and retrieval of electronic health information that supports LEXISNEXIS REED TECH Our mission & purpose. At LexisNexis Reed Tech, our mission and purpose is to enable the advancement of humanity by delivering better outcomes to the innovation community. Each and every day, the work our Team does supports the development of new technologies and CAREERS | LEXISNEXIS REED TECH LexisNexis Reed Tech, a division of RELX, is an equal opportunity employer: qualified applicants are considered for and treated during employment without regard to race, color, creed, religion, sex, national origin, citizenship status, disability status, protected veteran status, age, marital status, sexual orientation, gender identity, genetic information, or any other characteristic NAVIGATOR™ FOR MEDICAL DEVICES Navigator™ for Medical Devices is a unique, best-in-class solution designed to help: Find the best predicates to expedite your 510 (k) applications and discover and monitor safety events. Easily identify predicate devices to bring a new device to market through the FDA’s 510 (k) application process. Find the best predicates for your client SINGLESOURCE™ FOR MEDICAL DEVICES SingleSource™ for Medical Devices Features. Central Product Data Management. Store product data in a secure cloud repository. Ensure master data values are current, accurate and comprehensive. Assign roles and permissions for corporate and in-country legal representatives. Simple Data Syndication. FDA CLASS I UDI SUBMISSION DEADLINE Class I devices and devices that have not been classified into class I, class II or class III that are required to be labeled with a UDI code, must submit product data to the FDA’s GUDID by September 24, 2022 (moved from September 24, 2020).FDA announced a two-year shift of the enforcement of this compliance date on June 30, 2020. UDI LABELING (UNIQUE DEVICE IDENTIFICATION): BEST Unique Device Identification (UDI) labeling remains a hot topic among medical device manufacturers under pressure to comply with UDI requirements for US FDA GUDID and other emerging health authorities around the globe. EU EUDAMED is underway and China’s NMPA, South Korea and others have published guidelines.UDI is becoming integrated into medical device labeling all over the FDA CHANGES FOR PRIVATE LABEL DISTRIBUTORS AND CONTRACT These categories were established for listings performed by a CMO under its own NDC Labeler Code (read more about FDA’s drug listing requirements for CMOs here).Although each category references the CMO’s relationship with the distributor of the product (PLD), the FDA has recognized that the naming of these categories has resulted in confusion within the industry. NHS EPROCUREMENT REQUIREMENTS, TIMELINES AND STRATEGIES Live. •. Join Gary Saner and John Lorenc of Reed Tech for a discussion of the UK National Health Service’s (NHS) eProcurement requirements and how affected medical device labelers can comply. Read more about NHS eProcurement in this Q & A article. OVER-THE-COUNTER DRUG PRODUCT MONOGRAPHS: WHAT YOU NEED TO Register here for the next webinar in our Over-the-Counter Drug Monograph series taking place on June 16, 2021 at 2 PM ET- Over-the-Counter Drug Product Monographs: FDA Requirements for Hand Sanitizers and other COVID-19-focused products.. To learn more, please call us at +215-557-3010 or email us at . UNIQUE DEVICE IDENTIFICATION (UDI) Easily maintain a source data set and provide accurate product data to regulatory authorities and customers. Reed Tech is a leading FDA supplier of Unique Device Identification (UDI) information, submitting close to one-quarter of all NLM Access GUDID UDI records annually. With SingleSource™ for Medical Devices, UDI data is managedthroughout
PRODUCT LOGIN PAGE
LexisNexis, a division of RELX Inc., may contact you in your professional capacity with information about our other products, services and events that we believe may be of interest. ELECTRONIC DRUG REGISTRATION AND LISTING 3 Introduction 4 About FDA’s Electronic Drug Registration and Listing (eDRL) 5 Manufacturers’ Listings and Distributor’s Listings 6 Who is responsible to list a drug product 7 NDC Directory and DailyMed Publication 8 Manufacturer’s Listings Enforcement and Import Issues 9 Drug Listing and National Drug Codes (NDCs) 10 Prerequisites for drug listing submission REED TECH INSIGHTS: WHAT IS UDI-DI AND BUDI-DI? Put simply, BUDI-DI is focused on categorizing medical devices that use the same identifier in the same group. BUDI is the main identifier of a model, while the DI is the specific device unit. However, there is a lot more to BUDI-DI, especially when looking at the differences between BUDI-DI and UDI-DI, and BUDI’s use in the EU. FDA SPL - STRUCTURED PRODUCT & DRUG LABELING COMPOSITION Reed Tech offers a process to use the Content of Labeling portion of the FDA-accepted SPL Drug Listing file as the source for: Structured Product Labeling (SPL) is a Health Level Seven (HL7) International standard for regulatory guidance documents as a methodKNOWLEDGE CENTER
Drug & Biologic Product Submissions. This page provides blogs, short videos and recordings focused on industry insights for regulatory requirements and best practices for electronic submission compliance for manufacturers and distributors of drug and biologic products. SAUDI ARABIA (SFDA) UNIQUE DEVICE IDENTIFICATION UPDATE Saudi Food and Drug Authority (SFDA) issued final “Guidance on Requirements for Unique Device Identification (UDI) for Medical Devices” (MDS-G34) (MDS-G34 at HIBCC) in Apri l 2019. LS - PHARMA - WEBINARS - UNDERSTANDING REMS IN SPL The corporate mission is to advance humanity by delivering better outcomes to the innovation community. Reed Tech is a LexisNexis company. For more information: Pharma@ReedTech.com NHS EPROCUREMENT REQUIREMENTS, TIMELINES AND STRATEGIES Live. •. Join Gary Saner and John Lorenc of Reed Tech for a discussion of the UK National Health Service’s (NHS) eProcurement requirements and how affected medical device labelers can comply. Read more about NHS eProcurement in this Q & A article. REED TECH | BEST-IN-CLASS INFORMATION-BASED SOLUTIONS ANDCORPORATE RESPONSIBILITYMEDICAL DEVICE COMPLIANCE SOLUTIONSSTRUCTURED PRODUCTLABELING LOGISTICS
Reed Tech is a true partner – an extension of our internal resources. Their software solutions are enabling us to successfully meet UDI compliance dates in the US and UK.”. “Reed Tech has experts in Drug Listing and UDI and we can always count on them for their guidance for the best approach to be taken when needed.”. ABOUT US | REGULATORY PROFESSIONALS IN MEDICAL DEVICES, RX The Reed Tech Life Sciences team has an employee sitting on the standards setting board of this organization. Health Level Seven International (HL7) is a not-for-profit, ANSI-accredited standards developing organization dedicated to providing a comprehensive framework and related standards for the exchange, integration, sharing, and retrieval of electronic health information that supports LEXISNEXIS REED TECH Our mission & purpose. At LexisNexis Reed Tech, our mission and purpose is to enable the advancement of humanity by delivering better outcomes to the innovation community. Each and every day, the work our Team does supports the development of new technologies and CAREERS | LEXISNEXIS REED TECH LexisNexis Reed Tech, a division of RELX, is an equal opportunity employer: qualified applicants are considered for and treated during employment without regard to race, color, creed, religion, sex, national origin, citizenship status, disability status, protected veteran status, age, marital status, sexual orientation, gender identity, genetic information, or any other characteristic NAVIGATOR™ FOR MEDICAL DEVICES Navigator™ for Medical Devices is a unique, best-in-class solution designed to help: Find the best predicates to expedite your 510 (k) applications and discover and monitor safety events. Easily identify predicate devices to bring a new device to market through the FDA’s 510 (k) application process. Find the best predicates for your client SINGLESOURCE™ FOR MEDICAL DEVICES SingleSource™ for Medical Devices Features. Central Product Data Management. Store product data in a secure cloud repository. Ensure master data values are current, accurate and comprehensive. Assign roles and permissions for corporate and in-country legal representatives. Simple Data Syndication. FDA SPL - STRUCTURED PRODUCT & DRUG LABELING COMPOSITION Reed Tech offers a process to use the Content of Labeling portion of the FDA-accepted SPL Drug Listing file as the source for: Structured Product Labeling (SPL) is a Health Level Seven (HL7) International standard for regulatory guidance documents as a method UDI LABELING (UNIQUE DEVICE IDENTIFICATION): BEST Unique Device Identification (UDI) labeling remains a hot topic among medical device manufacturers under pressure to comply with UDI requirements for US FDA GUDID and other emerging health authorities around the globe. EU EUDAMED is underway and China’s NMPA, South Korea and others have published guidelines.UDI is becoming integrated into medical device labeling all over the FDA CHANGES FOR PRIVATE LABEL DISTRIBUTORS AND CONTRACT These categories were established for listings performed by a CMO under its own NDC Labeler Code (read more about FDA’s drug listing requirements for CMOs here).Although each category references the CMO’s relationship with the distributor of the product (PLD), the FDA has recognized that the naming of these categories has resulted in confusion within the industry. SAUDI ARABIA (SFDA) UNIQUE DEVICE IDENTIFICATION UPDATE Saudi Food and Drug Authority (SFDA) issued final “Guidance on Requirements for Unique Device Identification (UDI) for Medical Devices” (MDS-G34) (MDS-G34 at HIBCC) in Apri l 2019. REED TECH | BEST-IN-CLASS INFORMATION-BASED SOLUTIONS ANDCORPORATE RESPONSIBILITYMEDICAL DEVICE COMPLIANCE SOLUTIONSSTRUCTURED PRODUCTLABELING LOGISTICS
Reed Tech is a true partner – an extension of our internal resources. Their software solutions are enabling us to successfully meet UDI compliance dates in the US and UK.”. “Reed Tech has experts in Drug Listing and UDI and we can always count on them for their guidance for the best approach to be taken when needed.”. ABOUT US | REGULATORY PROFESSIONALS IN MEDICAL DEVICES, RX The Reed Tech Life Sciences team has an employee sitting on the standards setting board of this organization. Health Level Seven International (HL7) is a not-for-profit, ANSI-accredited standards developing organization dedicated to providing a comprehensive framework and related standards for the exchange, integration, sharing, and retrieval of electronic health information that supports LEXISNEXIS REED TECH Our mission & purpose. At LexisNexis Reed Tech, our mission and purpose is to enable the advancement of humanity by delivering better outcomes to the innovation community. Each and every day, the work our Team does supports the development of new technologies and CAREERS | LEXISNEXIS REED TECH LexisNexis Reed Tech, a division of RELX, is an equal opportunity employer: qualified applicants are considered for and treated during employment without regard to race, color, creed, religion, sex, national origin, citizenship status, disability status, protected veteran status, age, marital status, sexual orientation, gender identity, genetic information, or any other characteristic NAVIGATOR™ FOR MEDICAL DEVICES Navigator™ for Medical Devices is a unique, best-in-class solution designed to help: Find the best predicates to expedite your 510 (k) applications and discover and monitor safety events. Easily identify predicate devices to bring a new device to market through the FDA’s 510 (k) application process. Find the best predicates for your client SINGLESOURCE™ FOR MEDICAL DEVICES SingleSource™ for Medical Devices Features. Central Product Data Management. Store product data in a secure cloud repository. Ensure master data values are current, accurate and comprehensive. Assign roles and permissions for corporate and in-country legal representatives. Simple Data Syndication. FDA SPL - STRUCTURED PRODUCT & DRUG LABELING COMPOSITION Reed Tech offers a process to use the Content of Labeling portion of the FDA-accepted SPL Drug Listing file as the source for: Structured Product Labeling (SPL) is a Health Level Seven (HL7) International standard for regulatory guidance documents as a method UDI LABELING (UNIQUE DEVICE IDENTIFICATION): BEST Unique Device Identification (UDI) labeling remains a hot topic among medical device manufacturers under pressure to comply with UDI requirements for US FDA GUDID and other emerging health authorities around the globe. EU EUDAMED is underway and China’s NMPA, South Korea and others have published guidelines.UDI is becoming integrated into medical device labeling all over the FDA CHANGES FOR PRIVATE LABEL DISTRIBUTORS AND CONTRACT These categories were established for listings performed by a CMO under its own NDC Labeler Code (read more about FDA’s drug listing requirements for CMOs here).Although each category references the CMO’s relationship with the distributor of the product (PLD), the FDA has recognized that the naming of these categories has resulted in confusion within the industry. SAUDI ARABIA (SFDA) UNIQUE DEVICE IDENTIFICATION UPDATE Saudi Food and Drug Authority (SFDA) issued final “Guidance on Requirements for Unique Device Identification (UDI) for Medical Devices” (MDS-G34) (MDS-G34 at HIBCC) in Apri l 2019.PRODUCT LOGIN PAGE
LexisNexis, a division of RELX Inc., may contact you in your professional capacity with information about our other products, services and events that we believe may be of interest. UNIQUE DEVICE IDENTIFICATION (UDI) Easily maintain a source data set and provide accurate product data to regulatory authorities and customers. Reed Tech is a leading FDA supplier of Unique Device Identification (UDI) information, submitting close to one-quarter of all NLM Access GUDID UDI records annually. With SingleSource™ for Medical Devices, UDI data is managedthroughout
ALL BLOG POSTS
Reed Tech® Introduces Reed Tech Navigator™ for Medical Devices. Apr 10, 2017. Horsham, Penn. – April 10, 2017 – Reed Technology and Information Services Inc. (Reed Tech), a leader in data analytics and information services for the Life Sciences industry, announces the launch of Reed Tech Navigator™ for Medical Devices.OUR COMPANY
Our Company. Bringing together data and analytics to keep our customers innovating. At LexisNexis Reed Tech, our mission and purpose is to enable the advancement of humanity by delivering better outcomes to the innovation community. Our business is a division of LexisNexis Legal and Professional, a leading global provider of legal, regulatory ELECTRONIC DRUG REGISTRATION AND LISTING 3 Introduction 4 About FDA’s Electronic Drug Registration and Listing (eDRL) 5 Manufacturers’ Listings and Distributor’s Listings 6 Who is responsible to list a drug product 7 NDC Directory and DailyMed Publication 8 Manufacturer’s Listings Enforcement and Import Issues 9 Drug Listing and National Drug Codes (NDCs) 10 Prerequisites for drug listing submission NAVIGATOR™ FOR DRUG LABELS Navigator™ for Drug Labels helps pharmaceutical regulatory researchers ensure accurate and up-to-date drug label content. Search the database using a single word or leverage up to seven categories and 90+ parameters to build a multi-dimensional search.KNOWLEDGE CENTER
Drug & Biologic Product Submissions. This page provides blogs, short videos and recordings focused on industry insights for regulatory requirements and best practices for electronic submission compliance for manufacturers and distributors of drug and biologic products. REED TECH INSIGHTS: WHAT IS UDI-DI AND BUDI-DI? Put simply, BUDI-DI is focused on categorizing medical devices that use the same identifier in the same group. BUDI is the main identifier of a model, while the DI is the specific device unit. However, there is a lot more to BUDI-DI, especially when looking at the differences between BUDI-DI and UDI-DI, and BUDI’s use in the EU. NHS EPROCUREMENT REQUIREMENTS, TIMELINES AND STRATEGIES Live. •. Join Gary Saner and John Lorenc of Reed Tech for a discussion of the UK National Health Service’s (NHS) eProcurement requirements and how affected medical device labelers can comply. Read more about NHS eProcurement in this Q & A article. REEDTECH PASSWORD RECOVERY Enter Your User Name. Enter your user name (for example, domain\username or username@domain): Enter the characters you see on the picture. Get new image.Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0