Are you over 18 and want to see adult content?
More Annotations

A complete backup of itcentralstation.com
Are you over 18 and want to see adult content?
A complete backup of konicaminolta.com.cn
Are you over 18 and want to see adult content?
A complete backup of animalaigo-portal.info
Are you over 18 and want to see adult content?
A complete backup of sciencespacerobots.com
Are you over 18 and want to see adult content?
A complete backup of topcelebrityfakes.com
Are you over 18 and want to see adult content?
A complete backup of metropoliabierta.com
Are you over 18 and want to see adult content?
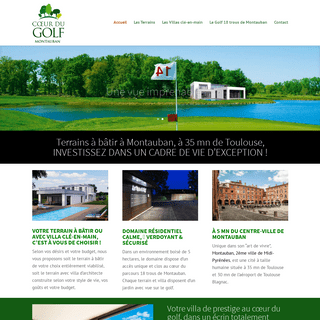
A complete backup of lecoeurdugolf.fr
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of comofuncionaque.com
Are you over 18 and want to see adult content?
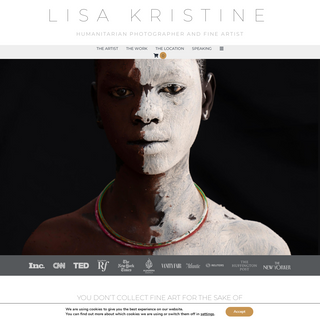
A complete backup of lisakristine.com
Are you over 18 and want to see adult content?

A complete backup of centralhousing.com.mx
Are you over 18 and want to see adult content?

A complete backup of saratovairlines.ru
Are you over 18 and want to see adult content?
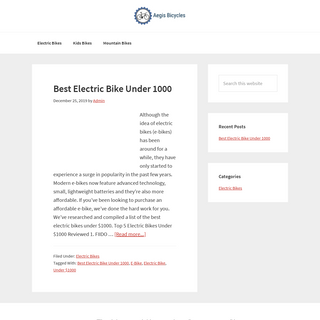
A complete backup of aegisbicycles.com
Are you over 18 and want to see adult content?
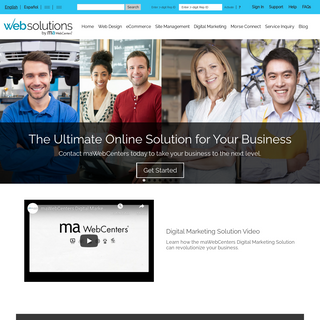
A complete backup of mawebcenters.com
Are you over 18 and want to see adult content?
Text
VONOPRAZAN FUMARATE
Vonoprazan Fumarate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on December 26, 2014. It was co-developed and marketed as Takecab ® by Takeda & Otsuka. Vonoprazan has a novel mechanism of action called potassium-competitive acid blockers (P-CABs) which competitively inhibits the binding the potassium ions to H +, K +-ATPase (also known as the 医药研发领域大数据信息平台 药渡是一家专注于医药研发领域的“互联网+”大数据信息平台,我们致力于整合药物研发相关数十个学科专业数据和资源信息,为全球药物研发人员提供“一站式信息解决方案”,助力创新,加速创造。 OLANEXIDINE GLUCONATE 12 rows · Olanexidine gluconate was approved by Pharmaceuticals and Medical Devices Agency of REVAPRAZAN HYDROCHLORIDE Revaprazan hydrochloride was approved by Korea Food and Drug Administration (KFDA) on September 1, 2005. It was developed and marketed as Revanex ® by Yuhan Corporation in KR.. Revaprazan is the first acid pump antagonist with a function similar to that of proton pump inhibitors (PPIs).RIVAROXABAN
51 rows · Rivaroxaban was first approved by Health Canada on Septemper 15, 2008, then approved by LANDIOLOL HYDROCHLORIDE Landiolol hydrochloride was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jul 5, 2002. It was developed and marketed as Onoact ® by Ono in Japan.. Landiolol is a highly cardioselective, ultra short-acting beta blocker. BOSUTINIB MONOHYDRATE Bosutinib hydrate was approved by the U.S. Food and Drug Administration (FDA) on September 4, 2012, then approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Sep 26, 2014, and approved by European Medicine Agency (EMA) on Mar 27, 2013. POLMACOXIB - PHARMACODIA.COM Polmacoxib was approved by the Korean Ministry of Food and Drug Safety (MFDS) on Feb 05, 2015. It was originally developed by CrystalGenomics, then it signed an agreement for the commercialization of polmacoxib in South Korea with Dong-A in August. EDOXABAN TOSILATE HYDRATE Edoxaban tosilate hydrate was first approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on April 22, 2011, then approved by the U.S. Food and Drug Administration (FDA) on January 8, 2015, and approved by European Medicine Agency (EMA) on June 19, 2015. ETANERCEPT - PHARMACODIA.COM Etanercept was first approved by the U.S. Food and Drug Administration (FDA) on November 2, 1998, then approved by European Medicine Agency (EMA) on February 3, 2000, Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on January 19, 2005, China Food and Drug Administration (CFDA) on February 26, 2010.VONOPRAZAN FUMARATE
Vonoprazan Fumarate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on December 26, 2014. It was co-developed and marketed as Takecab ® by Takeda & Otsuka. Vonoprazan has a novel mechanism of action called potassium-competitive acid blockers (P-CABs) which competitively inhibits the binding the potassium ions to H +, K +-ATPase (also known as the 医药研发领域大数据信息平台 药渡是一家专注于医药研发领域的“互联网+”大数据信息平台,我们致力于整合药物研发相关数十个学科专业数据和资源信息,为全球药物研发人员提供“一站式信息解决方案”,助力创新,加速创造。 OLANEXIDINE GLUCONATE 12 rows · Olanexidine gluconate was approved by Pharmaceuticals and Medical Devices Agency of REVAPRAZAN HYDROCHLORIDE Revaprazan hydrochloride was approved by Korea Food and Drug Administration (KFDA) on September 1, 2005. It was developed and marketed as Revanex ® by Yuhan Corporation in KR.. Revaprazan is the first acid pump antagonist with a function similar to that of proton pump inhibitors (PPIs).RIVAROXABAN
51 rows · Rivaroxaban was first approved by Health Canada on Septemper 15, 2008, then approved by LANDIOLOL HYDROCHLORIDE Landiolol hydrochloride was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jul 5, 2002. It was developed and marketed as Onoact ® by Ono in Japan.. Landiolol is a highly cardioselective, ultra short-acting beta blocker. BOSUTINIB MONOHYDRATE Bosutinib hydrate was approved by the U.S. Food and Drug Administration (FDA) on September 4, 2012, then approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Sep 26, 2014, and approved by European Medicine Agency (EMA) on Mar 27, 2013. POLMACOXIB - PHARMACODIA.COM Polmacoxib was approved by the Korean Ministry of Food and Drug Safety (MFDS) on Feb 05, 2015. It was originally developed by CrystalGenomics, then it signed an agreement for the commercialization of polmacoxib in South Korea with Dong-A in August. EDOXABAN TOSILATE HYDRATE Edoxaban tosilate hydrate was first approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on April 22, 2011, then approved by the U.S. Food and Drug Administration (FDA) on January 8, 2015, and approved by European Medicine Agency (EMA) on June 19, 2015.TOFACITINIB CITRATE
Tofacitinib citrate was approved by the U.S. Food and Drug Administration (FDA) on November 6, 2012, and then approved by Pharmaceuticals and Medicals Devices Agency of ADUCANUMAB - PHARMACODIA.COM Aducanumab is an amyloid plaque targeted monoclonal antibody in phase III clinical trials for the intravenous treatment of patients with early Alzheimer's disease. MIRABEGRON - PHARMACODIA.COM Mirabegron was first approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on July 1, 2011, then approved by the U.S. Food and Drug Administration (FDA) on June 28, 2012, and approved by European Medicine Agency (EMA) on Dec 20, 2012. IGURATIMOD - PHARMACODIA.COM Iguratimod was first approved by China Food and Drug Administration (CFDA) on August 15, 2011, then approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012.ESFLURBIPROFEN
Esflurbiprofen was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Sep 28, 2015. It was developed by Taisho and marketed by Taisho and Teijin as Loqoa ® in Japan.. Esflurbiprofen is a cyclooxygenase (COX) inhibitor indicated OLANEXIDINE GLUCONATE Olanexidine gluconate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jul 03, 2015. It was developed and marketed as Olanedine ® by Otsuka in Japan.. Olanexidine gluconate is an antiseptic/disinfectant compound with potent bactericidal activity against Gram-negative and Gram-positive bacteria, for use in preparing patients for surgery and preventing of LANDIOLOL HYDROCHLORIDE Landiolol hydrochloride was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jul 5, 2002. It was developed and marketed as Onoact ® by Ono in Japan.. Landiolol is a highly cardioselective, ultra short-acting beta blocker.NARTOGRASTIM
Nartograstim was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on April 1, 1994. It was originally developed by Kyowa Hakko Kiri, then marketed as Neu-up ® by Yakult Honsha.. Nartograstim is a mutant recombinant human granulocyte colony-stimulating factor, which regulates the production of neutrophils within the bone marrow and affects neutrophil progenitorPEGFILGRASTIM
Pegfilgrastim was first approved by the U.S. Food and Drug Administration (FDA) on January 31, 2001, then approved by European Medicine Agency (EMA) on August 22, 2002, Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on September 26, 2014.LENVATINIB MESYLATE
Lenvatinib mesylate was first approved by the U.S. Food and Drug Administration (FDA) on Feb 13, 2015, then approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Mar 26, 2015, and approved by European Medicine Agency (EMA) on May 28, 2015. 医药研发领域大数据信息平台 药渡是一家专注于医药研发领域的“互联网+”大数据信息平台,我们致力于整合药物研发相关数十个学科专业数据和资源信息,为全球药物研发人员提供“一站式信息解决方案”,助力创新,加速创造。EFINACONAZOLE
Efinaconazole was first approved by Health Canada on October 2, 2013, then approved by the U.S. Food and Drug Administration (FDA) on June 6, 2014, and approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on July 4, 2014.VONOPRAZAN FUMARATE
Vonoprazan Fumarate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on December 26, 2014. It was co-developed and marketed as Takecab ® by Takeda & Otsuka. Vonoprazan has a novel mechanism of action called potassium-competitive acid blockers (P-CABs) which competitively inhibits the binding the potassium ions to H +, K +-ATPase (also known as the ETANERCEPT - PHARMACODIA.COM Etanercept was first approved by the U.S. Food and Drug Administration (FDA) on November 2, 1998, then approved by European Medicine Agency (EMA) on February 3, 2000, Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on January 19, 2005, China Food and Drug Administration (CFDA) on February 26, 2010. IBRUTINIB - PHARMACODIA.COM In Vivo Efficacy. In ex vivo studies, Btk activated site occupancy in splenocyte in ibrutinib pre-treated mice.. Xenograft models in SCID mice: OCL-Ly-10 cell lines: Significantly inhibited tumor growth at 3 mg/kg oral daily at day 10. Mantle cell lymphoma (MCL) model: Decreased the clinical signs, mean white blood cell (WBC) counts, collective lymph node weights, PBMC, lymph nodes and hCD19 POLMACOXIB - PHARMACODIA.COM Polmacoxib was approved by the Korean Ministry of Food and Drug Safety (MFDS) on Feb 05, 2015. It was originally developed by CrystalGenomics, then it signed an agreement for the commercialization of polmacoxib in South Korea with Dong-A in August. TENELIGLIPTIN HYDROBROMIDE HYDRATE Teneligliptin hydrobromide hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. It was co-developed & co-marketed as Tenelia ® by Mitsubishi Tanabe and Daiichi Sankyo.. Teneligliptin hydrobromide hydrate is a dipeptidyl peptidase-4 inhibitor. FASUDIL HYDROCHLORIDE HYDRATE Fasudil hydrochloride Hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 30, 1995. It was developed and marketed as Eril ® by Asahi Kasei in Japan.. Fasudil hydrochloride is a selective RhoA/Rho kinase (ROCK) inhibitor. BOSUTINIB MONOHYDRATE Bosutinib hydrate was approved by the U.S. Food and Drug Administration (FDA) on September 4, 2012, then approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Sep 26, 2014, and approved by European Medicine Agency (EMA) on Mar 27, 2013. EDOXABAN TOSILATE HYDRATE Edoxaban tosilate hydrate was first approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on April 22, 2011, then approved by the U.S. Food and Drug Administration (FDA) on January 8, 2015, and approved by European Medicine Agency (EMA) on June 19, 2015. 医药研发领域大数据信息平台 药渡是一家专注于医药研发领域的“互联网+”大数据信息平台,我们致力于整合药物研发相关数十个学科专业数据和资源信息,为全球药物研发人员提供“一站式信息解决方案”,助力创新,加速创造。EFINACONAZOLE
Efinaconazole was first approved by Health Canada on October 2, 2013, then approved by the U.S. Food and Drug Administration (FDA) on June 6, 2014, and approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on July 4, 2014.VONOPRAZAN FUMARATE
Vonoprazan Fumarate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on December 26, 2014. It was co-developed and marketed as Takecab ® by Takeda & Otsuka. Vonoprazan has a novel mechanism of action called potassium-competitive acid blockers (P-CABs) which competitively inhibits the binding the potassium ions to H +, K +-ATPase (also known as the ETANERCEPT - PHARMACODIA.COM Etanercept was first approved by the U.S. Food and Drug Administration (FDA) on November 2, 1998, then approved by European Medicine Agency (EMA) on February 3, 2000, Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on January 19, 2005, China Food and Drug Administration (CFDA) on February 26, 2010. IBRUTINIB - PHARMACODIA.COM In Vivo Efficacy. In ex vivo studies, Btk activated site occupancy in splenocyte in ibrutinib pre-treated mice.. Xenograft models in SCID mice: OCL-Ly-10 cell lines: Significantly inhibited tumor growth at 3 mg/kg oral daily at day 10. Mantle cell lymphoma (MCL) model: Decreased the clinical signs, mean white blood cell (WBC) counts, collective lymph node weights, PBMC, lymph nodes and hCD19 POLMACOXIB - PHARMACODIA.COM Polmacoxib was approved by the Korean Ministry of Food and Drug Safety (MFDS) on Feb 05, 2015. It was originally developed by CrystalGenomics, then it signed an agreement for the commercialization of polmacoxib in South Korea with Dong-A in August. TENELIGLIPTIN HYDROBROMIDE HYDRATE Teneligliptin hydrobromide hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. It was co-developed & co-marketed as Tenelia ® by Mitsubishi Tanabe and Daiichi Sankyo.. Teneligliptin hydrobromide hydrate is a dipeptidyl peptidase-4 inhibitor. FASUDIL HYDROCHLORIDE HYDRATE Fasudil hydrochloride Hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 30, 1995. It was developed and marketed as Eril ® by Asahi Kasei in Japan.. Fasudil hydrochloride is a selective RhoA/Rho kinase (ROCK) inhibitor. BOSUTINIB MONOHYDRATE Bosutinib hydrate was approved by the U.S. Food and Drug Administration (FDA) on September 4, 2012, then approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Sep 26, 2014, and approved by European Medicine Agency (EMA) on Mar 27, 2013. EDOXABAN TOSILATE HYDRATE Edoxaban tosilate hydrate was first approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on April 22, 2011, then approved by the U.S. Food and Drug Administration (FDA) on January 8, 2015, and approved by European Medicine Agency (EMA) on June 19, 2015. ETANERCEPT - PHARMACODIA.COM Etanercept was first approved by the U.S. Food and Drug Administration (FDA) on November 2, 1998, then approved by European Medicine Agency (EMA) on February 3, 2000, Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on January 19, 2005, China Food and Drug Administration (CFDA) on February 26, 2010.EMPAGLIFLOZIN
Empagliflozin was first approved by European Medicine Agency (EMA) on May 22, 2014, then approved by the U.S. Food and Drug Administration (FDA) on Aug 1, 2014. IBRUTINIB - PHARMACODIA.COM In Vivo Efficacy. In ex vivo studies, Btk activated site occupancy in splenocyte in ibrutinib pre-treated mice.. Xenograft models in SCID mice: OCL-Ly-10 cell lines: Significantly inhibited tumor growth at 3 mg/kg oral daily at day 10. Mantle cell lymphoma (MCL) model: Decreased the clinical signs, mean white blood cell (WBC) counts, collective lymph node weights, PBMC, lymph nodes and hCD19 IGURATIMOD - PHARMACODIA.COM Iguratimod was first approved by China Food and Drug Administration (CFDA) on August 15, 2011, then approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. REVAPRAZAN HYDROCHLORIDE Revaprazan hydrochloride was approved by Korea Food and Drug Administration (KFDA) on September 1, 2005. It was developed and marketed as Revanex ® by Yuhan Corporation in KR.. Revaprazan is the first acid pump antagonist with a function similar to that of proton pump inhibitors (PPIs). LANDIOLOL HYDROCHLORIDE Landiolol hydrochloride was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jul 5, 2002. It was developed and marketed as Onoact ® by Ono in Japan.. Landiolol is a highly cardioselective, ultra short-acting beta blocker. TENELIGLIPTIN HYDROBROMIDE HYDRATE Teneligliptin hydrobromide hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. It was co-developed & co-marketed as Tenelia ® by Mitsubishi Tanabe and Daiichi Sankyo.. Teneligliptin hydrobromide hydrate is a dipeptidyl peptidase-4 inhibitor. IPRAGLIFLOZIN L-PROLINE Ipragliflozin L-proline was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jan 17, 2014. It was developed and marketed as Suglat ® by Astellas cooperating with Kotobuki Pharmaceutical and Merck Sharp & Dohme.. Ipragliflozin L-proline is a sodium-glucose cotransporter 2 (SGLT2) inhibitor.ROFLUMILAST
Roflumilast was first approved by the European Medicines Agency (EMA) on July 5, 2010, then approved by the U.S. Food and Drug Administration (FDA) on February 28, 2011.VILDAGLIPTIN
Repeated Dose Toxicity Repeated dose toxicity studies were performed in rats (up to 26 weeks) and dogs (up to 52 weeks), mice and monkeys (up to 13 weeks). For mice, NOAEL was 250 mg/kg/day, 43×MRHD, the main toxicological effect was the accumulation of clusters of foamy alveolar macrophages in the lung. For rats, NOAEL was 25 mg/kg/day, 6×MRHD, the main toxicological effect was the 医药研发领域大数据信息平台 药渡是一家专注于医药研发领域的“互联网+”大数据信息平台,我们致力于整合药物研发相关数十个学科专业数据和资源信息,为全球药物研发人员提供“一站式信息解决方案”,助力创新,加速创造。EFINACONAZOLE
Efinaconazole was first approved by Health Canada on October 2, 2013, then approved by the U.S. Food and Drug Administration (FDA) on June 6, 2014, and approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on July 4, 2014.VONOPRAZAN FUMARATE
Vonoprazan Fumarate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on December 26, 2014. It was co-developed and marketed as Takecab ® by Takeda & Otsuka. Vonoprazan has a novel mechanism of action called potassium-competitive acid blockers (P-CABs) which competitively inhibits the binding the potassium ions to H +, K +-ATPase (also known as the ETANERCEPT - PHARMACODIA.COM Etanercept was first approved by the U.S. Food and Drug Administration (FDA) on November 2, 1998, then approved by European Medicine Agency (EMA) on February 3, 2000, Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on January 19, 2005, China Food and Drug Administration (CFDA) on February 26, 2010. IBRUTINIB - PHARMACODIA.COM In Vivo Efficacy. In ex vivo studies, Btk activated site occupancy in splenocyte in ibrutinib pre-treated mice.. Xenograft models in SCID mice: OCL-Ly-10 cell lines: Significantly inhibited tumor growth at 3 mg/kg oral daily at day 10. Mantle cell lymphoma (MCL) model: Decreased the clinical signs, mean white blood cell (WBC) counts, collective lymph node weights, PBMC, lymph nodes and hCD19 POLMACOXIB - PHARMACODIA.COM Polmacoxib was approved by the Korean Ministry of Food and Drug Safety (MFDS) on Feb 05, 2015. It was originally developed by CrystalGenomics, then it signed an agreement for the commercialization of polmacoxib in South Korea with Dong-A in August. TENELIGLIPTIN HYDROBROMIDE HYDRATE Teneligliptin hydrobromide hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. It was co-developed & co-marketed as Tenelia ® by Mitsubishi Tanabe and Daiichi Sankyo.. Teneligliptin hydrobromide hydrate is a dipeptidyl peptidase-4 inhibitor. FASUDIL HYDROCHLORIDE HYDRATE Fasudil hydrochloride Hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 30, 1995. It was developed and marketed as Eril ® by Asahi Kasei in Japan.. Fasudil hydrochloride is a selective RhoA/Rho kinase (ROCK) inhibitor. BOSUTINIB MONOHYDRATE Bosutinib hydrate was approved by the U.S. Food and Drug Administration (FDA) on September 4, 2012, then approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Sep 26, 2014, and approved by European Medicine Agency (EMA) on Mar 27, 2013. EDOXABAN TOSILATE HYDRATE Edoxaban tosilate hydrate was first approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on April 22, 2011, then approved by the U.S. Food and Drug Administration (FDA) on January 8, 2015, and approved by European Medicine Agency (EMA) on June 19, 2015. 医药研发领域大数据信息平台 药渡是一家专注于医药研发领域的“互联网+”大数据信息平台,我们致力于整合药物研发相关数十个学科专业数据和资源信息,为全球药物研发人员提供“一站式信息解决方案”,助力创新,加速创造。EFINACONAZOLE
Efinaconazole was first approved by Health Canada on October 2, 2013, then approved by the U.S. Food and Drug Administration (FDA) on June 6, 2014, and approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on July 4, 2014.VONOPRAZAN FUMARATE
Vonoprazan Fumarate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on December 26, 2014. It was co-developed and marketed as Takecab ® by Takeda & Otsuka. Vonoprazan has a novel mechanism of action called potassium-competitive acid blockers (P-CABs) which competitively inhibits the binding the potassium ions to H +, K +-ATPase (also known as the ETANERCEPT - PHARMACODIA.COM Etanercept was first approved by the U.S. Food and Drug Administration (FDA) on November 2, 1998, then approved by European Medicine Agency (EMA) on February 3, 2000, Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on January 19, 2005, China Food and Drug Administration (CFDA) on February 26, 2010. IBRUTINIB - PHARMACODIA.COM In Vivo Efficacy. In ex vivo studies, Btk activated site occupancy in splenocyte in ibrutinib pre-treated mice.. Xenograft models in SCID mice: OCL-Ly-10 cell lines: Significantly inhibited tumor growth at 3 mg/kg oral daily at day 10. Mantle cell lymphoma (MCL) model: Decreased the clinical signs, mean white blood cell (WBC) counts, collective lymph node weights, PBMC, lymph nodes and hCD19 POLMACOXIB - PHARMACODIA.COM Polmacoxib was approved by the Korean Ministry of Food and Drug Safety (MFDS) on Feb 05, 2015. It was originally developed by CrystalGenomics, then it signed an agreement for the commercialization of polmacoxib in South Korea with Dong-A in August. TENELIGLIPTIN HYDROBROMIDE HYDRATE Teneligliptin hydrobromide hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. It was co-developed & co-marketed as Tenelia ® by Mitsubishi Tanabe and Daiichi Sankyo.. Teneligliptin hydrobromide hydrate is a dipeptidyl peptidase-4 inhibitor. FASUDIL HYDROCHLORIDE HYDRATE Fasudil hydrochloride Hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 30, 1995. It was developed and marketed as Eril ® by Asahi Kasei in Japan.. Fasudil hydrochloride is a selective RhoA/Rho kinase (ROCK) inhibitor. BOSUTINIB MONOHYDRATE Bosutinib hydrate was approved by the U.S. Food and Drug Administration (FDA) on September 4, 2012, then approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Sep 26, 2014, and approved by European Medicine Agency (EMA) on Mar 27, 2013. EDOXABAN TOSILATE HYDRATE Edoxaban tosilate hydrate was first approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on April 22, 2011, then approved by the U.S. Food and Drug Administration (FDA) on January 8, 2015, and approved by European Medicine Agency (EMA) on June 19, 2015. ETANERCEPT - PHARMACODIA.COM Etanercept was first approved by the U.S. Food and Drug Administration (FDA) on November 2, 1998, then approved by European Medicine Agency (EMA) on February 3, 2000, Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on January 19, 2005, China Food and Drug Administration (CFDA) on February 26, 2010.EMPAGLIFLOZIN
Empagliflozin was first approved by European Medicine Agency (EMA) on May 22, 2014, then approved by the U.S. Food and Drug Administration (FDA) on Aug 1, 2014. IBRUTINIB - PHARMACODIA.COM In Vivo Efficacy. In ex vivo studies, Btk activated site occupancy in splenocyte in ibrutinib pre-treated mice.. Xenograft models in SCID mice: OCL-Ly-10 cell lines: Significantly inhibited tumor growth at 3 mg/kg oral daily at day 10. Mantle cell lymphoma (MCL) model: Decreased the clinical signs, mean white blood cell (WBC) counts, collective lymph node weights, PBMC, lymph nodes and hCD19 IGURATIMOD - PHARMACODIA.COM Iguratimod was first approved by China Food and Drug Administration (CFDA) on August 15, 2011, then approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. REVAPRAZAN HYDROCHLORIDE Revaprazan hydrochloride was approved by Korea Food and Drug Administration (KFDA) on September 1, 2005. It was developed and marketed as Revanex ® by Yuhan Corporation in KR.. Revaprazan is the first acid pump antagonist with a function similar to that of proton pump inhibitors (PPIs). LANDIOLOL HYDROCHLORIDE Landiolol hydrochloride was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jul 5, 2002. It was developed and marketed as Onoact ® by Ono in Japan.. Landiolol is a highly cardioselective, ultra short-acting beta blocker. TENELIGLIPTIN HYDROBROMIDE HYDRATE Teneligliptin hydrobromide hydrate was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. It was co-developed & co-marketed as Tenelia ® by Mitsubishi Tanabe and Daiichi Sankyo.. Teneligliptin hydrobromide hydrate is a dipeptidyl peptidase-4 inhibitor. IPRAGLIFLOZIN L-PROLINE Ipragliflozin L-proline was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jan 17, 2014. It was developed and marketed as Suglat ® by Astellas cooperating with Kotobuki Pharmaceutical and Merck Sharp & Dohme.. Ipragliflozin L-proline is a sodium-glucose cotransporter 2 (SGLT2) inhibitor.ROFLUMILAST
Roflumilast was first approved by the European Medicines Agency (EMA) on July 5, 2010, then approved by the U.S. Food and Drug Administration (FDA) on February 28, 2011.VILDAGLIPTIN
Repeated Dose Toxicity Repeated dose toxicity studies were performed in rats (up to 26 weeks) and dogs (up to 52 weeks), mice and monkeys (up to 13 weeks). For mice, NOAEL was 250 mg/kg/day, 43×MRHD, the main toxicological effect was the accumulation of clusters of foamy alveolar macrophages in the lung. For rats, NOAEL was 25 mg/kg/day, 6×MRHD, the main toxicological effect was the 药渡-医药研发领域大数据信息平台首页 药渡-医药研发领域大数据信息平台首页温馨提示
您的账户已在别处登录,如果不是本人操作,请前往个人中心修改您账号登录密码!!! 咨询定制和售前售后服务 请您联系客服 400-851-9921,为您提供服务进行购买 您暂无使用该功能的权限 请您开通付费会员权限 开通付费会员权限 您暂无使用该功能的权限您可以:
1、去开通付费会员权限(全部权限) 2、申请试用,您就能获得免费试用(全部权限)机会 申请试用 开通付费会员权限 您暂无使用该功能的权限 请您登录(没有账号点我)去登录
欢迎您注册药渡数据账号_邮箱注册_
请使用您的企业邮箱进行注册,药渡客服将尽快给您发送激活链接企业名称
企业邮箱
设置密码
验证码_刷新_
阅读并同意《药渡数据用户须知》《隐私声明》
立即注册
手机号
*
China (中国)+86
*
*
Afghanistan (افغانستان)+93*
Albania (Shqipëri)+355*
Algeria (الجزائر)+213*
American Samoa+1684
*
Andorra+376
*
Angola+244
*
Anguilla+1264
*
Antigua and Barbuda+1268*
Argentina+54
*
Armenia (Հայաստան)+374*
Aruba+297
*
Australia+61
*
Austria (Österreich)+43*
Azerbaijan (Azərbaycan)+994*
Bahamas+1242
*
Bahrain (البحرين)+973*
Bangladesh (বাংলাদেশ)+880*
Barbados+1246
*
Belarus (Беларусь)+375*
Belgium (België)+32*
Belize+501
*
Benin (Bénin)+229
*
Bermuda+1441
*
Bhutan (འབྲུག)+975*
Bolivia+591
*
Bosnia and Herzegovina (Босна и Херцеговина)+387*
Botswana+267
*
Brazil (Brasil)+55
*
Brunei+673
*
Bulgaria (България)+359*
Burkina Faso+226
*
Burundi (Uburundi)+257*
Cambodia (កម្ពុជា)+855*
Cameroon (Cameroun)+237*
Canada+1
*
Cape Verde (Kabu Verdi)+238*
Cayman Islands+1345
*
Central African Republic (République centrafricaine)+236*
Chad (Tchad)+235
*
Chile+56
*
China (中国)+86
*
Colombia+57
*
Comoros (جزر القمر)+269*
Congo (DRC) (Jamhuri ya Kidemokrasia ya Kongo)+243*
Congo (Republic) (Congo-Brazzaville)+242*
Cook Islands+682
*
Costa Rica+506
*
Côte d’Ivoire+225*
Croatia (Hrvatska)+385*
Cuba+53
*
Curaçao+599
*
Cyprus (Κύπρος)+357*
Czech Republic (Česká republika)+420*
Denmark (Danmark)+45*
Djibouti+253
*
Dominica+1767
*
Dominican Republic (República Dominicana)+1809*
Ecuador+593
*
Egypt (مصر)+20*
El Salvador+503
*
Equatorial Guinea (Guinea Ecuatorial)+240*
Estonia (Eesti)+372
*
Ethiopia+251
*
Faroe Islands (Føroyar)+298*
Fiji+679
*
Finland (Suomi)+358
*
France+33
*
French Guiana (Guyane française)+594*
French Polynesia (Polynésie française)+689*
Gabon+241
*
Gambia+220
*
Georgia (საქართველო)+995*
Germany (Deutschland)+49*
Ghana (Gaana)+233
*
Gibraltar+350
*
Greece (Ελλάδα)+30*
Greenland (Kalaallit Nunaat)+299*
Grenada+1473
*
Guadeloupe+590
*
Guam+1671
*
Guatemala+502
*
Guinea (Guinée)+224*
Guinea-Bissau (Guiné Bissau)+245*
Guyana+592
*
Haiti+509
*
Honduras+504
*
Hong Kong (中国香港)+852*
Hungary (Magyarország)+36*
Iceland (Ísland)+354*
India (भारत)+91*
Indonesia+62
*
Iran (ایران)+98*
Iraq (العراق)+964*
Ireland+353
*
Israel (ישראל)+972*
Italy (Italia)+39
*
Jamaica+1876
*
Japan (日本)+81
*
Jordan (الأردن)+962*
Kazakhstan (Казахстан)+7*
Kenya+254
*
Kiribati+686
*
Kuwait (الكويت)+965*
Kyrgyzstan (Кыргызстан)+996*
Laos (ລາວ)+856*
Latvia (Latvija)+371*
Lebanon (لبنان)+961*
Lesotho+266
*
Liberia+231
*
Libya (ليبيا)+218*
Liechtenstein+423
*
Lithuania (Lietuva)+370*
Luxembourg+352
*
Macau (中国澳門)+853*
Madagascar (Madagasikara)+261*
Malawi+265
*
Malaysia+60
*
Maldives+960
*
Mali+223
*
Malta+356
*
Martinique+596
*
Mauritania (موريتانيا)+222*
Mauritius (Moris)+230*
Mayotte+269
*
Mexico (México)+52
*
Moldova (Republica Moldova)+373*
Monaco+377
*
Mongolia (Монгол)+976*
Montenegro (Crna Gora)+382*
Montserrat+1664
*
Morocco (المغرب)+212*
Mozambique (Moçambique)+258*
Myanmar (Burma) (မြန်မာ)+95*
Namibia (Namibië)+264*
Nepal (नेपाल)+977*
Netherlands (Nederland)+31*
New Caledonia (Nouvelle-Calédonie)+687*
New Zealand+64
*
Nicaragua+505
*
Niger (Nijar)+227
*
Nigeria+234
*
Norway (Norge)+47
*
Oman (عُمان)+968*
Pakistan (پاکستان)+92*
Palau+680
*
Palestine (فلسطين)+930*
Panama (Panamá)+507*
Papua New Guinea+675*
Paraguay+595
*
Peru (Perú)+51
*
Philippines+63
*
Poland (Polska)+48
*
Portugal+351
*
Puerto Rico+1787
*
Qatar (قطر)+974*
Réunion (La Réunion)+262*
Romania (România)+40*
Russia (Россия)+7*
Rwanda+250
*
Saint Kitts and Nevis+1869*
Saint Lucia+1758
*
Saint Martin (Saint-Martin (partie française))+590*
Saint Pierre and Miquelon (Saint-Pierre-et-Miquelon)+508*
Saint Vincent and the Grenadines+1784*
Samoa+685
*
San Marino+378
*
São Tomé and Príncipe (São Tomé e Príncipe)+239*
Saudi Arabia (المملكة العربية السعودية)+966*
Senegal (Sénégal)+221*
Serbia (Србија)+381*
Seychelles+248
*
Sierra Leone+232
*
Singapore+65
*
Sint Maarten+590
*
Slovakia (Slovensko)+421*
Slovenia (Slovenija)+386*
Solomon Islands+677
*
Somalia (Soomaaliya)+252*
South Africa+27
*
South Korea (대한민국)+82*
South Sudan (جنوب السودان)+211*
Spain (España)+34
*
Sri Lanka (ශ්රී ලංකාව)+94*
Sudan (السودان)+249*
Suriname+597
*
Swaziland+268
*
Sweden (Sverige)+46
*
Switzerland (Schweiz)+41*
Syria (سوريا)+963*
Taiwan (中国台灣)+886*
Tajikistan+992
*
Tanzania+255
*
Thailand (ไทย)+66*
Timor-Leste+670
*
Togo+228
*
Tonga+676
*
Trinidad and Tobago+1868*
Tunisia (تونس)+216*
Turkey (Türkiye)+90*
Turkmenistan+993
*
Turks and Caicos Islands+1649*
U.S. Virgin Islands+1340*
Uganda+256
*
Ukraine (Україна)+380*
United Arab Emirates (الإمارات العربية المتحدة)+971*
United Kingdom+44
*
United States+1
*
Uruguay+598
*
Uzbekistan (Oʻzbekiston)+998*
Vanuatu+678
*
Venezuela+58
*
Vietnam (Việt Nam)+84*
Yemen (اليمن)+967*
Zambia+260
*
Zimbabwe+263
企业名称
设置密码
验证码_刷新_
短信验证码
阅读并同意《药渡数据用户须知》《隐私声明》
立即注册
欢迎您使用药渡数据关闭购买
您的账号已在别的电脑登录,您在本机已经退出。 如确认不是您本人进行操作,请您重新登录及时修改密码或致电药渡数据客服400-851-9921.重新登录 关闭
欢迎登录
保持登录忘记密码登录
没有账号?
免费申请账号
为保障您的权益,请您获取短信验证码进行身份验证 您将获取短信验证码的手机号是:刷新
提交
如长时间无法接收短信验证码,请致电 药渡数据客服400-851-9921
公共头部 药渡-医药研发领域大数据信息平台首页 * 欢迎访问 药渡* 试用/购买
* APP
* 微信公众号
* English _|_
* 登录 登录 注册* 全部
* 药物名称
* 靶点
* 公司
* 适应症
结构式检索 组合检索 * 搜索历史 清空 无搜索历史,要加油哦~ 温馨提示:请您选择适宜的搜索范围,提高搜索准确度Pharmalaxy
数据库教程
咨询服务
药渡大学
药渡说
更多
香豆素在药物中的应用09:07
_药渡数据_
_研究进展及盘点_|_药渡_
珍藏版!中国TOP10生物类似药2020-01-06
_研究进展及盘点__热点解析_
|_药渡_
肿瘤免疫时代下:细胞因子药物研发现状和趋势2020-01-02
_药渡咨询_
|_药渡_
药渡盘点—2019年美国FDA批准新药报告(下)2020-01-02
_研究进展及盘点__热点解析_
|_药渡_
药渡盘点—2019年美国FDA批准新药报告(上)2020-01-02
_研究进展及盘点__热点解析_
|_药渡_
会议
更多
第104期论坛丨创新药研发项目的综合评估苏州
_新药研发_
2020-02-22
来啦:人气爆棚-CMC药物质量研究及技术服务年会2020(南京)南京
_方法与技术_
2020-02-20
金斯瑞生物科技全球产业论坛议程公布, 一月邀您相聚“JPM周”旧金山
_生物医药_
2020-01-14
2020年6月 |
第二届中国生物医药创新合作大会再度来袭!杭州
_生物医药_
2020-06-19
还等什么?DIA 2020年会早期注册优惠不容错过!苏州
__
2020-05-20
药渡数据使用教程 更多__
查询某靶点的在研公司__
查询某药物在FDA溶出中的信息__
查询药物的杂质信息__
查询在某年首批的药物__
检索含有某子结构的药物靶点情况__
检索含有某子结构的全球药物研发动态__
查询同靶点药物__
查询药物的工艺路线__
查询某个药物近5年的销量__
如何进行药物数据的对比分析__
查询某靶点的在研公司__
查询某药物在FDA溶出中的信息__
查询药物的杂质信息__
查询在某年首批的药物__
检索含有某子结构的药物靶点情况__
检索含有某子结构的全球药物研发动态 您的浏览器版本过低,请更换至IE9以上或者谷歌/火狐浏览器再次打开* 药物
* 合成路线
* 精确查找
* 子结构检索
* 相似度检索
* 90% 80% 70% 60% 50% 以上* 合成工艺
组合式检索
组合检索
* 通用检索
* 专利专用
(您可任意输入选择一项或多项关键词进行检索)药物名称
and
靶点
and
适应症
and
公司
and
研发阶段
请选择 批准上市 NDA 申请 BLA 申请 临床三期 临床二期 临床一期 临床申请 撤市 and临床状态
请选择 进行中 无进展 终止 and 化学药 and 生物药 and 中国1类申请号
and
专利标题
and
药物名称
and
公开号
and
技术分类
请选择 化合物 衍生物 制备/工艺 制剂 用途 联合用药/组合物 中间体 分析方法 器械 其它 and公开日
- and
中国同族最早失效日-
确定 取消 首页底部* 关于药渡
* 购买与服务
* 诚聘英才
* 网址地图
* 帮助中心
* 联系我们
客服电话:400-851-9921 客服邮箱:data@pharmacodia.com 公司地址:北京市海淀区上地五街昊海大厦 CopyRight©2019药渡经纬信息科技(北京)有限公司 电子与信息服务业务经营许可证151001号京ICP备14047345
| 京公安网备11010802017043号 | (京)-经营性-2015-0023(互联网药品信息服务资格证书)*
药渡APP
* 微信二维码
* 友情链接:
* 生物谷
* |
* 生物探索
* |
* 新浪医药
* |
* 火石创�
* |
* 投壶网
* |
* 贝壳社
* |
* 肽度时界
* |
* 易科学
* |
* 医药魔方
* |
* 序说DNASpeaking
* |
* 实验易购
* |
* 生物360
* |
* 化学加网
* |
* 人人实验
* |
* 超凡知识产权我要反馈
比
客服电话:400-851-9921有奖反馈
感谢您使用药渡数据库企业版,为不断提升我们的服务质量,现诚邀您参与有奖反馈活动。 您只需填写提交您对药渡数据的宝贵意见和您的联系方式,即可参与药渡数据的抽奖活动。 我们将定期抽出参与反馈的幸运用户,送上精美礼品一份。_*_姓名
_*_手机号
_*_工作单位
我的反馈
提交
正在提交...
您的留言已成功提交,感谢您的宝贵意见!对 比 工 具 _>_
药物信息对比
请点击“+对比”添加对比的药物 开始对比 最多6个 清空提示
确定 取消
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0