Are you over 18 and want to see adult content?
More Annotations
A complete backup of amaderbharat.com
Are you over 18 and want to see adult content?

A complete backup of estudiaraprender.com
Are you over 18 and want to see adult content?

A complete backup of lyfe-support.blogspot.com
Are you over 18 and want to see adult content?

A complete backup of currencyrate.today
Are you over 18 and want to see adult content?
A complete backup of saintsreport.com
Are you over 18 and want to see adult content?
A complete backup of esllimpeza.com.br
Are you over 18 and want to see adult content?
A complete backup of immigrationboards.com
Are you over 18 and want to see adult content?

A complete backup of makelaarsland.nl
Are you over 18 and want to see adult content?
Favourite Annotations
A complete backup of https://www.onestophindi.com/ofilmywap-movies/
Are you over 18 and want to see adult content?
A complete backup of https://www.kaufmich.com/Milchmami
Are you over 18 and want to see adult content?
A complete backup of http://dependent.ru.net/v/191774
Are you over 18 and want to see adult content?
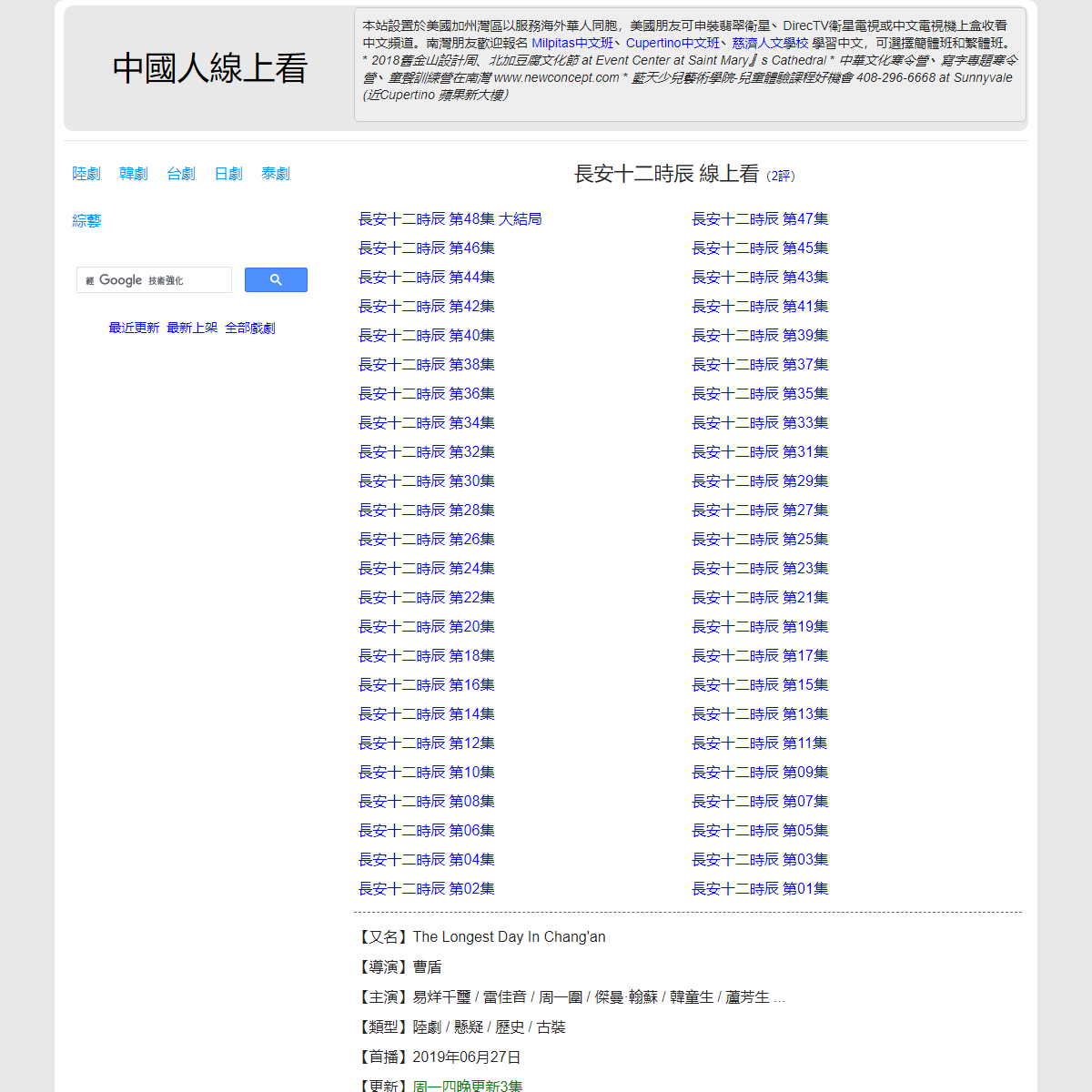
A complete backup of https://chinaq.tv/cn190627b/
Are you over 18 and want to see adult content?

A complete backup of http://www.linkzb.net/country/usa/
Are you over 18 and want to see adult content?
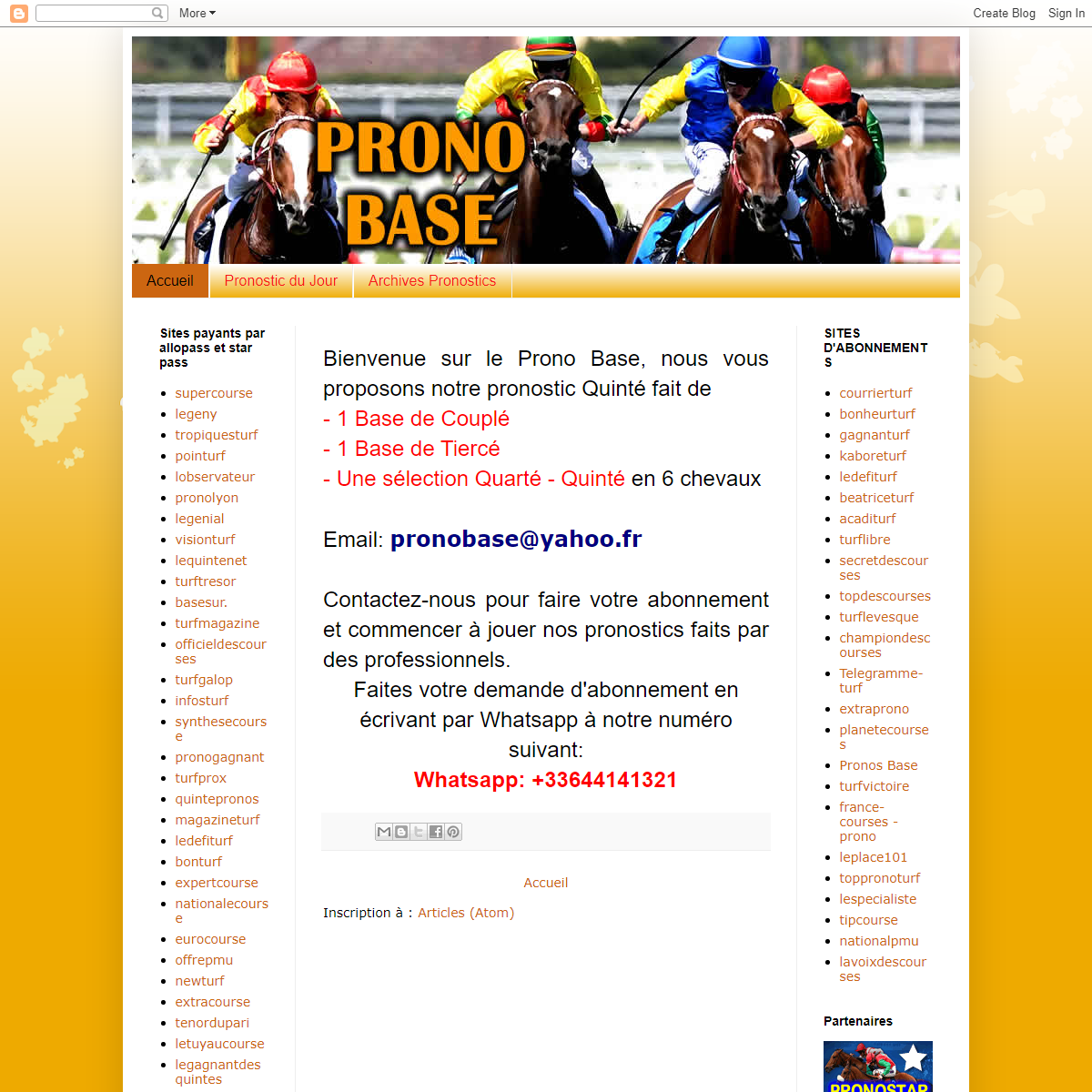
A complete backup of https://pronosbase.blogspot.com/
Are you over 18 and want to see adult content?
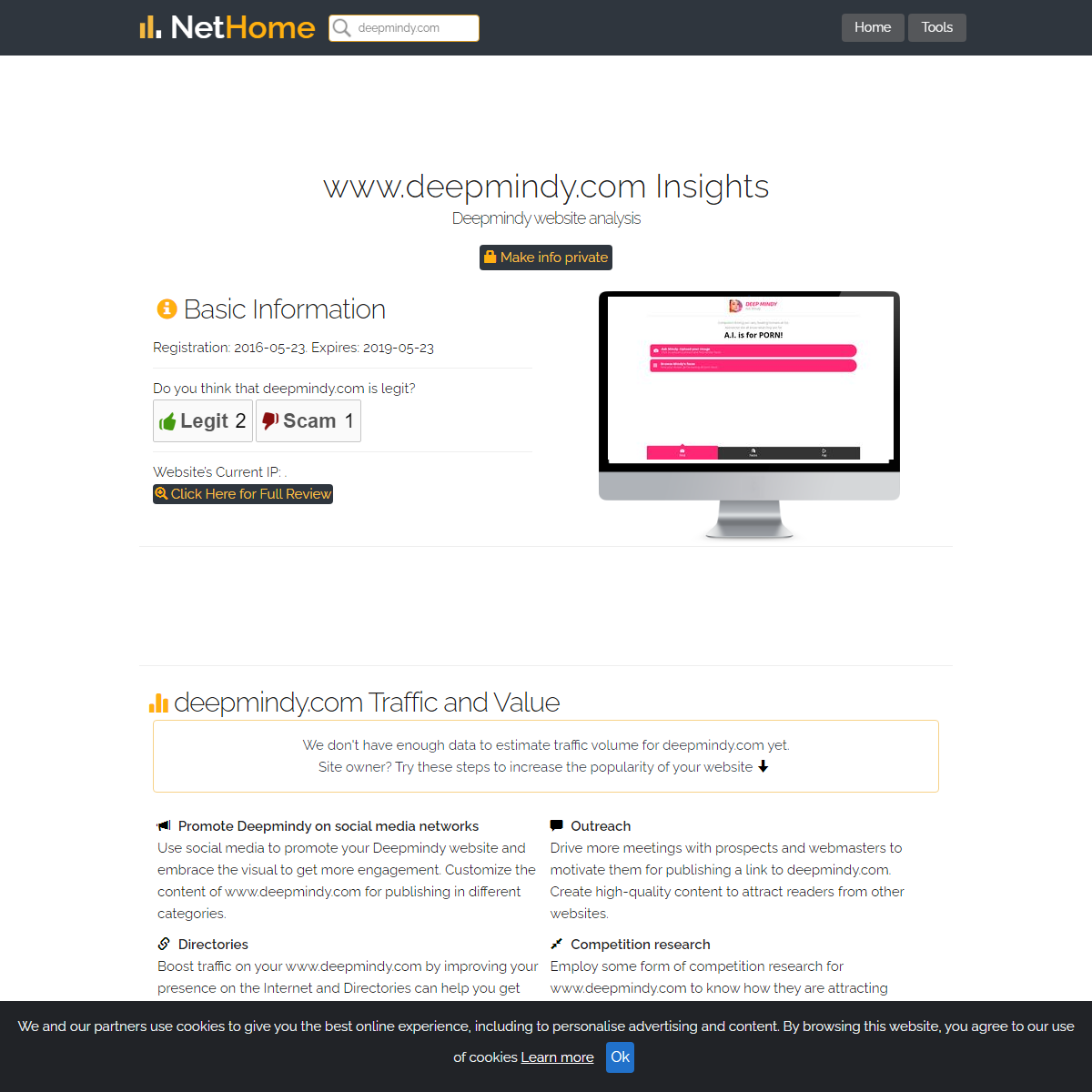
A complete backup of https://nethome.wiki/deepmindy.com
Are you over 18 and want to see adult content?
Text
throughout clinical
CAREER OPPORTUNITIES AT PAREXEL Our employees have the opportunity to make a difference globally while we support them locally in environments full of challenge, collaboration, flexibility and industry-leading growth potential. We invite you to learn more about the career opportunities available with Parexel across the globe. We invite you to join us in making adifference.
SEARCH OUR JOB OPPORTUNITIES AT PAREXEL Search for available job openings at Parexel. 2020. Joined the Scientific Data Organization as Scientific Data Technology Director, creating agile access to real world data, bringing best in class technology solutions to Parexel services, and harnessing data to transform therapy development for patients.PAREXEL - SIS
The Safety Information System is a secure password protected tool that distributes safety information to Investigators, replacing the previous paper based distribution process. This tool provides easy access to all relevant safety information online and in one place. Safety reports are made immediately available to investigators as soon as they are uploaded into the system.SITE ORDER MANAGER
Site Order Manager (SOM) Login. Username DEMYSTIFYING FDA’S KASA INITIATIVE… AND HOW IT AIMS TO 2. Risk Mitigation Assessment. Using the KASA-generated, risk-ranked, appropriate IQA template, the agency will perform the quality assessment of the drug product and its proposed manufacturing process to ensure that all inherent risks are mitigated to the extentpossible.
MEDICAL WRITING
2020. Promoted to Senior Medical Writer, focusing on the development and support of clinical documents intended for regulatory submission. 2017. Began a new role working remotely as a Medical Writer II, researching and coordinating the development of clinical documents intended for submission to the FDA or other regulatory authorities. STEVE WINITSKY, M.D. Education. Dr. Winitsky earned a B.A. in Chemistry from Columbia University in New York in 1992. He then went on to earn his M.D. from the University of Miami School of Medicine in Florida in 1996. He was an internal medicine resident at Emory University and completed his residency in the NHLBI/NIH Clinical Associate Program in 1999,continuing
IMPACT - LOG INIMPACT HARMONY CTMSIMPACT CTMSPAREXEL CTMSCTMS SYSTEMSIMPACT SITE MINECRAFTIMPACT CLINICAL TRIAL MANAGEMENT SYSTEM Unsupported browser found: Sorry, this version of your browser is unable to view this web page. A supported browser can be downloaded from: Microsoft Internet Explorer EUROPEAN COORDINATION HUB AND DISTRIBUTION CENTER PAREXEL’S EUROPEAN COORDINATION HUB AND DISTRIBUTION CENTER OFFERS: • 25,500 m facility offering more than 65,000 cubic meters of clinical supply storage space CLINICAL RESEARCH ORGANIZATION (CRO) & BIOPHARMACEUTICALCAREERSSOLUTIONSINNOVATIONS & THERAPEUTIC EXPERTISEVOLUNTEERS, STUDIES & INVESTIGATORS Risk-based Quality Management (RBQM) Services. The new world order of clinical research demands a fresh look at end-to-end clinical practices. Parexel’s approach to risk-based quality management (RBQM) is a holistic risk strategy rooted in Quality by Design principles, that relies on critical data to inform decision-makingthroughout clinical
CAREER OPPORTUNITIES AT PAREXEL Our employees have the opportunity to make a difference globally while we support them locally in environments full of challenge, collaboration, flexibility and industry-leading growth potential. We invite you to learn more about the career opportunities available with Parexel across the globe. We invite you to join us in making adifference.
SEARCH OUR JOB OPPORTUNITIES AT PAREXEL Search for available job openings at Parexel. 2020. Joined the Scientific Data Organization as Scientific Data Technology Director, creating agile access to real world data, bringing best in class technology solutions to Parexel services, and harnessing data to transform therapy development for patients.PAREXEL - SIS
The Safety Information System is a secure password protected tool that distributes safety information to Investigators, replacing the previous paper based distribution process. This tool provides easy access to all relevant safety information online and in one place. Safety reports are made immediately available to investigators as soon as they are uploaded into the system.SITE ORDER MANAGER
Site Order Manager (SOM) Login. Username DEMYSTIFYING FDA’S KASA INITIATIVE… AND HOW IT AIMS TO 2. Risk Mitigation Assessment. Using the KASA-generated, risk-ranked, appropriate IQA template, the agency will perform the quality assessment of the drug product and its proposed manufacturing process to ensure that all inherent risks are mitigated to the extentpossible.
MEDICAL WRITING
2020. Promoted to Senior Medical Writer, focusing on the development and support of clinical documents intended for regulatory submission. 2017. Began a new role working remotely as a Medical Writer II, researching and coordinating the development of clinical documents intended for submission to the FDA or other regulatory authorities. STEVE WINITSKY, M.D. Education. Dr. Winitsky earned a B.A. in Chemistry from Columbia University in New York in 1992. He then went on to earn his M.D. from the University of Miami School of Medicine in Florida in 1996. He was an internal medicine resident at Emory University and completed his residency in the NHLBI/NIH Clinical Associate Program in 1999,continuing
IMPACT - LOG INIMPACT HARMONY CTMSIMPACT CTMSPAREXEL CTMSCTMS SYSTEMSIMPACT SITE MINECRAFTIMPACT CLINICAL TRIAL MANAGEMENT SYSTEM Unsupported browser found: Sorry, this version of your browser is unable to view this web page. A supported browser can be downloaded from: Microsoft Internet Explorer EUROPEAN COORDINATION HUB AND DISTRIBUTION CENTER PAREXEL’S EUROPEAN COORDINATION HUB AND DISTRIBUTION CENTER OFFERS: • 25,500 m facility offering more than 65,000 cubic meters of clinical supply storage space ABOUT PAREXEL: A CLINICAL RESEARCH ORGANIZATION (CRO About Us. Headquartered near Boston, Massachusetts and in Durham, North Carolina, Parexel has over 16,000 employees, with offices that support clients in over 100 countries around the world. We provide the most comprehensive drug development capabilities of any PHASE 1 CLINICAL TRIALS The first stages of a clinical trial can be tense. So, choosing a partner to share this critical phase of your drug’s journey is key. By working with us, you’ll gain a simplified and dedicated approach to initial stage testing. Phase I is the first time a drug will be tested in people, and often involves a small group of volunteers withno
WORKING AT PAREXEL
At Parexel, we work every day to improve the world’s health. Find a career with heart – from clinical trials to regulatory, consulting,and market access.
LOGIN
If you are a registered physician and have connection issue or you forgot your password, please use the support functionality or contact Helpdesk. If you are a member of Pharmaceutical industry Program Team registered in PAREXEL My Access Programs please enter your Log In details on the left side or contact the Program Project Manager.SEARCH FOR A STUDY
Refer a friend to screen for study 246909 or 261043 and you could earn a referral bonus. TBD - TBD. Up to: $100. Healthy Volunteer. Smoking (No) 251720. 18-50. The Baltimore PAREXEL team is currently seeking healthy males and females (non-childbearing potenSITE ORDER MANAGER
Site Order Manager (SOM) Login. Username NEW REFERRAL OPPORTUNITIES Additional Information. Refer a friend to screen for the studies listed below ,#246909 ($100 bonus), 261043 ($100 bonus) • Your referral must be new to PAREXEL (or hasn’t screened for a study in 2 years). • Referral must give the full name of the person who referred him or her when scheduling an appointment. • Referral mustpass
INITIATION CLINICAL RESEARCH ASSOCIATE II AT PAREXEL 2020. Promoted to Senior Medical Writer, focusing on the development and support of clinical documents intended for regulatory submission. 2017. Began a new role working remotely as a Medical Writer II, researching and coordinating the development of clinical documents intended for submission to the FDA or other regulatory authorities. WATCH REPLAY: INNOVATE IN PROJECT LEADERSHIP 2020. Promoted to Senior Medical Writer, focusing on the development and support of clinical documents intended for regulatory submission. 2017. Began a new role working remotely as a Medical Writer II, researching and coordinating the development of clinical documents intended for submission to the FDA or other regulatory authorities. MANAGER, DATA MANAGEMENT (REMOTE) AT PAREXEL 2020. Promoted to Senior Medical Writer, focusing on the development and support of clinical documents intended for regulatory submission. 2017. Began a new role working remotely as a Medical Writer II, researching and coordinating the development of clinical documents intended for submission to the FDA or other regulatory authorities. This website uses cookies. You may choose to continue browsing with cookies, or leave thesite.Show Cookies
Continue
Browsing
Essential Cookies - The site owner has indicated that these are essential to the running of the site.*
Cookie Guard
- This cookie is essential for storing the status of your cookie choices whilst using this site.*
Concrete5
- This session id cookie remembers your recent page requests on our site and is required for the website to run. This cookie expires when you close your browser.*
__sharethis_cookie_test__ - This session id cookie remembers your recent page requests on our site and is required for the website to run. This cookie expires when you close your browser. Non-Essential Cookies - The site owner has approved these cookies but you may turn them off.*
- These cookies are used to collect information about how visitors use our site. We use the information to compile reports and to help us improve the site. The cookies collect information in an anonymous form, including the number of visitors to the site, where visitors have come to the site from and the pages they visited. See list of all Non-Essential Cookies on the Privacy Policy.__
__
* Solutions
* Innovations & Therapeutic Expertise * Volunteers, Studies & Investigators * Ecosystem, Education & Careers * News, Events & Resources* About Us
__ Contact Us
__ Preference Center__ Client Login
__ Global Locations
* __
* __
* __
* __
275 Grove St., Suite 101C, Newton, MA 02466+1 617 454 9300
2520 Meridian Parkway, Research Triangle Park, Suite 200, Durham, NC27713
+1 919 544 3170
� 2000-2020. Parexel International Corporation. RECOMMENDED FOR YOU: HEAR FROM PARTICIPANTS SHARE THEIR REASONS FOR VOLUNTEERING FORCLINICAL TRIALS
Watch Video
SEE HOW PAREXEL IS DOING ITS PART #WITH HEARTWatch Video
RECOMMENDED FOR YOU: SEE HOW PAREXEL IS DOING ITS PART #WITH HEARTWatch Video
FIND OUT WHY THE BEST-DESIGNED STUDIES START WITH THE PATIENTWatch Video
DISCOVER THE BENEFITS OF PAREXEL'S DEDICATED BIOTECH DIVISIONWatch Video
RECOMMENDED FOR YOU: SEE HOW PAREXEL IS DOING ITS PART #WITH HEARTWatch Video
LEARN HOW THE RIGHT PEOPLE, PROCESSES AND CULTURE CAN UNLOCKINNOVATION
Corporate VP, Alberto GrignoloWatch Video
RECOMMENDED FOR YOU: INNOVATING PRECISION MEDICINE TRIALSWatch Video
SEE HOW PAREXEL IS DOING ITS PART #WITH HEARTWatch Video
PAREXEL'S SY PRETORIUS ON OUR PATIENT INNOVATION CENTERWatch Video
RECOMMENDED FOR YOU: PAREXEL CAREERS: SEE HOW WE GROW TOGETHERWatch Video
SEE HOW PAREXEL IS DOING ITS PART #WITH HEARTWatch Video
RECOMMENDED FOR YOU: SEE HOW PAREXEL IS DOING ITS PART #WITH HEARTWatch Video
PAREXEL'S SY PRETORIUS ON OUR PATIENT INNOVATION CENTERWatch Video
REGARDING CORONAVIRUS (COVID-19) During this time, we continue to be committed to our clients’ business while putting the safety of patients, client partners and our employees at the heart of everything we do. Message from our CEO Expert Insights: Webinars & Blogs Parexel COVID-19 Resource CenterSolutions
Innovations & Therapeutic Expertise Volunteers, Studies & Investigators FIGHTING CANCER TOGETHER. Every year there are new scientific discoveries and treatments developed that help improve patient outcomes and even cure some cancers. But at the same time, there is still more to do. At Parexel we’re working with more urgency than ever before to reduce the burden of cancer and support the goal of a cancer-free world. Read Monica’s Story DISRUPTING THE BENCH TO BEDSIDE PHILOSOPHY Precision medicine brings insights from the bench to bedside, but we need to share information from the bedside back to the bench to innovate drug development. Learn more about expanding this philosophy from industry leadersWatch the video
THE FUTURE OF CLINICAL TRIALS IS NOW Now more than ever, the industry is shifting to Decentralized Clinical Trials – either fully virtual or hybrid approaches – to improve patient access and enrollment in research studies during the COVID-19 pandemic. Watch our new video clip spotlighting a patient’s perspective on the positive impact Decentralized Clinical Trials canhave on daily life.
Learn More
FEATURED CONTENT
3 Ways to Reap Benefits of Adaptive TrialsView Now __
A harmonized approach to data integrityView Now __
Decentralized clinical trials: Are we ready to make the leap?Read Now __
How to Take the Lead in Reinventing the Life Sciences IndustryRead Now __
Launch Into Market With Real-World EvidenceView Now __
__ SEE FULL CONTENT STREAM 3 Ways to Reap Benefits of Adaptive TrialsView Now __
A harmonized approach to data integrityView Now __
Decentralized clinical trials: Are we ready to make the leap?Read Now __
How to Take the Lead in Reinventing the Life Sciences IndustryRead Now __
Launch Into Market With Real-World EvidenceView Now __
CLOSE
* CORPORATE LINKS
* About Us
* News
* Events
* Careers
* Education
* Investigators
* My Preferences
* SOLUTIONS
* Parexel Biotech
* Clinical Development * Integrated Technologies* Consulting
* Medical & Scientific Services * Real-World Evidence * STUDIES & VOLUNTEERS* About Trials
* Participate in a Trial*
* GLOBAL STUDY LOCATIONS* Baltimore, US
* Los Angeles, US
* London, UK
* Berlin, DE
* __
* __
* __
* __
275 Grove St., Suite 101C, Newton, MA 02466+1 617 454 9300
2520 Meridian Parkway, Research Triangle Park, Suite 200, Durham, NC27713
+1 919 544 3170
� 2000-2020. Parexel International Corporation. All Rights Reserved.* Privacy Policy
* Terms of Service
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0