Are you over 18 and want to see adult content?
More Annotations

A complete backup of sport24.ru/news/football/2020-02-24-liverpul-zabil-v-36-y-igre-apl-podryad-boleye-dlitelnaya-golevaya-seriy
Are you over 18 and want to see adult content?

A complete backup of ec.ltn.com.tw/article/breakingnews/3078875
Are you over 18 and want to see adult content?
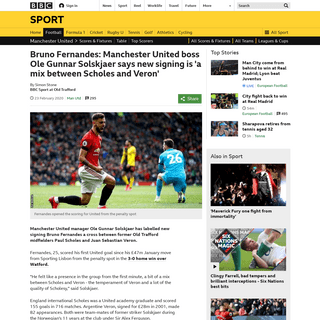
A complete backup of www.bbc.co.uk/sport/football/51607355
Are you over 18 and want to see adult content?

A complete backup of www.superhaber.tv/eskiya-dunyaya-hukumdar-olmaz-158-bolum-izle-eskiya-dunyaya-hukumdar-olmaz-son-bolum-izle
Are you over 18 and want to see adult content?
Favourite Annotations

Behind The Scene | مجتمع بيهايند | اقوى قاعدة بيانات للأفلام والمراجعات
Are you over 18 and want to see adult content?

ADMIRAL Sportwetten: Online-Sportwetten mit Top-Wettquoten
Are you over 18 and want to see adult content?

Mitsubishi Motors Malaysia | Drive Your Ambition
Are you over 18 and want to see adult content?
Bedrijfswagen en bestelwagen specialist | Derks Bedrijfswagens Uden
Are you over 18 and want to see adult content?
ラジコンカーを始めたい初心者の入門ブログ「Get ready RC CAR」
Are you over 18 and want to see adult content?
Historia Interactiva
Are you over 18 and want to see adult content?
Text
NAACCR
NAACCR. Chapter I: Problem Statement, Goals, and Scope of this Document. Chapter II: Historical Background and Status of North American Standards. Chapter III: Standards for Tumor Inclusion and Reportability. Chapter IV: Recommended Data Editors and Software Coordination of Standards. Chapter V: Unresolved Issues.LISTSERVS & PRESS
ListServ – Sending Messages. NAACCR has employed the use of an innovative cross-collaborative software, SHARE by Higher Logic to distribute information to its membership. If you are interested in forwarding useful and pertinent information to the NAACCR membership, simply send an email and format it exactly as you would like it to bedistributed.
NORTH AMERICAN ASSOCIATION OF CENTRAL CANCER REGISTRIES North American Association of Central Cancer Registries, Inc. (NAACCR) 2021 Implementation Guidelines and Recommendations (For NAACCR Data Standards and Data Dictionary, Version 21, effective CANCER REGISTRAR TRAINING GUIDE OVERVIEW . The NAACCR Cancer Registrar Training Guide is intended for use by central cancer registries (CCRs) in training individuals new to the cancer registry profession. PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014.NAACCRWEB VIEW
2300. 7692. TABLES EDITS EDIT SETS FIELDS Deleted LAYOUT Edit name change x Logic Change Edit Set Change Description, Administrative Notes, Error Messages Change GRADE CODING INSTRUCTIONS AND TABLES Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published August 2019 . Version 1.7 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEER2018 IMPLEMENTATION
2018 Implementation Information. This page is intended to be a source of information for central registries, hospital registries, and software vendors. On this site you will find information concerning new data items, edits, rules for determining multiple primaries and histologies, updates to histology codes, and educational activities. ICD O 3 CODING UPDATES These documents address the implementation of ICD-O-3 for cases diagnosed on or after January 1, 2021. ICD O 3.2 Implementation Documents. 2021 ICD O 3.2 Coding Guidelines – 10/05/2020; 2021 ICD O 3.2 Tables 1-5 (tables with new term, new codes, changed behaviors, etc) – 10/01/2020; 2021 ICD O 3.2 Table 6 Numeric (combined tables 1-5 in numeric order) – 11/10/2020 HIPAA RESOURCES FOR CANCER REGISTRIES Only 10 more days to take advantage of the Early Bird savings for the 2021 NAACCR Forum. Get Registered today at t.co/I6cOdbqWEg. Only 15 days left to take advantage of the Early Bird Registration Savings. Get registered before June 6th to save t.co/NFlAfZ9z1X.NAACCR
NAACCR. Chapter I: Problem Statement, Goals, and Scope of this Document. Chapter II: Historical Background and Status of North American Standards. Chapter III: Standards for Tumor Inclusion and Reportability. Chapter IV: Recommended Data Editors and Software Coordination of Standards. Chapter V: Unresolved Issues.LISTSERVS & PRESS
ListServ – Sending Messages. NAACCR has employed the use of an innovative cross-collaborative software, SHARE by Higher Logic to distribute information to its membership. If you are interested in forwarding useful and pertinent information to the NAACCR membership, simply send an email and format it exactly as you would like it to bedistributed.
NORTH AMERICAN ASSOCIATION OF CENTRAL CANCER REGISTRIES North American Association of Central Cancer Registries, Inc. (NAACCR) 2021 Implementation Guidelines and Recommendations (For NAACCR Data Standards and Data Dictionary, Version 21, effective CANCER REGISTRAR TRAINING GUIDE OVERVIEW . The NAACCR Cancer Registrar Training Guide is intended for use by central cancer registries (CCRs) in training individuals new to the cancer registry profession. PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014.NAACCRWEB VIEW
2300. 7692. TABLES EDITS EDIT SETS FIELDS Deleted LAYOUT Edit name change x Logic Change Edit Set Change Description, Administrative Notes, Error Messages Change GRADE CODING INSTRUCTIONS AND TABLES Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published August 2019 . Version 1.7 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEER2018 IMPLEMENTATION
2018 Implementation Information. This page is intended to be a source of information for central registries, hospital registries, and software vendors. On this site you will find information concerning new data items, edits, rules for determining multiple primaries and histologies, updates to histology codes, and educational activities. MEMBERSHIP APPLICATION All International Member applications require an endorsement from an employee of a current NAACCR Full or Sponsoring Member Organization. Prior to completing the membership application, please obtain a name and email of an employee of a current NAACCR Full or Sponsoring Member Organization and enter their name and email PRIOR TO SUBMITTING YOUR NAACCR EXECUTIVE OFFICE STAFF The NAACCR Executive Office is located in Springfield, Illinois. Springfield is the Illinois state capital with a population of 120,000residents.
NORTH AMERICAN ASSOCIATION OF CENTRAL CANCER REGISTRIES North American Association of Central Cancer Registries, Inc. (NAACCR) 2021 Implementation Guidelines and Recommendations (For NAACCR Data Standards and Data Dictionary, Version 21, effectiveLISTSERVS & PRESS
ListServ – Sending Messages. NAACCR has employed the use of an innovative cross-collaborative software, SHARE by Higher Logic to distribute information to its membership. If you are interested in forwarding useful and pertinent information to the NAACCR membership, simply send an email and format it exactly as you would like it to bedistributed.
PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014. GRADE CODING INSTRUCTIONS AND TABLES V2 Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published January 2021 . Version 2.01 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEERSCHEMAS | SSDI DATA
Version 1.7 (For use with NAACCR v18 Layout) SSDI Manual. SSDI Manual Appendix A. SSDI Manual Appendix B. SSDI Manual Appendix C. Grade Manual. Change Log. Comments or suggestions concerning the SSDI’s are welcome and can be posted at the American College of Surgeons CAnswer Forum. Adnexa Uterine Other Adrenal Gland Ampulla of VaterAnus
SSDI MANUAL: APPENDIX A Schema ID 00060: Cervical Lymph Nodes and Unknown Primary . Primary Site(s)/Histology(ies) Primary Site Histology Schema Discriminator 1 . C760 8000-8700, 8720-8790, 8941, 9700-9701 2, 3, 4, 5 NATIONAL CHILDHOOD CANCER REGISTRY Associate Director. shill@naaccr.org. Introducing the National Childhood Cancer Registry. Cancers among children, adolescents, and young adults pose unique challenges and opportunities in the realm of public health surveillance. The rarity of pediatric cancers often poses a barrier to state-by-state investigation and analysis intoissues of
1: MEDICAL TERMINOLOGY AND GENERAL ANATOMY Scenario 2 A patient presents for a routine mammogram and is found to have a 6 cm abnormal mass in the right breast. Additional workup showed the mass extending into the pectoral muscle. HOME EDUCATION & TRAINING CALENDAROTHER RESOURCESPAST ANNUAL CONFERENCESFUTURE ANNUAL CONFERENCES The Virtual Pooled Registry Cancer Linkage System (VPR-CLS) is a one-stop-shop designed to efficiently connect researchers with multiple cancer registries, thereby increasing access to, linkage with, and use of this valuable source of cancer surveillance information. The project is coordinated by the North American Association of Central Cancer ICD O 3 CODING UPDATES These documents address the implementation of ICD-O-3 for cases diagnosed on or after January 1, 2021. ICD O 3.2 Implementation Documents. 2021 ICD O 3.2 Coding Guidelines – 10/05/2020; 2021 ICD O 3.2 Tables 1-5 (tables with new term, new codes, changed behaviors, etc) – 10/01/2020; 2021 ICD O 3.2 Table 6 Numeric (combined tables 1-5 in numeric order) – 11/10/2020 HIPAA RESOURCES FOR CANCER REGISTRIES Only 10 more days to take advantage of the Early Bird savings for the 2021 NAACCR Forum. Get Registered today at t.co/I6cOdbqWEg. Only 15 days left to take advantage of the Early Bird Registration Savings. Get registered before June 6th to save t.co/NFlAfZ9z1X.NAACCR
NAACCR. Chapter I: Problem Statement, Goals, and Scope of this Document. Chapter II: Historical Background and Status of North American Standards. Chapter III: Standards for Tumor Inclusion and Reportability. Chapter IV: Recommended Data Editors and Software Coordination of Standards. Chapter V: Unresolved Issues. STANDARDS FOR CANCER REGISTRIES, STANDARD DATA EDITS For previous versions, or questions please contact Jim Hofferkamp, NAACCR Program Manager of Education and Training at jhofferkamp@naaccr.org or (217) 698-0800 x5. Questions on current edits or suggestions for new edits may be submitted to the Edits Forum for Vendors and Central Registry Metafile Administrators. FUTURE ANNUAL CONFERENCE DATES AND LOCATIONS 2022– Boise, Idaho. The Grove Hotel. June 06-14, 2022. Single/Double Room Rate: $182 US. Residence Inn. June 06-14, 2022. Single/DoubleRoom Rate: $169 US.
CANCER REGISTRAR TRAINING GUIDE OVERVIEW . The NAACCR Cancer Registrar Training Guide is intended for use by central cancer registries (CCRs) in training individuals new to the cancer registry profession. PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014. GRADE CODING INSTRUCTIONS AND TABLES Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published August 2019 . Version 1.7 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEER2018 IMPLEMENTATION
2018 Implementation Information. This page is intended to be a source of information for central registries, hospital registries, and software vendors. On this site you will find information concerning new data items, edits, rules for determining multiple primaries and histologies, updates to histology codes, and educational activities. HOME EDUCATION & TRAINING CALENDAROTHER RESOURCESPAST ANNUAL CONFERENCESFUTURE ANNUAL CONFERENCES The Virtual Pooled Registry Cancer Linkage System (VPR-CLS) is a one-stop-shop designed to efficiently connect researchers with multiple cancer registries, thereby increasing access to, linkage with, and use of this valuable source of cancer surveillance information. The project is coordinated by the North American Association of Central Cancer ICD O 3 CODING UPDATES These documents address the implementation of ICD-O-3 for cases diagnosed on or after January 1, 2021. ICD O 3.2 Implementation Documents. 2021 ICD O 3.2 Coding Guidelines – 10/05/2020; 2021 ICD O 3.2 Tables 1-5 (tables with new term, new codes, changed behaviors, etc) – 10/01/2020; 2021 ICD O 3.2 Table 6 Numeric (combined tables 1-5 in numeric order) – 11/10/2020 HIPAA RESOURCES FOR CANCER REGISTRIES Only 10 more days to take advantage of the Early Bird savings for the 2021 NAACCR Forum. Get Registered today at t.co/I6cOdbqWEg. Only 15 days left to take advantage of the Early Bird Registration Savings. Get registered before June 6th to save t.co/NFlAfZ9z1X.NAACCR
NAACCR. Chapter I: Problem Statement, Goals, and Scope of this Document. Chapter II: Historical Background and Status of North American Standards. Chapter III: Standards for Tumor Inclusion and Reportability. Chapter IV: Recommended Data Editors and Software Coordination of Standards. Chapter V: Unresolved Issues. STANDARDS FOR CANCER REGISTRIES, STANDARD DATA EDITS For previous versions, or questions please contact Jim Hofferkamp, NAACCR Program Manager of Education and Training at jhofferkamp@naaccr.org or (217) 698-0800 x5. Questions on current edits or suggestions for new edits may be submitted to the Edits Forum for Vendors and Central Registry Metafile Administrators. FUTURE ANNUAL CONFERENCE DATES AND LOCATIONS 2022– Boise, Idaho. The Grove Hotel. June 06-14, 2022. Single/Double Room Rate: $182 US. Residence Inn. June 06-14, 2022. Single/DoubleRoom Rate: $169 US.
CANCER REGISTRAR TRAINING GUIDE OVERVIEW . The NAACCR Cancer Registrar Training Guide is intended for use by central cancer registries (CCRs) in training individuals new to the cancer registry profession. PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014. GRADE CODING INSTRUCTIONS AND TABLES Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published August 2019 . Version 1.7 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEER2018 IMPLEMENTATION
2018 Implementation Information. This page is intended to be a source of information for central registries, hospital registries, and software vendors. On this site you will find information concerning new data items, edits, rules for determining multiple primaries and histologies, updates to histology codes, and educational activities. MEMBERSHIP APPLICATION All International Member applications require an endorsement from an employee of a current NAACCR Full or Sponsoring Member Organization. Prior to completing the membership application, please obtain a name and email of an employee of a current NAACCR Full or Sponsoring Member Organization and enter their name and email PRIOR TO SUBMITTING YOURNAACCR
NAACCR. Chapter I: Problem Statement, Goals, and Scope of this Document. Chapter II: Historical Background and Status of North American Standards. Chapter III: Standards for Tumor Inclusion and Reportability. Chapter IV: Recommended Data Editors and Software Coordination of Standards. Chapter V: Unresolved Issues.LISTSERVS & PRESS
ListServ – Sending Messages. NAACCR has employed the use of an innovative cross-collaborative software, SHARE by Higher Logic to distribute information to its membership. If you are interested in forwarding useful and pertinent information to the NAACCR membership, simply send an email and format it exactly as you would like it to bedistributed.
NAACCR EXECUTIVE OFFICE STAFF The NAACCR Executive Office is located in Springfield, Illinois. Springfield is the Illinois state capital with a population of 120,000residents.
NORTH AMERICAN ASSOCIATION OF CENTRAL CANCER REGISTRIES North American Association of Central Cancer Registries, Inc. (NAACCR) 2021 Implementation Guidelines and Recommendations (For NAACCR Data Standards and Data Dictionary, Version 21, effectiveABOUT VPR-CLS
The Virtual Pooled Registry Cancer Linkage System (VPR-CLS) is a secure online service designed to: efficiently connect researchers performing minimal risk linkage studies with multiple U.S. population-based cancer registries; perform linkages utilize a single cohort file, standard linkage software, and consistent matchingalgorithms;
PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014.NAACCRWEB VIEW
2300. 7692. TABLES EDITS EDIT SETS FIELDS Deleted LAYOUT Edit name change x Logic Change Edit Set Change Description, Administrative Notes, Error Messages Change GRADE CODING INSTRUCTIONS AND TABLES V2 Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published January 2021 . Version 2.01 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEER NATIONAL CHILDHOOD CANCER REGISTRY Associate Director. shill@naaccr.org. Introducing the National Childhood Cancer Registry. Cancers among children, adolescents, and young adults pose unique challenges and opportunities in the realm of public health surveillance. The rarity of pediatric cancers often poses a barrier to state-by-state investigation and analysis intoissues of
HOME EDUCATION & TRAINING CALENDAROTHER RESOURCESPAST ANNUAL CONFERENCESFUTURE ANNUAL CONFERENCES The Virtual Pooled Registry Cancer Linkage System (VPR-CLS) is a one-stop-shop designed to efficiently connect researchers with multiple cancer registries, thereby increasing access to, linkage with, and use of this valuable source of cancer surveillance information. The project is coordinated by the North American Association of Central Cancer ICD O 3 CODING UPDATES These documents address the implementation of ICD-O-3 for cases diagnosed on or after January 1, 2021. ICD O 3.2 Implementation Documents. 2021 ICD O 3.2 Coding Guidelines – 10/05/2020; 2021 ICD O 3.2 Tables 1-5 (tables with new term, new codes, changed behaviors, etc) – 10/01/2020; 2021 ICD O 3.2 Table 6 Numeric (combined tables 1-5 in numeric order) – 11/10/2020 HIPAA RESOURCES FOR CANCER REGISTRIES Only 10 more days to take advantage of the Early Bird savings for the 2021 NAACCR Forum. Get Registered today at t.co/I6cOdbqWEg. Only 15 days left to take advantage of the Early Bird Registration Savings. Get registered before June 6th to save t.co/NFlAfZ9z1X.NAACCR
NAACCR. Chapter I: Problem Statement, Goals, and Scope of this Document. Chapter II: Historical Background and Status of North American Standards. Chapter III: Standards for Tumor Inclusion and Reportability. Chapter IV: Recommended Data Editors and Software Coordination of Standards. Chapter V: Unresolved Issues. STANDARDS FOR CANCER REGISTRIES, STANDARD DATA EDITS For previous versions, or questions please contact Jim Hofferkamp, NAACCR Program Manager of Education and Training at jhofferkamp@naaccr.org or (217) 698-0800 x5. Questions on current edits or suggestions for new edits may be submitted to the Edits Forum for Vendors and Central Registry Metafile Administrators. FUTURE ANNUAL CONFERENCE DATES AND LOCATIONS 2022– Boise, Idaho. The Grove Hotel. June 06-14, 2022. Single/Double Room Rate: $182 US. Residence Inn. June 06-14, 2022. Single/DoubleRoom Rate: $169 US.
CANCER REGISTRAR TRAINING GUIDE OVERVIEW . The NAACCR Cancer Registrar Training Guide is intended for use by central cancer registries (CCRs) in training individuals new to the cancer registry profession. PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014.NAACCRWEB VIEW
2300. 7692. TABLES EDITS EDIT SETS FIELDS Deleted LAYOUT Edit name change x Logic Change Edit Set Change Description, Administrative Notes, Error Messages Change GRADE CODING INSTRUCTIONS AND TABLES Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published August 2019 . Version 1.7 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEER HOME EDUCATION & TRAINING CALENDAROTHER RESOURCESPAST ANNUAL CONFERENCESFUTURE ANNUAL CONFERENCES The Virtual Pooled Registry Cancer Linkage System (VPR-CLS) is a one-stop-shop designed to efficiently connect researchers with multiple cancer registries, thereby increasing access to, linkage with, and use of this valuable source of cancer surveillance information. The project is coordinated by the North American Association of Central Cancer ICD O 3 CODING UPDATES These documents address the implementation of ICD-O-3 for cases diagnosed on or after January 1, 2021. ICD O 3.2 Implementation Documents. 2021 ICD O 3.2 Coding Guidelines – 10/05/2020; 2021 ICD O 3.2 Tables 1-5 (tables with new term, new codes, changed behaviors, etc) – 10/01/2020; 2021 ICD O 3.2 Table 6 Numeric (combined tables 1-5 in numeric order) – 11/10/2020 HIPAA RESOURCES FOR CANCER REGISTRIES Only 10 more days to take advantage of the Early Bird savings for the 2021 NAACCR Forum. Get Registered today at t.co/I6cOdbqWEg. Only 15 days left to take advantage of the Early Bird Registration Savings. Get registered before June 6th to save t.co/NFlAfZ9z1X.NAACCR
NAACCR. Chapter I: Problem Statement, Goals, and Scope of this Document. Chapter II: Historical Background and Status of North American Standards. Chapter III: Standards for Tumor Inclusion and Reportability. Chapter IV: Recommended Data Editors and Software Coordination of Standards. Chapter V: Unresolved Issues. STANDARDS FOR CANCER REGISTRIES, STANDARD DATA EDITS For previous versions, or questions please contact Jim Hofferkamp, NAACCR Program Manager of Education and Training at jhofferkamp@naaccr.org or (217) 698-0800 x5. Questions on current edits or suggestions for new edits may be submitted to the Edits Forum for Vendors and Central Registry Metafile Administrators. FUTURE ANNUAL CONFERENCE DATES AND LOCATIONS 2022– Boise, Idaho. The Grove Hotel. June 06-14, 2022. Single/Double Room Rate: $182 US. Residence Inn. June 06-14, 2022. Single/DoubleRoom Rate: $169 US.
CANCER REGISTRAR TRAINING GUIDE OVERVIEW . The NAACCR Cancer Registrar Training Guide is intended for use by central cancer registries (CCRs) in training individuals new to the cancer registry profession. PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014.NAACCRWEB VIEW
2300. 7692. TABLES EDITS EDIT SETS FIELDS Deleted LAYOUT Edit name change x Logic Change Edit Set Change Description, Administrative Notes, Error Messages Change GRADE CODING INSTRUCTIONS AND TABLES Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published August 2019 . Version 1.7 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEER MEMBERSHIP APPLICATION All International Member applications require an endorsement from an employee of a current NAACCR Full or Sponsoring Member Organization. Prior to completing the membership application, please obtain a name and email of an employee of a current NAACCR Full or Sponsoring Member Organization and enter their name and email PRIOR TO SUBMITTING YOUR STANDARDS FOR CANCER REGISTRIES, STANDARD DATA EDITS For previous versions, or questions please contact Jim Hofferkamp, NAACCR Program Manager of Education and Training at jhofferkamp@naaccr.org or (217) 698-0800 x5. Questions on current edits or suggestions for new edits may be submitted to the Edits Forum for Vendors and Central Registry Metafile Administrators. NORTH AMERICAN ASSOCIATION OF CENTRAL CANCER REGISTRIES North American Association of Central Cancer Registries, Inc. (NAACCR) 2021 Implementation Guidelines and Recommendations (For NAACCR Data Standards and Data Dictionary, Version 21, effective NAACCR EXECUTIVE OFFICE STAFF The NAACCR Executive Office is located in Springfield, Illinois. Springfield is the Illinois state capital with a population of 120,000residents.
PATHOLOGY LABORATORY ELECTRONIC REPORTING, VOLUME V Pathology Laboratory Electronic Reporting Version 4.0 – April 2011. Includes HL7 Version 2.5.1 Implementation Guidelines with rules for formatting messages carrying synoptic reports (Chapter 3). Summary of Changes from Version 3 to 4. List of Mnemonic Codes for Standards Volume V, Version 4.0, Appendix A Code Tables – May 2014. NAACCR CTR EXAM READINESS WEBINAR SYLLABUS NAACCR CTR Exam Preparation & Review Webinar Syllabus . Preparation for the June 2021 CTR Exam . 1:30 pm until 3:30 pm Eastern Time (times are subject to change) GRADE CODING INSTRUCTIONS AND TABLES Grade Coding Instructions and Tables . Effective with Cases Diagnosed 1/1/2018 and Forward . Published September 2020 . Version 2.0 . Editors: Jennifer Ruhl, MSHCA, RHIT, CCS, CTR, NCI SEERNAACCRWEB VIEW
2300. 7692. TABLES EDITS EDIT SETS FIELDS Deleted LAYOUT Edit name change x Logic Change Edit Set Change Description, Administrative Notes, Error Messages Change NATIONAL CHILDHOOD CANCER REGISTRY Associate Director. shill@naaccr.org. Introducing the National Childhood Cancer Registry. Cancers among children, adolescents, and young adults pose unique challenges and opportunities in the realm of public health surveillance. The rarity of pediatric cancers often poses a barrier to state-by-state investigation and analysis intoissues of
2018 IMPLEMENTATION
2018 Implementation Information. This page is intended to be a source of information for central registries, hospital registries, and software vendors. On this site you will find information concerning new data items, edits, rules for determining multiple primaries and histologies, updates to histology codes, and educational activities. NAACCR Education and Training Education & Training Calendar now available!Text Layer
2020 Fall Narrative is now availableLogin
Register
*
*
*
*
Toggle navigation
* Education
* Annual Conference
* NAACCR Annual Conference * Past Annual Conferences(Programs,
Presentations, CE Certificate, Photos) * Future Annual Conferences(Dates
and Locations)
* NAACCR Forum
* 2021 NAACCR FORUM
* Resources & Training * NAACCR Education and Training (NET) * Recruitment & Retention Toolkit* Certification
* Certification
* Certification Criteria(Levels and Logos)
* Certified Registries(Who is Certified?)
* Call for Data (Each year, NAACCR has a Call for Data for its certification process and to produce a new set of Cancer in North America (CiNA) data products.) * Central Registry Standards * Central Registry Standards * Data Exchange Standards & Record Descriptions(Volume
I)
* Data Standards & Data Dictionary(Volume II)
* Standards for Completeness, Quality, Analysis, and Management ofData
(Volume
III)
* Standards for Cancer Registries, Standard Data Edits(Volume IV)
* Pathology Laboratory Electronic Reporting(Volume
V)
* Cancer Surveillance Timeline * ICD O 3 Coding Updates * Implementation Guidelines* Meaningful Use
* National Interstate Data Exchange Agreement * Registry Operations Guidelines * Site Specific Data Items (SSDI) * v21 Reference Page * XML Data Exchange Standard* Data & Statistics
* NAACCR Data & Statistics * Annual Report to Nation* Top 5 Cancers
* Cancer in North America CiNA Volumes * On-Line Cancer Data(Interactive)
* Other Data Links
* Canadian Cancer Statistics* Call For Data
* Cancer in North America CiNA Data Products * Research & Analytic Tools * Access Data for Research * Cancer in North America CiNA Data Products * Data Quality Assessments and Evaluations * IRB & Common Rule Materials * Analysis Resources & Tools Overview * Analysis and Data Improvement Tools* Delay Adjustment
* GIS Resources
* Confidentiality Issues * Path Lab Search Terms * Crosswalks (Interoperability)* Other Resources
* Annual Report to the Nation* Data Dictionary
* National Data Exchange Agreements * Research & Data Use Blog(NAACCReview)
* Research & Data Use Cancer Surveillance Talks * Virtual Pooled Registry * Virtual Pooled Registry * Virtual Pooled Registry* About VPR-CLS
* How it works
* Apply for Linkage
(Application, Fees)
* IRB Activities
* Inter-Registry Deduplication * ORGANIZATION & MEMBERSHIP* ORGANIZATION
* About NAACCR
* Strategic Management Plan* NAACCR Board
* NAACCR Staff
* Bylaws, Policies, Standing Rules* Board Resolutions
* Call for Nominations* Annual Report
* Business Meetings
* Partners and Sponsors * Institutional Liaisons* Become a Member
* MEMBERSHIP & COMMUNICATIONS * Steering Committees(Rosters, Contact,
Minutes)
* Membership Directory* Job Opportunities
* Member Awards
* Contact Us
* NAACCReview Blog
* ListServs & Press
* Forums
* Narrative Newsletter* MyNAACCR Login
(Access
to Membership Account) * SHARE Workspace Login (Collaboration workspace for Committees& Workgroups)
NAACCR Mission
NAACCR is a professional organization that develops and promotes uniform data standards for cancer registration Promotes the use of cancer surveillance data and systems for cancercontrol and
epidemiologic research, public health programs, and patient care Makes available a variety of standards and technical assistance documents as well as cancerincidence data
RESOURCES AND PROJECTS Data Standards & Data Dictionary (Volume II) Resources for International Registries Cancer Surveillance Timeline Site Specific Data Items (SSDI) Cancer Data & Maps(interactive)
ANNOUNCEMENTS
* 2021 Forum Abstract Submission* 2021 NAACCR Forum
* NAACCR XML Resources and Timeline * Fall Narrative Now Available * 2020 Annual Report to the Nation * NAACCR Call for Nominations * Enrico Anglesio Prize 2020 Virtual * New CiNA Prevalence Now AvailableVPR-CLS
VIRTUAL POOLED REGISTRY CANCER LINKAGE SYSTEM (VPR‐CLS) The Virtual Pooled Registry Cancer Linkage System (VPR-CLS) is a one-stop-shop designed to efficiently connect researchers with multiple cancer registries, thereby increasing access to, linkage with, and use of this valuable source of cancer surveillance information. The project is coordinated by the North American Association of Central Cancer Registries (NAACCR) and funded by the National Cancer Institute (NCI). The VPR-CLS is currently in the development and testing phase and is progressing with input from national partners, researchers, and central registry staff.Read More
USEFUL LINKS
* Annual Conference
* Get Cancer Data
* Strategic Management Plan* MyNAACCR
* SHARE Committee Workspace* NAACCR Fast Stats
* Contact Us
* ListServ Archives
* NAACCReview
* Forums
* Narrative
* Job Opportunities
Click here to send a ListServFOLLOW US
Jill Barnholtz-Sloan, PhD@JBarnholtzSloan
So thrilled to highlight my colleagues Gino Cioffi, MPH, and Nirav Patil, MBBS, MPH, who will present virtually, No… t.co/ec0rBp9rsq __ Retweeted by NAACCR, Inc.NAACCR, Inc.
@NAACCR
We are pleased to introduce NAACCR Summer FORUM June 15-17. This remote experience will offer customary high-qualit… t.co/umTK4snOpNRECENT POSTS
Cancer Surveillance and the 2020 Economy: Thinking about Industrial/Occupational (I/O) Data in Current ContextAugust 28, 2020
Disaggregated Hispanic Groups and Cancer: Importance, Methodology, and Current KnowledgeMarch 31, 2020
CONTACT
(217) 698-0800
(217) 698-0188
info@naaccr.org
Contact Us
NAACCR Staff
NAACCR Board
Funding for this web site content was made possible in part by a contract with Federal funds from the National Cancer Institute, National Institutes of Health and Department of Health & Human Services under Contract number HHSN261201400004I / HHSN26100002. Additionally, funding for this web site content was made possible in part by a cooperative agreement with Federal funds from the Centers for Disease Control and Prevention Cooperative Agreement number 5NU58DP006458. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NCIand CDC.
Copyright © 2018 NAACCR , Inc. All Rights Reserved | See NAACCR Partners and SponsorsScroll To Top
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0