Are you over 18 and want to see adult content?
More Annotations
A complete backup of fanoosphysio.com
Are you over 18 and want to see adult content?
A complete backup of sigtunaedu.sharepoint.com
Are you over 18 and want to see adult content?
A complete backup of online-puzzle.ru
Are you over 18 and want to see adult content?
A complete backup of heidelbergcement.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of metalandmineral.wordpress.com
Are you over 18 and want to see adult content?

A complete backup of worshipandpraisesongs.blogspot.com
Are you over 18 and want to see adult content?

A complete backup of vikingeborgen.dk
Are you over 18 and want to see adult content?

A complete backup of newgrandmart.com
Are you over 18 and want to see adult content?

A complete backup of sam-cv.herokuapp.com
Are you over 18 and want to see adult content?

A complete backup of aceperipherals.com
Are you over 18 and want to see adult content?

A complete backup of online-webradio.com
Are you over 18 and want to see adult content?
Text
in the world. 5.
OXFORD UNIVERSITY BREAKTHROUGH ON GLOBAL COVID-19 VACCINE Oxford, UK – Vaccitech’s scientific founders at University of Oxford announce positive high-level results from an interim analysis of clinical trials of AZD1222 in the UK and Brazil. Phase 3 interim analysis including 131 Covid-19 cases indicates that the vaccine is 70.4% effective when combining data from two dosing regimens In thetwo different dose
INVESTORS | VACCITECH Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centres in the world. Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologousprime-boost
TECHNOLOGY - VACCITECH A clinical stage company utilizing a world leading T cell-inducing platform. The patented Vaccitech adenovirus vectors are known as chimpanzee adenovirus Oxford 1 and 2 (ChAdOx1 and ChAdOx2), and are in the group E simian adenovirus family, similar to the widely-studied chimpanzee adenovirus 63. These viruses have been engineered to be PIPELINE - VACCITECH Vaccitech is developing VTP-500, its prophylactic vaccine product candidate, to prevent infection and subsequent disease caused by MERS-CoV. VTP-500 is based on the use of one or two doses of ChAdOx1 encoding the spike glycoprotein of MERS-CoV and was developed at the University of Oxford. Development status. OUR TEAM - VACCITECH Dr. Margaret (Meg) Marshall has more than 25 years of experience in the preclinical and clinical development of cell and antibody-based immunotherapies for infectious diseases and cancer, including vaccines, checkpoint inhibitors, and cell therapies such as CAR T cells. Prior to joining Vaccitech, she held directorship positions inthe clinical
VACANCY: SENIOR SCIENTIST Vacancy: Senior Scientist – Process Development. Aug 24, 2020. Due to exciting expansion across the company, with offices in the UK (headquarters), USA, Italy and Australia, Vaccitech is now recruiting for a Senior Scientist – Process Development, which is VACCITECH RECEIVES £6M INVESTMENT FROM GENEMATRIX AND Vaccitech will progress existing vaccine programs and the collaboration will combine Vaccitech’s know-how with GeneMatrix’s clinical network. Oxford, UK and Seoul, South Korea Vaccitech, a clinical stage developer of a universal flu vaccine and other vaccine-related products, has received a joint £6m investment from a Korean biotech company, GeneMatrix, and venture capital and private INDUCING T CELLS TO TREAT AND PREVENT DISEASE AZ-OUI licensed COVID program(1) may provide us with revenue beginning as early as H2 2021 Clinical data in both therapeutic (prostate cancer) and prophylactic (COVID, MERS) programs supports further development with our platform Application of the platform for pandemic vaccine programs provides potential for response to ongoing emerginginfections
CMC PROJECT MANAGER
2 CMC Project Manager Essential knowledge, experience and skills: • BSc or equivalent experience in science/technical. • Minimum 3 years’ experience of biologics, VACCITECH - CREATING WAYS TO TREAT AND PREVENT DISEASE.ABOUTTECHNOLOGYPIPELINEPARTNERSHIPSNEWS & UPDATESCAREERS We discover and develop immunotherapeutics products and vaccines for the treatment and prevention of infectious diseases and cancer. Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centresin the world. 5.
OXFORD UNIVERSITY BREAKTHROUGH ON GLOBAL COVID-19 VACCINE Oxford, UK – Vaccitech’s scientific founders at University of Oxford announce positive high-level results from an interim analysis of clinical trials of AZD1222 in the UK and Brazil. Phase 3 interim analysis including 131 Covid-19 cases indicates that the vaccine is 70.4% effective when combining data from two dosing regimens In thetwo different dose
INVESTORS | VACCITECH Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centres in the world. Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologousprime-boost
TECHNOLOGY - VACCITECH A clinical stage company utilizing a world leading T cell-inducing platform. The patented Vaccitech adenovirus vectors are known as chimpanzee adenovirus Oxford 1 and 2 (ChAdOx1 and ChAdOx2), and are in the group E simian adenovirus family, similar to the widely-studied chimpanzee adenovirus 63. These viruses have been engineered to be PIPELINE - VACCITECH Vaccitech is developing VTP-500, its prophylactic vaccine product candidate, to prevent infection and subsequent disease caused by MERS-CoV. VTP-500 is based on the use of one or two doses of ChAdOx1 encoding the spike glycoprotein of MERS-CoV and was developed at the University of Oxford. Development status. OUR TEAM - VACCITECH Dr. Margaret (Meg) Marshall has more than 25 years of experience in the preclinical and clinical development of cell and antibody-based immunotherapies for infectious diseases and cancer, including vaccines, checkpoint inhibitors, and cell therapies such as CAR T cells. Prior to joining Vaccitech, she held directorship positions inthe clinical
VACANCY: SENIOR SCIENTIST Vacancy: Senior Scientist – Process Development. Aug 24, 2020. Due to exciting expansion across the company, with offices in the UK (headquarters), USA, Italy and Australia, Vaccitech is now recruiting for a Senior Scientist – Process Development, which is VACCITECH RECEIVES £6M INVESTMENT FROM GENEMATRIX AND Vaccitech will progress existing vaccine programs and the collaboration will combine Vaccitech’s know-how with GeneMatrix’s clinical network. Oxford, UK and Seoul, South Korea Vaccitech, a clinical stage developer of a universal flu vaccine and other vaccine-related products, has received a joint £6m investment from a Korean biotech company, GeneMatrix, and venture capital and private INDUCING T CELLS TO TREAT AND PREVENT DISEASE AZ-OUI licensed COVID program(1) may provide us with revenue beginning as early as H2 2021 Clinical data in both therapeutic (prostate cancer) and prophylactic (COVID, MERS) programs supports further development with our platform Application of the platform for pandemic vaccine programs provides potential for response to ongoing emerginginfections
CMC PROJECT MANAGER
2 CMC Project Manager Essential knowledge, experience and skills: • BSc or equivalent experience in science/technical. • Minimum 3 years’ experience of biologics, TECHNOLOGY - VACCITECH A clinical stage company utilizing a world leading T cell-inducing platform. The patented Vaccitech adenovirus vectors are known as chimpanzee adenovirus Oxford 1 and 2 (ChAdOx1 and ChAdOx2), and are in the group E simian adenovirus family, similar to the widely-studied chimpanzee adenovirus 63. These viruses have been engineered to beABOUT - VACCITECH
Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologous prime-boost platform comprises Chimpanzee Adenovirus Oxford (ChAdOx) and Modified Vaccinia Ankara (MVA), two non-replicating viral vectors which safely mimic NEW MERS CORONAVIRUS VACCINE CLINICAL TRIAL STARTS IN Vaccitech’s partner, the University of Oxford’s Jenner Institute, in collaboration with The King Abdullah International Medical Research Centre (KAIMRC), announced that it has started a Phase I clinical trial in the Kingdom of Saudi Arabia (KSA) for a vaccine against Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Vaccitech retains commercial rights to this vaccine. The trial NEWSROOM - VACCITECH Mar 17, 2021. Oxford, UK - Vaccitech Ltd (“Vaccitech”), a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious diseases and cancer, today announces thatit has
VACCITECH UNIVERSAL FLU VACCINE PASSES PHASE 2B CLINICAL Vaccitech initiates Phase 2b clinical studies for its MVA-NP+M1 universal flu vaccine (VTP-100) in Belgium and completes recruitment in Australia Oxford, United Kingdom – 5 June 2019: Vaccitech Ltd. announces that it has administered its pandemic universal influenza A vaccine MVA-NP+M1 (VTP-100) to the first participants in a Flu 010 study – a Phase 2b,PARTNERSHIPS
Our Partners. Vaccitech has established partnerships with leading institutions across the globe to support the development of its programs. Hover over the logos below for more information. Vaccitech has entered into non-exclusive licensure of its T cell induction platform in oncology, and is committed to advancing the use of itsplatform for
FIRST PARTICIPANT DOSED IN PHASE 1 CLINICAL STUDY WITH VTP Oxford, UK – Vaccitech Ltd, a clinical stage biopharmaceutical company developing immunotherapies to treat and prevent infectious diseases and cancer, announces today that it has dosed the first person in a Phase 1 clinical study to investigate the safety and immunogenicity of the VTP-300 prime in both healthy participants and patients with chronic hepatitis B VACCITECH SECURES £20M SERIES A WITH GV, OSI AND SEQUOIA Vaccitech, an Oxford University spinout company developing a universal flu vaccine, among other vaccine-related products, has secured £20m ($27.1m) in Series A financing. The round was co-led by new investors GV, Sequoia China, and existing backer Oxford Sciences Innovation, which manages a £600m fund aimed at Oxford University spinouts. Neptune Funds joined in participation. In PHASE 1 DATA ON THE VACCITECH PROSTATE CANCER VACCINE Vaccitech Limited, a UK company developing products to treat and prevent multiple infectious diseases and cancer, today announced the presentation of Phase 1 prostate cancer vaccine data at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. In this “window” study, funded by the European Commission FP7 programme, 40 patients with low orCMC PROJECT MANAGER
2 CMC Project Manager Essential knowledge, experience and skills: • BSc or equivalent experience in science/technical. • Minimum 3 years’ experience of biologics, VACCITECH - CREATING WAYS TO TREAT AND PREVENT DISEASE.ABOUTTECHNOLOGYPIPELINEPARTNERSHIPSNEWS & UPDATESCAREERS We discover and develop immunotherapeutics products and vaccines for the treatment and prevention of infectious diseases and cancer. Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centresin the world. 5.
INVESTORS | VACCITECH Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centres in the world. Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologousprime-boost
OUR TEAM - VACCITECH Dr. Margaret (Meg) Marshall has more than 25 years of experience in the preclinical and clinical development of cell and antibody-based immunotherapies for infectious diseases and cancer, including vaccines, checkpoint inhibitors, and cell therapies such as CAR T cells. Prior to joining Vaccitech, she held directorship positions inthe clinical
VACCITECH RECEIVES £6M INVESTMENT FROM GENEMATRIX AND Vaccitech will progress existing vaccine programs and the collaboration will combine Vaccitech’s know-how with GeneMatrix’s clinical network. Oxford, UK and Seoul, South Korea Vaccitech, a clinical stage developer of a universal flu vaccine and other vaccine-related products, has received a joint £6m investment from a Korean biotech company, GeneMatrix, and venture capital and private VACCITECH UNIVERSAL FLU VACCINE PASSES PHASE 2B CLINICAL Vaccitech initiates Phase 2b clinical studies for its MVA-NP+M1 universal flu vaccine (VTP-100) in Belgium and completes recruitment in Australia Oxford, United Kingdom – 5 June 2019: Vaccitech Ltd. announces that it has administered its pandemic universal influenza A vaccine MVA-NP+M1 (VTP-100) to the first participants in a Flu 010 study – a Phase 2b, VACANCY: SENIOR SCIENTIST Vacancy: Senior Scientist – Process Development. Aug 24, 2020. Due to exciting expansion across the company, with offices in the UK (headquarters), USA, Italy and Australia, Vaccitech is now recruiting for a Senior Scientist – Process Development, which is VACCITECH AND OXFORD UNIVERSITY ANNOUNCE PROMISING PHASE Oxford, UK – Vaccitech Limited and the University of Oxford today announce initial efficacy and safety data for ADVANCE, a Phase 2a study testing VTP-800, an immunotherapeutic product candidate in patients with metastatic castration resistant prostate cancer (mCRPC). The study demonstrated that VTP-800 is safe and showed an encouraging efficacy trend in patients with VACCITECH APPOINTS BILL ENRIGHT AS NEW CHIEF EXECUTIVE Vaccitech, a clinical-stage T cell immunotherapy company developing products to treat and prevent cancer and infectious diseases (ID), announced today that it has appointed Bill Enright as its new Chief Executive Officer (CEO). Mr. Enright succeeds Tom Evans, MD who will make a planned transition to the role of Vaccitech’s Chief Scientific Officer. Bill joins PHASE 1 DATA ON THE VACCITECH PROSTATE CANCER VACCINE Vaccitech Limited, a UK company developing products to treat and prevent multiple infectious diseases and cancer, today announced the presentation of Phase 1 prostate cancer vaccine data at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. In this “window” study, funded by the European Commission FP7 programme, 40 patients with low or INDUCING T CELLS TO TREAT AND PREVENT DISEASE AZ-OUI licensed COVID program(1) may provide us with revenue beginning as early as H2 2021 Clinical data in both therapeutic (prostate cancer) and prophylactic (COVID, MERS) programs supports further development with our platform Application of the platform for pandemic vaccine programs provides potential for response to ongoing emerginginfections
VACCITECH - CREATING WAYS TO TREAT AND PREVENT DISEASE.ABOUTTECHNOLOGYPIPELINEPARTNERSHIPSNEWS & UPDATESCAREERS We discover and develop immunotherapeutics products and vaccines for the treatment and prevention of infectious diseases and cancer. Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centresin the world. 5.
INVESTORS | VACCITECH Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centres in the world. Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologousprime-boost
OUR TEAM - VACCITECH Dr. Margaret (Meg) Marshall has more than 25 years of experience in the preclinical and clinical development of cell and antibody-based immunotherapies for infectious diseases and cancer, including vaccines, checkpoint inhibitors, and cell therapies such as CAR T cells. Prior to joining Vaccitech, she held directorship positions inthe clinical
VACCITECH RECEIVES £6M INVESTMENT FROM GENEMATRIX AND Vaccitech will progress existing vaccine programs and the collaboration will combine Vaccitech’s know-how with GeneMatrix’s clinical network. Oxford, UK and Seoul, South Korea Vaccitech, a clinical stage developer of a universal flu vaccine and other vaccine-related products, has received a joint £6m investment from a Korean biotech company, GeneMatrix, and venture capital and private VACCITECH UNIVERSAL FLU VACCINE PASSES PHASE 2B CLINICAL Vaccitech initiates Phase 2b clinical studies for its MVA-NP+M1 universal flu vaccine (VTP-100) in Belgium and completes recruitment in Australia Oxford, United Kingdom – 5 June 2019: Vaccitech Ltd. announces that it has administered its pandemic universal influenza A vaccine MVA-NP+M1 (VTP-100) to the first participants in a Flu 010 study – a Phase 2b, VACANCY: SENIOR SCIENTIST Vacancy: Senior Scientist – Process Development. Aug 24, 2020. Due to exciting expansion across the company, with offices in the UK (headquarters), USA, Italy and Australia, Vaccitech is now recruiting for a Senior Scientist – Process Development, which is VACCITECH AND OXFORD UNIVERSITY ANNOUNCE PROMISING PHASE Oxford, UK – Vaccitech Limited and the University of Oxford today announce initial efficacy and safety data for ADVANCE, a Phase 2a study testing VTP-800, an immunotherapeutic product candidate in patients with metastatic castration resistant prostate cancer (mCRPC). The study demonstrated that VTP-800 is safe and showed an encouraging efficacy trend in patients with VACCITECH APPOINTS BILL ENRIGHT AS NEW CHIEF EXECUTIVE Vaccitech, a clinical-stage T cell immunotherapy company developing products to treat and prevent cancer and infectious diseases (ID), announced today that it has appointed Bill Enright as its new Chief Executive Officer (CEO). Mr. Enright succeeds Tom Evans, MD who will make a planned transition to the role of Vaccitech’s Chief Scientific Officer. Bill joins PHASE 1 DATA ON THE VACCITECH PROSTATE CANCER VACCINE Vaccitech Limited, a UK company developing products to treat and prevent multiple infectious diseases and cancer, today announced the presentation of Phase 1 prostate cancer vaccine data at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. In this “window” study, funded by the European Commission FP7 programme, 40 patients with low or INDUCING T CELLS TO TREAT AND PREVENT DISEASE AZ-OUI licensed COVID program(1) may provide us with revenue beginning as early as H2 2021 Clinical data in both therapeutic (prostate cancer) and prophylactic (COVID, MERS) programs supports further development with our platform Application of the platform for pandemic vaccine programs provides potential for response to ongoing emerginginfections
OXFORD UNIVERSITY BREAKTHROUGH ON GLOBAL COVID-19 VACCINE Oxford, UK – Vaccitech’s scientific founders at University of Oxford announce positive high-level results from an interim analysis of clinical trials of AZD1222 in the UK and Brazil. Phase 3 interim analysis including 131 Covid-19 cases indicates that the vaccine is 70.4% effective when combining data from two dosing regimens In thetwo different dose
NEW MERS CORONAVIRUS VACCINE CLINICAL TRIAL STARTS IN Vaccitech’s partner, the University of Oxford’s Jenner Institute, in collaboration with The King Abdullah International Medical Research Centre (KAIMRC), announced that it has started a Phase I clinical trial in the Kingdom of Saudi Arabia (KSA) for a vaccine against Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Vaccitech retains commercial rights to this vaccine. The trial TECHNOLOGY - VACCITECH A clinical stage company utilizing a world leading T cell-inducing platform. The patented Vaccitech adenovirus vectors are known as chimpanzee adenovirus Oxford 1 and 2 (ChAdOx1 and ChAdOx2), and are in the group E simian adenovirus family, similar to the widely-studied chimpanzee adenovirus 63. These viruses have been engineered to be NEWSROOM - VACCITECH Mar 17, 2021. Oxford, UK - Vaccitech Ltd (“Vaccitech”), a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious diseases and cancer, today announces thatit has
ABOUT - VACCITECH
Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologous prime-boost platform comprises Chimpanzee Adenovirus Oxford (ChAdOx) and Modified Vaccinia Ankara (MVA), two non-replicating viral vectors which safely mimic PIPELINE - VACCITECH Vaccitech is developing VTP-500, its prophylactic vaccine product candidate, to prevent infection and subsequent disease caused by MERS-CoV. VTP-500 is based on the use of one or two doses of ChAdOx1 encoding the spike glycoprotein of MERS-CoV and was developed at the University of Oxford. Development status.PARTNERSHIPS
Our Partners. Vaccitech has established partnerships with leading institutions across the globe to support the development of its programs. Hover over the logos below for more information. Vaccitech has entered into non-exclusive licensure of its T cell induction platform in oncology, and is committed to advancing the use of itsplatform for
VACCITECH AND OXFORD UNIVERSITY ANNOUNCE PROMISING PHASE Oxford, UK – Vaccitech Limited and the University of Oxford today announce initial efficacy and safety data for ADVANCE, a Phase 2a study testing VTP-800, an immunotherapeutic product candidate in patients with metastatic castration resistant prostate cancer (mCRPC).The study demonstrated that VTP-800 is safe and showed an encouraging efficacy trend in patients with mCRPC. UNIVERSAL INFLUENZA A VACCINE INVICTUS study –Phase 2b study, VTP-100 co-administered in >65 • Hypothesis: Inducing T-cells will reduce viral shedding attributable to Influenza A and will reduce the number and severity of symptoms during an influenza season • Participants: Age >65 randomized 1:1 to QIV (Fluzone) + VTP100, or QIV + placebo • Plan: Two year study in UK to enrol 2,000 participantsCMC PROJECT MANAGER
2 CMC Project Manager Essential knowledge, experience and skills: • BSc or equivalent experience in science/technical. • Minimum 3 years’ experience of biologics, VACCITECH - CREATING WAYS TO TREAT AND PREVENT DISEASE.ABOUTTECHNOLOGYPIPELINEPARTNERSHIPSNEWS & UPDATESCAREERS We discover and develop immunotherapeutics products and vaccines for the treatment and prevention of infectious diseases and cancer. Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centresin the world. 5.
INVESTORS | VACCITECH Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centres in the world. Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologousprime-boost
OUR TEAM - VACCITECH Dr. Margaret (Meg) Marshall has more than 25 years of experience in the preclinical and clinical development of cell and antibody-based immunotherapies for infectious diseases and cancer, including vaccines, checkpoint inhibitors, and cell therapies such as CAR T cells. Prior to joining Vaccitech, she held directorship positions inthe clinical
VACCITECH RECEIVES £6M INVESTMENT FROM GENEMATRIX AND Vaccitech will progress existing vaccine programs and the collaboration will combine Vaccitech’s know-how with GeneMatrix’s clinical network. Oxford, UK and Seoul, South Korea Vaccitech, a clinical stage developer of a universal flu vaccine and other vaccine-related products, has received a joint £6m investment from a Korean biotech company, GeneMatrix, and venture capital and private VACCITECH UNIVERSAL FLU VACCINE PASSES PHASE 2B CLINICAL Vaccitech initiates Phase 2b clinical studies for its MVA-NP+M1 universal flu vaccine (VTP-100) in Belgium and completes recruitment in Australia Oxford, United Kingdom – 5 June 2019: Vaccitech Ltd. announces that it has administered its pandemic universal influenza A vaccine MVA-NP+M1 (VTP-100) to the first participants in a Flu 010 study – a Phase 2b, VACANCY: SENIOR SCIENTIST Vacancy: Senior Scientist – Process Development. Aug 24, 2020. Due to exciting expansion across the company, with offices in the UK (headquarters), USA, Italy and Australia, Vaccitech is now recruiting for a Senior Scientist – Process Development, which is VACCITECH AND OXFORD UNIVERSITY ANNOUNCE PROMISING PHASE Oxford, UK – Vaccitech Limited and the University of Oxford today announce initial efficacy and safety data for ADVANCE, a Phase 2a study testing VTP-800, an immunotherapeutic product candidate in patients with metastatic castration resistant prostate cancer (mCRPC). The study demonstrated that VTP-800 is safe and showed an encouraging efficacy trend in patients with VACCITECH APPOINTS BILL ENRIGHT AS NEW CHIEF EXECUTIVE Vaccitech, a clinical-stage T cell immunotherapy company developing products to treat and prevent cancer and infectious diseases (ID), announced today that it has appointed Bill Enright as its new Chief Executive Officer (CEO). Mr. Enright succeeds Tom Evans, MD who will make a planned transition to the role of Vaccitech’s Chief Scientific Officer. Bill joins PHASE 1 DATA ON THE VACCITECH PROSTATE CANCER VACCINE Vaccitech Limited, a UK company developing products to treat and prevent multiple infectious diseases and cancer, today announced the presentation of Phase 1 prostate cancer vaccine data at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. In this “window” study, funded by the European Commission FP7 programme, 40 patients with low or INDUCING T CELLS TO TREAT AND PREVENT DISEASE AZ-OUI licensed COVID program(1) may provide us with revenue beginning as early as H2 2021 Clinical data in both therapeutic (prostate cancer) and prophylactic (COVID, MERS) programs supports further development with our platform Application of the platform for pandemic vaccine programs provides potential for response to ongoing emerginginfections
VACCITECH - CREATING WAYS TO TREAT AND PREVENT DISEASE.ABOUTTECHNOLOGYPIPELINEPARTNERSHIPSNEWS & UPDATESCAREERS We discover and develop immunotherapeutics products and vaccines for the treatment and prevention of infectious diseases and cancer. Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centresin the world. 5.
INVESTORS | VACCITECH Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centres in the world. Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologousprime-boost
OUR TEAM - VACCITECH Dr. Margaret (Meg) Marshall has more than 25 years of experience in the preclinical and clinical development of cell and antibody-based immunotherapies for infectious diseases and cancer, including vaccines, checkpoint inhibitors, and cell therapies such as CAR T cells. Prior to joining Vaccitech, she held directorship positions inthe clinical
VACCITECH RECEIVES £6M INVESTMENT FROM GENEMATRIX AND Vaccitech will progress existing vaccine programs and the collaboration will combine Vaccitech’s know-how with GeneMatrix’s clinical network. Oxford, UK and Seoul, South Korea Vaccitech, a clinical stage developer of a universal flu vaccine and other vaccine-related products, has received a joint £6m investment from a Korean biotech company, GeneMatrix, and venture capital and private VACCITECH UNIVERSAL FLU VACCINE PASSES PHASE 2B CLINICAL Vaccitech initiates Phase 2b clinical studies for its MVA-NP+M1 universal flu vaccine (VTP-100) in Belgium and completes recruitment in Australia Oxford, United Kingdom – 5 June 2019: Vaccitech Ltd. announces that it has administered its pandemic universal influenza A vaccine MVA-NP+M1 (VTP-100) to the first participants in a Flu 010 study – a Phase 2b, VACANCY: SENIOR SCIENTIST Vacancy: Senior Scientist – Process Development. Aug 24, 2020. Due to exciting expansion across the company, with offices in the UK (headquarters), USA, Italy and Australia, Vaccitech is now recruiting for a Senior Scientist – Process Development, which is VACCITECH AND OXFORD UNIVERSITY ANNOUNCE PROMISING PHASE Oxford, UK – Vaccitech Limited and the University of Oxford today announce initial efficacy and safety data for ADVANCE, a Phase 2a study testing VTP-800, an immunotherapeutic product candidate in patients with metastatic castration resistant prostate cancer (mCRPC). The study demonstrated that VTP-800 is safe and showed an encouraging efficacy trend in patients with VACCITECH APPOINTS BILL ENRIGHT AS NEW CHIEF EXECUTIVE Vaccitech, a clinical-stage T cell immunotherapy company developing products to treat and prevent cancer and infectious diseases (ID), announced today that it has appointed Bill Enright as its new Chief Executive Officer (CEO). Mr. Enright succeeds Tom Evans, MD who will make a planned transition to the role of Vaccitech’s Chief Scientific Officer. Bill joins PHASE 1 DATA ON THE VACCITECH PROSTATE CANCER VACCINE Vaccitech Limited, a UK company developing products to treat and prevent multiple infectious diseases and cancer, today announced the presentation of Phase 1 prostate cancer vaccine data at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. In this “window” study, funded by the European Commission FP7 programme, 40 patients with low or INDUCING T CELLS TO TREAT AND PREVENT DISEASE AZ-OUI licensed COVID program(1) may provide us with revenue beginning as early as H2 2021 Clinical data in both therapeutic (prostate cancer) and prophylactic (COVID, MERS) programs supports further development with our platform Application of the platform for pandemic vaccine programs provides potential for response to ongoing emerginginfections
OXFORD UNIVERSITY BREAKTHROUGH ON GLOBAL COVID-19 VACCINE Oxford, UK – Vaccitech’s scientific founders at University of Oxford announce positive high-level results from an interim analysis of clinical trials of AZD1222 in the UK and Brazil. Phase 3 interim analysis including 131 Covid-19 cases indicates that the vaccine is 70.4% effective when combining data from two dosing regimens In thetwo different dose
NEW MERS CORONAVIRUS VACCINE CLINICAL TRIAL STARTS IN Vaccitech’s partner, the University of Oxford’s Jenner Institute, in collaboration with The King Abdullah International Medical Research Centre (KAIMRC), announced that it has started a Phase I clinical trial in the Kingdom of Saudi Arabia (KSA) for a vaccine against Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Vaccitech retains commercial rights to this vaccine. The trial TECHNOLOGY - VACCITECH A clinical stage company utilizing a world leading T cell-inducing platform. The patented Vaccitech adenovirus vectors are known as chimpanzee adenovirus Oxford 1 and 2 (ChAdOx1 and ChAdOx2), and are in the group E simian adenovirus family, similar to the widely-studied chimpanzee adenovirus 63. These viruses have been engineered to be NEWSROOM - VACCITECH Mar 17, 2021. Oxford, UK - Vaccitech Ltd (“Vaccitech”), a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious diseases and cancer, today announces thatit has
ABOUT - VACCITECH
Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologous prime-boost platform comprises Chimpanzee Adenovirus Oxford (ChAdOx) and Modified Vaccinia Ankara (MVA), two non-replicating viral vectors which safely mimic PIPELINE - VACCITECH Vaccitech is developing VTP-500, its prophylactic vaccine product candidate, to prevent infection and subsequent disease caused by MERS-CoV. VTP-500 is based on the use of one or two doses of ChAdOx1 encoding the spike glycoprotein of MERS-CoV and was developed at the University of Oxford. Development status.PARTNERSHIPS
Our Partners. Vaccitech has established partnerships with leading institutions across the globe to support the development of its programs. Hover over the logos below for more information. Vaccitech has entered into non-exclusive licensure of its T cell induction platform in oncology, and is committed to advancing the use of itsplatform for
VACCITECH AND OXFORD UNIVERSITY ANNOUNCE PROMISING PHASE Oxford, UK – Vaccitech Limited and the University of Oxford today announce initial efficacy and safety data for ADVANCE, a Phase 2a study testing VTP-800, an immunotherapeutic product candidate in patients with metastatic castration resistant prostate cancer (mCRPC).The study demonstrated that VTP-800 is safe and showed an encouraging efficacy trend in patients with mCRPC. UNIVERSAL INFLUENZA A VACCINE INVICTUS study –Phase 2b study, VTP-100 co-administered in >65 • Hypothesis: Inducing T-cells will reduce viral shedding attributable to Influenza A and will reduce the number and severity of symptoms during an influenza season • Participants: Age >65 randomized 1:1 to QIV (Fluzone) + VTP100, or QIV + placebo • Plan: Two year study in UK to enrol 2,000 participantsCMC PROJECT MANAGER
2 CMC Project Manager Essential knowledge, experience and skills: • BSc or equivalent experience in science/technical. • Minimum 3 years’ experience of biologics, VACCITECH - CREATING WAYS TO TREAT AND PREVENT DISEASE.ABOUTTECHNOLOGYPIPELINEPARTNERSHIPSNEWS & UPDATESCAREERS We discover and develop immunotherapeutics products and vaccines for the treatment and prevention of infectious diseases and cancer. Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centresin the world. 5.
OXFORD UNIVERSITY BREAKTHROUGH ON GLOBAL COVID-19 VACCINE Oxford, UK – Vaccitech’s scientific founders at University of Oxford announce positive high-level results from an interim analysis of clinical trials of AZD1222 in the UK and Brazil. Phase 3 interim analysis including 131 Covid-19 cases indicates that the vaccine is 70.4% effective when combining data from two dosing regimens In thetwo different dose
INVESTORS | VACCITECH Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centres in the world. Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologousprime-boost
NEW MERS CORONAVIRUS VACCINE CLINICAL TRIAL STARTS IN Vaccitech’s partner, the University of Oxford’s Jenner Institute, in collaboration with The King Abdullah International Medical Research Centre (KAIMRC), announced that it has started a Phase I clinical trial in the Kingdom of Saudi Arabia (KSA) for a vaccine against Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Vaccitech retains commercial rights to this vaccine. The trial PIPELINE - VACCITECH Vaccitech is developing VTP-500, its prophylactic vaccine product candidate, to prevent infection and subsequent disease caused by MERS-CoV. VTP-500 is based on the use of one or two doses of ChAdOx1 encoding the spike glycoprotein of MERS-CoV and was developed at the University of Oxford. Development status. OUR TEAM - VACCITECH Dr. Margaret (Meg) Marshall has more than 25 years of experience in the preclinical and clinical development of cell and antibody-based immunotherapies for infectious diseases and cancer, including vaccines, checkpoint inhibitors, and cell therapies such as CAR T cells. Prior to joining Vaccitech, she held directorship positions inthe clinical
VACCITECH RECEIVES £6M INVESTMENT FROM GENEMATRIX AND Vaccitech will progress existing vaccine programs and the collaboration will combine Vaccitech’s know-how with GeneMatrix’s clinical network. Oxford, UK and Seoul, South Korea Vaccitech, a clinical stage developer of a universal flu vaccine and other vaccine-related products, has received a joint £6m investment from a Korean biotech company, GeneMatrix, and venture capital and private VACANCY: SENIOR SCIENTIST Vacancy: Senior Scientist – Process Development. Aug 24, 2020. Due to exciting expansion across the company, with offices in the UK (headquarters), USA, Italy and Australia, Vaccitech is now recruiting for a Senior Scientist – Process Development, which is PHASE 1 DATA ON THE VACCITECH PROSTATE CANCER VACCINE Vaccitech Limited, a UK company developing products to treat and prevent multiple infectious diseases and cancer, today announced the presentation of Phase 1 prostate cancer vaccine data at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. In this “window” study, funded by the European Commission FP7 programme, 40 patients with low orCMC PROJECT MANAGER
2 CMC Project Manager Essential knowledge, experience and skills: • BSc or equivalent experience in science/technical. • Minimum 3 years’ experience of biologics, VACCITECH - CREATING WAYS TO TREAT AND PREVENT DISEASE.ABOUTTECHNOLOGYPIPELINEPARTNERSHIPSNEWS & UPDATESCAREERS We discover and develop immunotherapeutics products and vaccines for the treatment and prevention of infectious diseases and cancer. Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centresin the world. 5.
OXFORD UNIVERSITY BREAKTHROUGH ON GLOBAL COVID-19 VACCINE Oxford, UK – Vaccitech’s scientific founders at University of Oxford announce positive high-level results from an interim analysis of clinical trials of AZD1222 in the UK and Brazil. Phase 3 interim analysis including 131 Covid-19 cases indicates that the vaccine is 70.4% effective when combining data from two dosing regimens In thetwo different dose
INVESTORS | VACCITECH Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centres in the world. Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologousprime-boost
NEW MERS CORONAVIRUS VACCINE CLINICAL TRIAL STARTS IN Vaccitech’s partner, the University of Oxford’s Jenner Institute, in collaboration with The King Abdullah International Medical Research Centre (KAIMRC), announced that it has started a Phase I clinical trial in the Kingdom of Saudi Arabia (KSA) for a vaccine against Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Vaccitech retains commercial rights to this vaccine. The trial PIPELINE - VACCITECH Vaccitech is developing VTP-500, its prophylactic vaccine product candidate, to prevent infection and subsequent disease caused by MERS-CoV. VTP-500 is based on the use of one or two doses of ChAdOx1 encoding the spike glycoprotein of MERS-CoV and was developed at the University of Oxford. Development status. OUR TEAM - VACCITECH Dr. Margaret (Meg) Marshall has more than 25 years of experience in the preclinical and clinical development of cell and antibody-based immunotherapies for infectious diseases and cancer, including vaccines, checkpoint inhibitors, and cell therapies such as CAR T cells. Prior to joining Vaccitech, she held directorship positions inthe clinical
VACCITECH RECEIVES £6M INVESTMENT FROM GENEMATRIX AND Vaccitech will progress existing vaccine programs and the collaboration will combine Vaccitech’s know-how with GeneMatrix’s clinical network. Oxford, UK and Seoul, South Korea Vaccitech, a clinical stage developer of a universal flu vaccine and other vaccine-related products, has received a joint £6m investment from a Korean biotech company, GeneMatrix, and venture capital and private VACANCY: SENIOR SCIENTIST Vacancy: Senior Scientist – Process Development. Aug 24, 2020. Due to exciting expansion across the company, with offices in the UK (headquarters), USA, Italy and Australia, Vaccitech is now recruiting for a Senior Scientist – Process Development, which is PHASE 1 DATA ON THE VACCITECH PROSTATE CANCER VACCINE Vaccitech Limited, a UK company developing products to treat and prevent multiple infectious diseases and cancer, today announced the presentation of Phase 1 prostate cancer vaccine data at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. In this “window” study, funded by the European Commission FP7 programme, 40 patients with low orCMC PROJECT MANAGER
2 CMC Project Manager Essential knowledge, experience and skills: • BSc or equivalent experience in science/technical. • Minimum 3 years’ experience of biologics, TECHNOLOGY - VACCITECH A clinical stage company utilizing a world leading T cell-inducing platform. The patented Vaccitech adenovirus vectors are known as chimpanzee adenovirus Oxford 1 and 2 (ChAdOx1 and ChAdOx2), and are in the group E simian adenovirus family, similar to the widely-studied chimpanzee adenovirus 63. These viruses have been engineered to beABOUT - VACCITECH
Vaccitech is a clinical stage T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancer. The company’s proprietary heterologous prime-boost platform comprises Chimpanzee Adenovirus Oxford (ChAdOx) and Modified Vaccinia Ankara (MVA), two non-replicating viral vectors which safely mimic NEWSROOM - VACCITECH Mar 17, 2021. Oxford, UK - Vaccitech Ltd (“Vaccitech”), a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious diseases and cancer, today announces thatit has
NEW MERS CORONAVIRUS VACCINE CLINICAL TRIAL STARTS IN Vaccitech’s partner, the University of Oxford’s Jenner Institute, in collaboration with The King Abdullah International Medical Research Centre (KAIMRC), announced that it has started a Phase I clinical trial in the Kingdom of Saudi Arabia (KSA) for a vaccine against Middle East Respiratory Syndrome Coronavirus (MERS-CoV).Vaccitech retains commercial rights to this vaccine.CONTACT - VACCITECH
Thank you for your interest in Vaccitech and your enquiry about our ongoing clinical trials. Please note that all information on ongoing or closed clinical trials run by Vaccitech, including eligibility criteria and contact information are posted on the https://clinicaltrials.gov/ website.. Chronic Hepatitis B: Vaccitech is conducting a Phase 1 study to determine the safety and tolerabilityof
PHASE 2 CLINICAL RESULTS FOR VACCITECH’S UNIVERSAL Oxford, UK – Vaccitech Ltd, a clinical-stage T cell immunotherapy company developing products to treat cancer and chronic infectious diseases, announces top line Phase 2 data and plans for its recombinant Modified Vaccinia Ankara (MVA) Universal Influenza A prophylactic vaccine, VTP-100, clinically developed to address both pandemic and seasonal influenza. A Data Monitoring Committee VACCITECH UNIVERSAL FLU VACCINE PASSES PHASE 2B CLINICAL Vaccitech initiates Phase 2b clinical studies for its MVA-NP+M1 universal flu vaccine (VTP-100) in Belgium and completes recruitment in Australia Oxford, United Kingdom – 5 June 2019: Vaccitech Ltd. announces that it has administered its pandemic universal influenza A vaccine MVA-NP+M1 (VTP-100) to the first participants in a Flu 010 study – a Phase 2b,PARTNERSHIPS
Our Partners. Vaccitech has established partnerships with leading institutions across the globe to support the development of its programs. Hover over the logos below for more information. Vaccitech has entered into non-exclusive licensure of its T cell induction platform in oncology, and is committed to advancing the use of itsplatform for
PHASE 1 DATA ON THE VACCITECH PROSTATE CANCER VACCINE Vaccitech Limited, a UK company developing products to treat and prevent multiple infectious diseases and cancer, today announced the presentation of Phase 1 prostate cancer vaccine data at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago. In this “window” study, funded by the European Commission FP7 programme, 40 patients with low or INDUCING T CELLS TO TREAT AND PREVENT DISEASE AZ-OUI licensed COVID program(1) may provide us with revenue beginning as early as H2 2021 Clinical data in both therapeutic (prostate cancer) and prophylactic (COVID, MERS) programs supports further development with our platform Application of the platform for pandemic vaccine programs provides potential for response to ongoing emerginginfections
* Home
* Our Team
* About
* Technology
* Pipeline
* Partnerships
* Newsroom
* Events
* Investors
* News & Events
* Stock Information
* Corporate Governance* Financials
* Investor Resources* Careers
* Contact
Select Page
* Home
* Our Team
* About
* Technology
* Pipeline
* Partnerships
* Newsroom
* Events
* Investors
* News & Events
* Stock Information
* Corporate Governance* Financials
* Investor Resources* Careers
* Contact
VACCITECH INDUCES T CELLS AND ANTIBODIES TO TREAT AND PREVENT DISEASELearn More
3
Learn more Vaccitech induces T cells and antibodies to treatand
prevent disease Learn more Human Papillomavirus causes nearly all cases of cervical cancer Learn more Chronic hepatitis B affects an estimated 257million
people worldwide Learn more SARS-CoV-2 has caused a worldwide pandemic of respiratory illness, commonly referredto as COVID-19
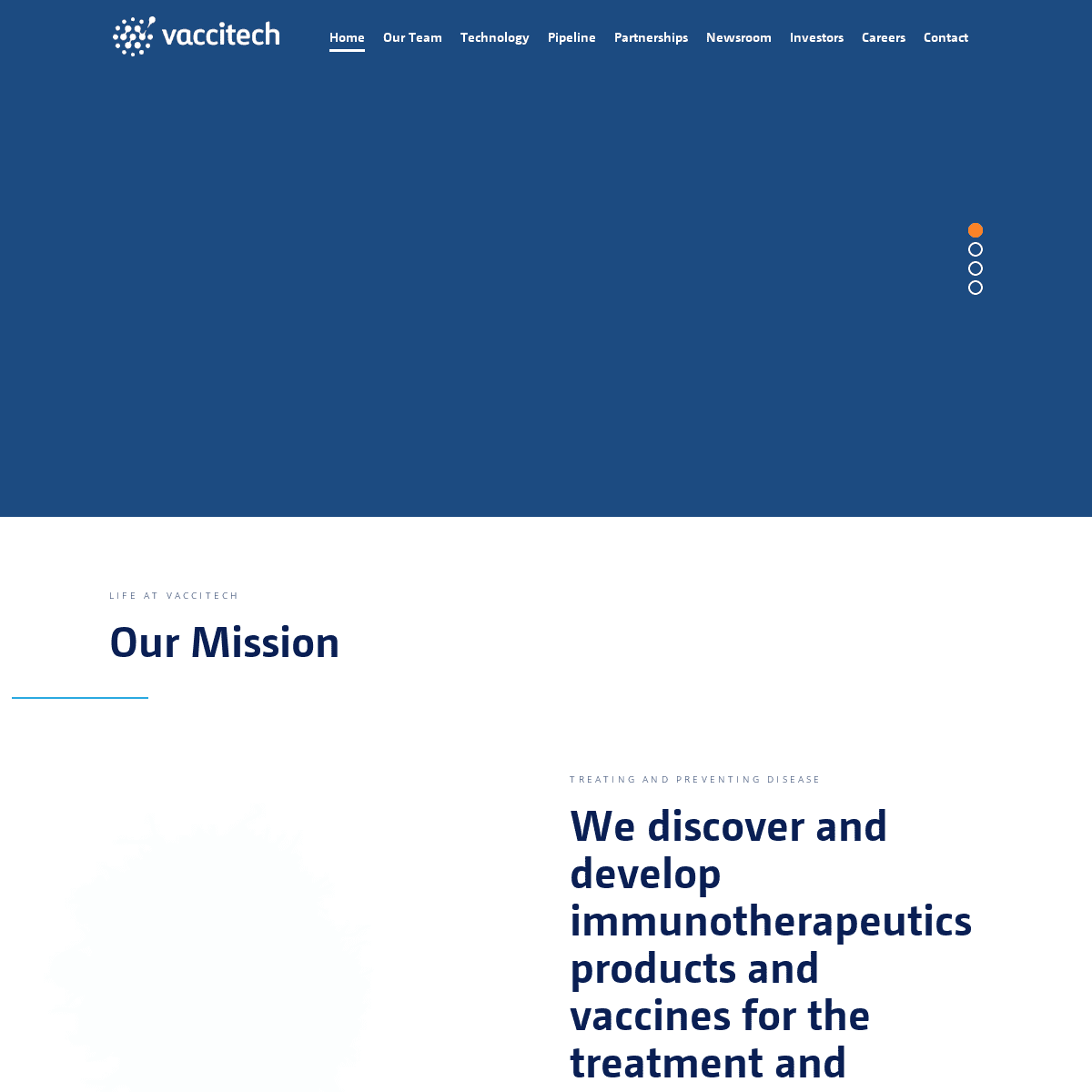
life at vaccitech
OUR MISSION
Image: NIAID
Treating and Preventing Disease WE DISCOVER AND DEVELOP IMMUNOTHERAPEUTICS PRODUCTS AND VACCINES FOR THE TREATMENT AND PREVENTION OF INFECTIOUS DISEASES AND CANCER Vaccitech is a spin-out company from the University of Oxford’s Jenner Institute, one of the most prestigious vaccine research centresin the world.
5
ABOUT VACCITECH
5
OUR TECHNOLOGY
5
MEET OUR TEAM
Vaccitech Announces Pricing of Initial Public Offering Oxford, UK - Vaccitech plc (“Vaccitech”) (Nasdaq: VACC), a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious... Vaccitech Appoints Joseph Scheeren, PharmD, to its Board of Directors Oxford, UK - Vaccitech Ltd (“Vaccitech”), a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious diseases and... Vaccitech Doses First Patient in HPV001, a Phase 1/2 Clinical Trial of VTP-200 Immunotherapeutic for High-risk Persistent HPV Infection Oxford, UK - Vaccitech Ltd, a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious diseasesand cancer,...
Vaccitech Completes $168 Million Series B Financing to Advance Three Clinical Programs Through Phase 2 Results Oxford, UK - Vaccitech Ltd (“Vaccitech”), a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious diseases and... Vaccitech Appoints Two Independent Board Directors Oxford, UK - Vaccitech Ltd., a clinical-stage biopharmaceutical company engaged in the discovery and development of novel immunotherapeutics and vaccines for the treatment and prevention of infectious diseases and cancer,...OUR APPROACH
TEACHING THE IMMUNE SYSTEM TO CONFRONT DEADLY INFECTIONS Traditional vaccine technology creates a pronounced antibody response to the infectious agent. Vaccitech’s platform is also able to induce potent and durable T cell responses from the cellular arm of theimmune system.
5
OUR TECHNOLOGY
5
OUR PIPELINE
5
OUR CAREERS
VACCITECH PLC
The Schrödinger BuildingHeatley Road
The Oxford Science ParkOxford
OX4 4GE
T: +44 (0)1865 818808* Home
* About
* Sitemap
* Privacy Policy
* Privacy Notice
* Careers
* Contact
Vaccitech is a T cell immunotherapy and vaccine company developing products to treat and prevent infectious disease and cancerDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0