Are you over 18 and want to see adult content?
More Annotations
A complete backup of sharedlivesplus.org.uk
Are you over 18 and want to see adult content?

A complete backup of bostondigital.com
Are you over 18 and want to see adult content?

A complete backup of familysavings.com
Are you over 18 and want to see adult content?

A complete backup of guardian-series.co.uk
Are you over 18 and want to see adult content?
A complete backup of therugseller.co.uk
Are you over 18 and want to see adult content?

A complete backup of dharmacrafts.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of https://paintsquare.com
Are you over 18 and want to see adult content?
A complete backup of https://arwmisure.it
Are you over 18 and want to see adult content?

A complete backup of https://eharmony.co.uk
Are you over 18 and want to see adult content?
A complete backup of https://theshopsatlacantera.com
Are you over 18 and want to see adult content?

A complete backup of https://ovahealth.in
Are you over 18 and want to see adult content?
A complete backup of https://qrcargo.com
Are you over 18 and want to see adult content?

A complete backup of https://bootyalbum.com
Are you over 18 and want to see adult content?
A complete backup of https://ocpinfo.com
Are you over 18 and want to see adult content?

A complete backup of https://hoster.kz
Are you over 18 and want to see adult content?

A complete backup of https://monnaiedeparis.fr
Are you over 18 and want to see adult content?

A complete backup of https://veskobel.by
Are you over 18 and want to see adult content?
Text
NEWS - STARPHARMA
Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More STARPHARMA | IN THE MEDIA Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More VIVAGEL® - STARPHARMA VivaGel® VivaGel ® is an innovative technology being applied to a range of sexual/women’s health products by Starpharma and its partners, including:. VivaGel® condom; VivaGel® for treatment & prevention of bacterial vaginosis (BV) VivaGel® for the prevention of VIRALEZE™ ANTIVIRAL NASAL SPRAY What is VIRALEZE™ Antiviral Nasal Spray? VIRALEZE™ is an antiviral nasal spray that contains astodrimer sodium (SPL7013), which has been shown in laboratory studies to inactivate a broad spectrum of respiratory viruses, including >99.9% of coronavirusVIVAGEL® CONDOM
VivaGel® Condom . VivaGel ® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria.. The physical barrier of the condom provides primary protection against sexually transmitted infections (STIs). The condom is lubricated with VivaGel ® lubricant. VivaGel ® is an antiviral agent proven, in laboratory studies only, to inactivate HIV, herpes simplex virus (HSV) and human VIVAGEL® AVAILABILITY VivaGel® Availability. VivaGel ® BV . VivaGel ® BV is available for sale under the brand names Betafem ® BV Gel (UK), Betadine BV TM (Europe), Betadine TM BV Gel (Asia) and Fleurstat BVgel (Australia & New Zealand).. Regulatory processes are underway to enable marketing in the US and other geographic regions. VivaGel ® condom . The VivaGel ® condom has been launched in Japan underDEP® DOCETAXEL
PARTNERED-DEP® PRODUCTS AZD0466: is a highly optimised nanomedicine formulation of Astra Zeneca cancer drug AZD4320; is currently in a phase 1 clinical trial (FDA IND) is a Bcl2/xL inhibitor which has improved preclinical efficacy and therapeutic margin STARPHARMA SIGNS NEW DEP® PARTNERSHIP WITH CHASE SUN 2 . DEP® ®- Dendrimer Enhanced Product : Starpharma’s DEP® drug delivery platform has demonstrated reproducible preclinical benefits across multiple internal and partnered DEP® programs, including improved efficacy, safety, and survival. Starpharma has three internal DEP® products – DEP ® docetaxel, ®DEP cabazitaxel and DEP irinotecan - in clinical development in patients with solid HOME - STARPHARMAABOUT USTECHNOLOGYVIVAGELDEP® DRUG DELIVERYNEWSQUARTERLY CASHFLOW REPORT Starpharma Holdings Limited (ASX:SPL, OTCQX:SPHRY) is a global biopharmaceutical company and a world leader in the development of new pharmaceutical and medical products based on proprietary polymers called dendrimers, with programs for COVID-19, DEP ® drug delivery and VivaGel ®.Starpharma has developed VIRALEZE™, an antiviral nasal spray for COVID-19, which is complementary to vaccinesNEWS - STARPHARMA
Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More STARPHARMA | IN THE MEDIA Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More VIVAGEL® - STARPHARMA VivaGel® VivaGel ® is an innovative technology being applied to a range of sexual/women’s health products by Starpharma and its partners, including:. VivaGel® condom; VivaGel® for treatment & prevention of bacterial vaginosis (BV) VivaGel® for the prevention of VIRALEZE™ ANTIVIRAL NASAL SPRAY What is VIRALEZE™ Antiviral Nasal Spray? VIRALEZE™ is an antiviral nasal spray that contains astodrimer sodium (SPL7013), which has been shown in laboratory studies to inactivate a broad spectrum of respiratory viruses, including >99.9% of coronavirusVIVAGEL® CONDOM
VivaGel® Condom . VivaGel ® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria.. The physical barrier of the condom provides primary protection against sexually transmitted infections (STIs). The condom is lubricated with VivaGel ® lubricant. VivaGel ® is an antiviral agent proven, in laboratory studies only, to inactivate HIV, herpes simplex virus (HSV) and human VIVAGEL® AVAILABILITY VivaGel® Availability. VivaGel ® BV . VivaGel ® BV is available for sale under the brand names Betafem ® BV Gel (UK), Betadine BV TM (Europe), Betadine TM BV Gel (Asia) and Fleurstat BVgel (Australia & New Zealand).. Regulatory processes are underway to enable marketing in the US and other geographic regions. VivaGel ® condom . The VivaGel ® condom has been launched in Japan underDEP® DOCETAXEL
PARTNERED-DEP® PRODUCTS AZD0466: is a highly optimised nanomedicine formulation of Astra Zeneca cancer drug AZD4320; is currently in a phase 1 clinical trial (FDA IND) is a Bcl2/xL inhibitor which has improved preclinical efficacy and therapeutic margin STARPHARMA SIGNS NEW DEP® PARTNERSHIP WITH CHASE SUN 2 . DEP® ®- Dendrimer Enhanced Product : Starpharma’s DEP® drug delivery platform has demonstrated reproducible preclinical benefits across multiple internal and partnered DEP® programs, including improved efficacy, safety, and survival. Starpharma has three internal DEP® products – DEP ® docetaxel, ®DEP cabazitaxel and DEP irinotecan - in clinical development in patients with solidNEWS - STARPHARMA
VIRALEZE SPL7013 highly active in UK variant of coronavirus Jun 1st, 2021 New antiviral testing at the Scripps Research Institute has confirmed SPL7013 (VIRALEZE ™ active) has potent antiviral activity against the UK (B.1.1.7) SARS-CoV-2 coronavirus mutant/variantstrain.. Read More
MANAGEMENT - STARPHARMA Jackie Fairley, BSc, BVSc (Hons), MBA, GAICD, FTSE Chief Executive Officer. Dr Jackie Fairley has more than 30 years of operational experience in the pharmaceutical and biotechnology industries working in business development and senior management roles with companies including CSL and Faulding/Hospira (now part of Pfizer).CLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®)) CONTACT US - STARPHARMA 4-6 Southampton Crescent Abbotsford VICTORIA 3067 AUSTRALIA. T: +61 3 8532 2700 F: +61 3 9510 5955. View Location in Google Maps : Postal Address: PO Box 2022 STARPHARMA | BOARD OF DIRECTORS Board of Directors Rob B Thomas AO, BEc, MSAA, SF Fin, FAICD, FRSN. Independent non-executive director (appointed 4 December 2013) Chairman from 13 June 2014. Mr Thomas has a strong background in financial services and capital markets and is a non-executive director of several Australian listed companies.DEP® DOCETAXEL
DEP® docetaxel Summary and Commercial Opportunity. Starpharma is developing an enhanced form of docetaxel known as DEP ® docetaxel (dendrimer-docetaxel).. Docetaxel is a leading chemotherapy drug used to treat a wide range of solid tumours including breast, lung andprostate.
SHAREHOLDER NEWSLETTER Apr 28, 2021. Shareholder Newsletter. The Starpharma Holdings Limited (ASX: SPL, OTCQX: SPHRY) Shareholder Update – April 2021 is now available to view online. This contains certain forward-lookingstatements.
DEP® IRINOTECAN
DEP® irinotecan. DEP ® irinotecan is a novel, patented nanoparticle formulation of SN-38, the active metabolite of irinotecan, delivered using Starpharma’s proprietary DEP ® technology. DEP ® irinotecan has shown a number of benefits compared to the original form of irinotecan, including significant improvements in anti-cancer efficacy and improved survival in multiple human cancer models. VIVAGEL® CLINICAL TRIALS Study No. Summary Schedule No. of Participants; SPL 7013-001. Safety, tolerability and PK: Vaginal administration, once daily for 7 days: 37SPL 7013-002
STARPHARMA | VIRALEZE™ ANTIVIRAL NASAL SPRAY REGISTERED IN Feb 23, 2021. VIRALEZE™ antiviral nasal spray registered in Europe . VIRALEZE™ has been successfully registered, to allow for its sale in Europe, including in HOME - STARPHARMAABOUT USTECHNOLOGYVIVAGELDEP® DRUG DELIVERYNEWSQUARTERLY CASHFLOW REPORT Starpharma Holdings Limited (ASX:SPL, OTCQX:SPHRY) is a global biopharmaceutical company and a world leader in the development of new pharmaceutical and medical products based on proprietary polymers called dendrimers, with programs for COVID-19, DEP ® drug delivery and VivaGel ®.Starpharma has developed VIRALEZE™, an antiviral nasal spray for COVID-19, which is complementary to vaccinesNEWS - STARPHARMA
Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More STARPHARMA | IN THE MEDIA Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More VIRALEZE™ ANTIVIRAL NASAL SPRAY VIRALEZE™ is an antiviral nasal spray that contains astodrimer sodium (SPL7013), which has been shown in laboratory studies to inactivate a broad spectrum of respiratory viruses, including >99.9% of coronavirus SARS-CoV-2, the virus that causes COVID-19.^. ^Paull, J.R.A. et al. Virucidal and antiviral activity of astodrimer sodiumagainst
VIVAGEL® CONDOM
VivaGel® Condom . VivaGel ® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria.. The physical barrier of the condom provides primary protection against sexually transmitted infections (STIs). The condom is lubricated with VivaGel ® lubricant. VivaGel ® is an antiviral agent proven, in laboratory studies only, to inactivate HIV, herpes simplex virus (HSV) and humanCLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®))2020 ESG REPORT
STARPHARMA ESG REPORT 2020 2 This Report . Starpharma is proud to publish its first standalone Environment, Social and Governance Report(“ESG Report”).
CHANNEL 7 EVENING NEWS FEATURES STARPHARMA'S VIRALEZE Channel 7 evening news features Starpharma's VIRALEZE. Watch Laurel Irving Channel 7 News (Melbourne) interview Dr Jackie Fairley, CEO of Starpharma about VIRALEZE™ and the need for other COVID-19 preventative tools alongside vaccines. This contains certain forward-looking statements. STARPHARMA | DEP® CABAZITAXEL PROGRESSES TO PHASE 2 ON DEP® cabazitaxel progresses to phase 2 on positive results. Phase 1 part of the DEP® cabazitaxel phase 1 / 2 trial has been successfully completed, with phase 2 to commence immediately. were observed in cancers not usually responsive to conventional cabazitaxel (Jevtana®), such as ovarian cancer, and at doses lower than used forJevtana®.
STARPHARMA SIGNS NEW DEP® PARTNERSHIP WITH CHASE SUN 2 . DEP® ®- Dendrimer Enhanced Product : Starpharma’s DEP® drug delivery platform has demonstrated reproducible preclinical benefits across multiple internal and partnered DEP® programs, including improved efficacy, safety, and survival. Starpharma has three internal DEP® products – DEP ® docetaxel, ®DEP cabazitaxel and DEP irinotecan - in clinical development in patients with solid HOME - STARPHARMAABOUT USTECHNOLOGYVIVAGELDEP® DRUG DELIVERYNEWSQUARTERLY CASHFLOW REPORT Starpharma Holdings Limited (ASX:SPL, OTCQX:SPHRY) is a global biopharmaceutical company and a world leader in the development of new pharmaceutical and medical products based on proprietary polymers called dendrimers, with programs for COVID-19, DEP ® drug delivery and VivaGel ®.Starpharma has developed VIRALEZE™, an antiviral nasal spray for COVID-19, which is complementary to vaccinesNEWS - STARPHARMA
Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More STARPHARMA | IN THE MEDIA Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More VIRALEZE™ ANTIVIRAL NASAL SPRAY VIRALEZE™ is an antiviral nasal spray that contains astodrimer sodium (SPL7013), which has been shown in laboratory studies to inactivate a broad spectrum of respiratory viruses, including >99.9% of coronavirus SARS-CoV-2, the virus that causes COVID-19.^. ^Paull, J.R.A. et al. Virucidal and antiviral activity of astodrimer sodiumagainst
VIVAGEL® CONDOM
VivaGel® Condom . VivaGel ® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria.. The physical barrier of the condom provides primary protection against sexually transmitted infections (STIs). The condom is lubricated with VivaGel ® lubricant. VivaGel ® is an antiviral agent proven, in laboratory studies only, to inactivate HIV, herpes simplex virus (HSV) and humanCLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®))2020 ESG REPORT
STARPHARMA ESG REPORT 2020 2 This Report . Starpharma is proud to publish its first standalone Environment, Social and Governance Report(“ESG Report”).
CHANNEL 7 EVENING NEWS FEATURES STARPHARMA'S VIRALEZE Channel 7 evening news features Starpharma's VIRALEZE. Watch Laurel Irving Channel 7 News (Melbourne) interview Dr Jackie Fairley, CEO of Starpharma about VIRALEZE™ and the need for other COVID-19 preventative tools alongside vaccines. This contains certain forward-looking statements. STARPHARMA | DEP® CABAZITAXEL PROGRESSES TO PHASE 2 ON DEP® cabazitaxel progresses to phase 2 on positive results. Phase 1 part of the DEP® cabazitaxel phase 1 / 2 trial has been successfully completed, with phase 2 to commence immediately. were observed in cancers not usually responsive to conventional cabazitaxel (Jevtana®), such as ovarian cancer, and at doses lower than used forJevtana®.
STARPHARMA SIGNS NEW DEP® PARTNERSHIP WITH CHASE SUN 2 . DEP® ®- Dendrimer Enhanced Product : Starpharma’s DEP® drug delivery platform has demonstrated reproducible preclinical benefits across multiple internal and partnered DEP® programs, including improved efficacy, safety, and survival. Starpharma has three internal DEP® products – DEP ® docetaxel, ®DEP cabazitaxel and DEP irinotecan - in clinical development in patients with solidNEWS - STARPHARMA
Starpharma (ASX:SPL) launches antiviral nasal spray in Europe. May 6th, 2021. The Market Herald reported on Starpharma's launch of VIRALEZE™ in Europe. Read the full article here. Starpharma’s (SPL) antiviral nasal spray is now available for purchase by consumers in Europe via the company’s online store. VIRALEZE is a broadspectrum
MANAGEMENT - STARPHARMA Jackie Fairley, BSc, BVSc (Hons), MBA, GAICD, FTSE Chief Executive Officer. Dr Jackie Fairley has more than 30 years of operational experience in the pharmaceutical and biotechnology industries working in business development and senior management roles with companies including CSL and Faulding/Hospira (now part of Pfizer).MEDIA - STARPHARMA
Media Contacts. For further information about Starpharma's business, please contact: Media. Grant Titmus. Sumit Media. M: +61 419 388 161. grant@sumitmedia.com.au VIVAGEL® - STARPHARMA VivaGel®. VivaGel ® is an innovative technology being applied to a range of sexual/women’s health products by Starpharma and its partners, including: VivaGel® for the prevention of sexually transmitted infections (STIs) VivaGel® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria. Several clinical studieshave successfully
CLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®)) STARPHARMA | SCRIPPS RESEARCH INSTITUTE TESTING SHOWS Jun 01, 2021. Scripps Research Institute testing shows Viraleze highly active against UK COVID strain. Markets Insider featured Viraleze and Starpharma following the announcement of SPL7013's potent antiviral activity against the UK coronavirus variant. STARPHARMA | BOARD OF DIRECTORS Board of Directors Rob B Thomas AO, BEc, MSAA, SF Fin, FAICD, FRSN. Independent non-executive director (appointed 4 December 2013) Chairman from 13 June 2014. Mr Thomas has a strong background in financial services and capital markets and is a non-executive director of several Australian listed companies. PARTNERED-DEP® PRODUCTS Partnered-DEP® products - AZD0466. Starpharma’s DEP® platform enhances the commercial and therapeutic value of a wide range of drugs, making it a highly valuable partnering technology. AZD0466: is a highly optimised nanomedicine formulation of Astra Zeneca cancer drug AZD4320. is currently in a phase 1 clinical trial (FDA IND) STARPHARMA | LLOYDS PHARMACY OFFERING FIRST-EVER COVID Mar 30, 2021. Lloyds Pharmacy offering first-ever Covid nasal spray Viraleze that 'blocks 99.9% of germs' CambridgeshireLive featured news of the VIRALEZE launch in the UK. LEADING BROKER NAMES THE ASX HEALTHCARE SHARES TO BUY IN Leading broker names the ASX healthcare shares to buy in 2021. The Motley Fool reported that Starpharma has been named by analysts at Bell Potter "one of ASX shares from several industries that they believe are best placed to have a strong 2021". HOME - STARPHARMAABOUT USTECHNOLOGYVIVAGELDEP® DRUG DELIVERYNEWSQUARTERLY CASHFLOW REPORT Starpharma Holdings Limited (ASX:SPL, OTCQX:SPHRY) is a global biopharmaceutical company and a world leader in the development of new pharmaceutical and medical products based on proprietary polymers called dendrimers, with programs for COVID-19, DEP ® drug delivery and VivaGel ®.Starpharma has developed VIRALEZE™, an antiviral nasal spray for COVID-19, which is complementary to vaccinesNEWS - STARPHARMA
Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More STARPHARMA | IN THE MEDIA Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More VIRALEZE™ ANTIVIRAL NASAL SPRAY VIRALEZE™ is an antiviral nasal spray that contains astodrimer sodium (SPL7013), which has been shown in laboratory studies to inactivate a broad spectrum of respiratory viruses, including >99.9% of coronavirus SARS-CoV-2, the virus that causes COVID-19.^. ^Paull, J.R.A. et al. Virucidal and antiviral activity of astodrimer sodiumagainst
VIVAGEL® CONDOM
VivaGel® Condom . VivaGel ® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria.. The physical barrier of the condom provides primary protection against sexually transmitted infections (STIs). The condom is lubricated with VivaGel ® lubricant. VivaGel ® is an antiviral agent proven, in laboratory studies only, to inactivate HIV, herpes simplex virus (HSV) and humanCLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®))2020 ESG REPORT
STARPHARMA ESG REPORT 2020 2 This Report . Starpharma is proud to publish its first standalone Environment, Social and Governance Report(“ESG Report”).
CHANNEL 7 EVENING NEWS FEATURES STARPHARMA'S VIRALEZE Channel 7 evening news features Starpharma's VIRALEZE. Watch Laurel Irving Channel 7 News (Melbourne) interview Dr Jackie Fairley, CEO of Starpharma about VIRALEZE™ and the need for other COVID-19 preventative tools alongside vaccines. This contains certain forward-looking statements. STARPHARMA | DEP® CABAZITAXEL PROGRESSES TO PHASE 2 ON DEP® cabazitaxel progresses to phase 2 on positive results. Phase 1 part of the DEP® cabazitaxel phase 1 / 2 trial has been successfully completed, with phase 2 to commence immediately. were observed in cancers not usually responsive to conventional cabazitaxel (Jevtana®), such as ovarian cancer, and at doses lower than used forJevtana®.
STARPHARMA SIGNS NEW DEP® PARTNERSHIP WITH CHASE SUN 2 . DEP® ®- Dendrimer Enhanced Product : Starpharma’s DEP® drug delivery platform has demonstrated reproducible preclinical benefits across multiple internal and partnered DEP® programs, including improved efficacy, safety, and survival. Starpharma has three internal DEP® products – DEP ® docetaxel, ®DEP cabazitaxel and DEP irinotecan - in clinical development in patients with solid HOME - STARPHARMAABOUT USTECHNOLOGYVIVAGELDEP® DRUG DELIVERYNEWSQUARTERLY CASHFLOW REPORT Starpharma Holdings Limited (ASX:SPL, OTCQX:SPHRY) is a global biopharmaceutical company and a world leader in the development of new pharmaceutical and medical products based on proprietary polymers called dendrimers, with programs for COVID-19, DEP ® drug delivery and VivaGel ®.Starpharma has developed VIRALEZE™, an antiviral nasal spray for COVID-19, which is complementary to vaccinesNEWS - STARPHARMA
Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More STARPHARMA | IN THE MEDIA Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More VIRALEZE™ ANTIVIRAL NASAL SPRAY VIRALEZE™ is an antiviral nasal spray that contains astodrimer sodium (SPL7013), which has been shown in laboratory studies to inactivate a broad spectrum of respiratory viruses, including >99.9% of coronavirus SARS-CoV-2, the virus that causes COVID-19.^. ^Paull, J.R.A. et al. Virucidal and antiviral activity of astodrimer sodiumagainst
VIVAGEL® CONDOM
VivaGel® Condom . VivaGel ® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria.. The physical barrier of the condom provides primary protection against sexually transmitted infections (STIs). The condom is lubricated with VivaGel ® lubricant. VivaGel ® is an antiviral agent proven, in laboratory studies only, to inactivate HIV, herpes simplex virus (HSV) and humanCLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®))2020 ESG REPORT
STARPHARMA ESG REPORT 2020 2 This Report . Starpharma is proud to publish its first standalone Environment, Social and Governance Report(“ESG Report”).
CHANNEL 7 EVENING NEWS FEATURES STARPHARMA'S VIRALEZE Channel 7 evening news features Starpharma's VIRALEZE. Watch Laurel Irving Channel 7 News (Melbourne) interview Dr Jackie Fairley, CEO of Starpharma about VIRALEZE™ and the need for other COVID-19 preventative tools alongside vaccines. This contains certain forward-looking statements. STARPHARMA | DEP® CABAZITAXEL PROGRESSES TO PHASE 2 ON DEP® cabazitaxel progresses to phase 2 on positive results. Phase 1 part of the DEP® cabazitaxel phase 1 / 2 trial has been successfully completed, with phase 2 to commence immediately. were observed in cancers not usually responsive to conventional cabazitaxel (Jevtana®), such as ovarian cancer, and at doses lower than used forJevtana®.
STARPHARMA SIGNS NEW DEP® PARTNERSHIP WITH CHASE SUN 2 . DEP® ®- Dendrimer Enhanced Product : Starpharma’s DEP® drug delivery platform has demonstrated reproducible preclinical benefits across multiple internal and partnered DEP® programs, including improved efficacy, safety, and survival. Starpharma has three internal DEP® products – DEP ® docetaxel, ®DEP cabazitaxel and DEP irinotecan - in clinical development in patients with solidNEWS - STARPHARMA
Starpharma (ASX:SPL) launches antiviral nasal spray in Europe. May 6th, 2021. The Market Herald reported on Starpharma's launch of VIRALEZE™ in Europe. Read the full article here. Starpharma’s (SPL) antiviral nasal spray is now available for purchase by consumers in Europe via the company’s online store. VIRALEZE is a broadspectrum
MANAGEMENT - STARPHARMA Jackie Fairley, BSc, BVSc (Hons), MBA, GAICD, FTSE Chief Executive Officer. Dr Jackie Fairley has more than 30 years of operational experience in the pharmaceutical and biotechnology industries working in business development and senior management roles with companies including CSL and Faulding/Hospira (now part of Pfizer).MEDIA - STARPHARMA
Media Contacts. For further information about Starpharma's business, please contact: Media. Grant Titmus. Sumit Media. M: +61 419 388 161. grant@sumitmedia.com.au VIVAGEL® - STARPHARMA VivaGel®. VivaGel ® is an innovative technology being applied to a range of sexual/women’s health products by Starpharma and its partners, including: VivaGel® for the prevention of sexually transmitted infections (STIs) VivaGel® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria. Several clinical studieshave successfully
CLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®)) STARPHARMA | SCRIPPS RESEARCH INSTITUTE TESTING SHOWS Jun 01, 2021. Scripps Research Institute testing shows Viraleze highly active against UK COVID strain. Markets Insider featured Viraleze and Starpharma following the announcement of SPL7013's potent antiviral activity against the UK coronavirus variant. STARPHARMA | BOARD OF DIRECTORS Board of Directors Rob B Thomas AO, BEc, MSAA, SF Fin, FAICD, FRSN. Independent non-executive director (appointed 4 December 2013) Chairman from 13 June 2014. Mr Thomas has a strong background in financial services and capital markets and is a non-executive director of several Australian listed companies. PARTNERED-DEP® PRODUCTS Partnered-DEP® products - AZD0466. Starpharma’s DEP® platform enhances the commercial and therapeutic value of a wide range of drugs, making it a highly valuable partnering technology. AZD0466: is a highly optimised nanomedicine formulation of Astra Zeneca cancer drug AZD4320. is currently in a phase 1 clinical trial (FDA IND) STARPHARMA | LLOYDS PHARMACY OFFERING FIRST-EVER COVID Mar 30, 2021. Lloyds Pharmacy offering first-ever Covid nasal spray Viraleze that 'blocks 99.9% of germs' CambridgeshireLive featured news of the VIRALEZE launch in the UK. LEADING BROKER NAMES THE ASX HEALTHCARE SHARES TO BUY IN Leading broker names the ASX healthcare shares to buy in 2021. The Motley Fool reported that Starpharma has been named by analysts at Bell Potter "one of ASX shares from several industries that they believe are best placed to have a strong 2021". HOME - STARPHARMAABOUT USTECHNOLOGYVIVAGELDEP® DRUG DELIVERYNEWSQUARTERLY CASHFLOW REPORT Starpharma Holdings Limited (ASX:SPL, OTCQX:SPHRY) is a global biopharmaceutical company and a world leader in the development of new pharmaceutical and medical products based on proprietary polymers called dendrimers, with programs for COVID-19, DEP ® drug delivery and VivaGel ®.Starpharma has developed VIRALEZE™, an antiviral nasal spray for COVID-19, which is complementary to vaccinesNEWS - STARPHARMA
Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More STARPHARMA | IN THE MEDIA Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More VIRALEZE™ ANTIVIRAL NASAL SPRAY VIRALEZE™ is an antiviral nasal spray that contains astodrimer sodium (SPL7013), which has been shown in laboratory studies to inactivate a broad spectrum of respiratory viruses, including >99.9% of coronavirus SARS-CoV-2, the virus that causes COVID-19.^. ^Paull, J.R.A. et al. Virucidal and antiviral activity of astodrimer sodiumagainst
VIVAGEL® - STARPHARMA VivaGel®. VivaGel ® is an innovative technology being applied to a range of sexual/women’s health products by Starpharma and its partners, including: VivaGel® for the prevention of sexually transmitted infections (STIs) VivaGel® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria. Several clinical studieshave successfully
MEDIA - STARPHARMA
Media Contacts. For further information about Starpharma's business, please contact: Media. Grant Titmus. Sumit Media. M: +61 419 388 161. grant@sumitmedia.com.auVIVAGEL® CONDOM
VivaGel® Condom . VivaGel ® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria.. The physical barrier of the condom provides primary protection against sexually transmitted infections (STIs). The condom is lubricated with VivaGel ® lubricant. VivaGel ® is an antiviral agent proven, in laboratory studies only, to inactivate HIV, herpes simplex virus (HSV) and humanCLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®)) STARPHARMA | DEP® CABAZITAXEL PROGRESSES TO PHASE 2 ON DEP® cabazitaxel progresses to phase 2 on positive results. Phase 1 part of the DEP® cabazitaxel phase 1 / 2 trial has been successfully completed, with phase 2 to commence immediately. were observed in cancers not usually responsive to conventional cabazitaxel (Jevtana®), such as ovarian cancer, and at doses lower than used forJevtana®.
STARPHARMA SIGNS NEW DEP® PARTNERSHIP WITH CHASE SUN 2 . DEP® ®- Dendrimer Enhanced Product : Starpharma’s DEP® drug delivery platform has demonstrated reproducible preclinical benefits across multiple internal and partnered DEP® programs, including improved efficacy, safety, and survival. Starpharma has three internal DEP® products – DEP ® docetaxel, ®DEP cabazitaxel and DEP irinotecan - in clinical development in patients with solid HOME - STARPHARMAABOUT USTECHNOLOGYVIVAGELDEP® DRUG DELIVERYNEWSQUARTERLY CASHFLOW REPORT Starpharma Holdings Limited (ASX:SPL, OTCQX:SPHRY) is a global biopharmaceutical company and a world leader in the development of new pharmaceutical and medical products based on proprietary polymers called dendrimers, with programs for COVID-19, DEP ® drug delivery and VivaGel ®.Starpharma has developed VIRALEZE™, an antiviral nasal spray for COVID-19, which is complementary to vaccinesNEWS - STARPHARMA
Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More STARPHARMA | IN THE MEDIA Starpharma (ASX:SPL)'s nasal spray achieves 98% efficacy against COVID-19 Jun 1st, 2021 "Biopharmaceutical company Starpharma claims the company’s nasal spray product is a highly efficacious means of protection from the UK variant of the coronavirus," Finance News Network reports. Read the full article here.. Read More VIRALEZE™ ANTIVIRAL NASAL SPRAY VIRALEZE™ is an antiviral nasal spray that contains astodrimer sodium (SPL7013), which has been shown in laboratory studies to inactivate a broad spectrum of respiratory viruses, including >99.9% of coronavirus SARS-CoV-2, the virus that causes COVID-19.^. ^Paull, J.R.A. et al. Virucidal and antiviral activity of astodrimer sodiumagainst
VIVAGEL® - STARPHARMA VivaGel®. VivaGel ® is an innovative technology being applied to a range of sexual/women’s health products by Starpharma and its partners, including: VivaGel® for the prevention of sexually transmitted infections (STIs) VivaGel® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria. Several clinical studieshave successfully
MEDIA - STARPHARMA
Media Contacts. For further information about Starpharma's business, please contact: Media. Grant Titmus. Sumit Media. M: +61 419 388 161. grant@sumitmedia.com.auVIVAGEL® CONDOM
VivaGel® Condom . VivaGel ® (SPL7013, or astodrimer sodium) is antiviral and blocks bacteria.. The physical barrier of the condom provides primary protection against sexually transmitted infections (STIs). The condom is lubricated with VivaGel ® lubricant. VivaGel ® is an antiviral agent proven, in laboratory studies only, to inactivate HIV, herpes simplex virus (HSV) and humanCLINICAL TRIALS
DEP ® docetaxel – Phase 2. Status: Recruiting patients. Open-label, two-stage design; Objectives are to establish anti-tumour activity (efficacy) and safety of DEP ® docetaxel (at the Recommended phase 2 Dose (RP2D) of 60mg/m2); First stage to enrol ~20 patients with either lung or prostate cancer (the key approved indications for standard docetaxel (e.g. Taxotere ®)) STARPHARMA | DEP® CABAZITAXEL PROGRESSES TO PHASE 2 ON DEP® cabazitaxel progresses to phase 2 on positive results. Phase 1 part of the DEP® cabazitaxel phase 1 / 2 trial has been successfully completed, with phase 2 to commence immediately. were observed in cancers not usually responsive to conventional cabazitaxel (Jevtana®), such as ovarian cancer, and at doses lower than used forJevtana®.
STARPHARMA SIGNS NEW DEP® PARTNERSHIP WITH CHASE SUN 2 . DEP® ®- Dendrimer Enhanced Product : Starpharma’s DEP® drug delivery platform has demonstrated reproducible preclinical benefits across multiple internal and partnered DEP® programs, including improved efficacy, safety, and survival. Starpharma has three internal DEP® products – DEP ® docetaxel, ®DEP cabazitaxel and DEP irinotecan - in clinical development in patients with solidNEWS - STARPHARMA
Starpharma (ASX:SPL) launches antiviral nasal spray in Europe. May 6th, 2021. The Market Herald reported on Starpharma's launch of VIRALEZE™ in Europe. Read the full article here. Starpharma’s (SPL) antiviral nasal spray is now available for purchase by consumers in Europe via the company’s online store. VIRALEZE is a broadspectrum
ASX ANNOUNCEMENTS
Oct 30th, 2020. Quarterly Cashflow and Activities Report. Oct 29th, 2020. Completion of Placement and SPP raises $48.9M in total. Oct 21st, 2020. Starpharma receives $5.7M R&D tax incentive refund. Oct 20th, 2020. Shareholder Update October 2020. Oct 19th, 2020. CONTACT US - STARPHARMA 4-6 Southampton Crescent Abbotsford VICTORIA 3067 AUSTRALIA. T: +61 3 8532 2700 F: +61 3 9510 5955. View Location in Google Maps : Postal Address: PO Box 2022MEDIA - STARPHARMA
Media Contacts. For further information about Starpharma's business, please contact: Media. Grant Titmus. Sumit Media. M: +61 419 388 161. grant@sumitmedia.com.auFINANCIAL REPORTS
Annual Report 2003 ( pdf file, 738kb) Annual Report 2002 ( pdf file, 1MB) Annual Report 2001 ( pdf file, 343kb) Annual Report 2000 ( pdf file, 532kb) Half Year Reports. Half Year Report December 2019 ( pdf file, 555kb) Half Year Report December 2018 ( pdf file, 766kb) Half Year Report December 2017 ( MANAGEMENT - STARPHARMA Jackie Fairley, BSc, BVSc (Hons), MBA, GAICD, FTSE Chief Executive Officer. Dr Jackie Fairley has more than 30 years of operational experience in the pharmaceutical and biotechnology industries working in business development and senior management roles with companies including CSL and Faulding/Hospira (now part of Pfizer). STARPHARMA | SCRIPPS RESEARCH INSTITUTE TESTING SHOWS Jun 01, 2021. Scripps Research Institute testing shows Viraleze highly active against UK COVID strain. Markets Insider featured Viraleze and Starpharma following the announcement of SPL7013's potent antiviral activity against the UK coronavirus variant.DEP® DRUG DELIVERY
Dendrimer Drug Delivery (DEP®) Starpharma’s dendrimers can be used to enhance the properties of other drugs. The approach is known as “drug delivery” because it is about working to ensure that the drug is delivered to the right part of the body at the right time. Starpharma's dendrimer drug delivery technology is known as DEP ®. STARPHARMA | BOARD OF DIRECTORS Board of Directors Rob B Thomas AO, BEc, MSAA, SF Fin, FAICD, FRSN. Independent non-executive director (appointed 4 December 2013) Chairman from 13 June 2014. Mr Thomas has a strong background in financial services and capital markets and is a non-executive director of several Australian listed companies. PARTNERED-DEP® PRODUCTS Partnered-DEP® products - AZD0466. Starpharma’s DEP® platform enhances the commercial and therapeutic value of a wide range of drugs, making it a highly valuable partnering technology. AZD0466: is a highly optimised nanomedicine formulation of Astra Zeneca cancer drug AZD4320. is currently in a phase 1 clinical trial (FDA IND)* About Us
* Technology
* VIRALEZE
* VivaGel®
* DEP® Drug Delivery* News
Search
Starpharma is a world leader in the development of dendrimer products for pharmaceutical, life science and other applicationsPrevious
VIRALEZE™ launches in Europe Shareholder Newsletter Quarterly Cashflow and Activities Report VIRALEZE SPL7013 highly active in UK variant of coronavirus VIRALEZE™ partners with Harlequins in the UK VIRALEZE™ launches in Europe Shareholder Newsletter Quarterly Cashflow and Activities Report VIRALEZE SPL7013 highly active in UK variant of coronavirus VIRALEZE™ partners with Harlequins in the UK VIRALEZE™ launches in Europe Shareholder Newsletter Quarterly Cashflow and Activities Report Next Starpharma Holdings Limited (ASX:SPL, OTCQX:SPHRY) is a global biopharmaceutical company and a world leader in the development of new pharmaceutical and medical products based on proprietary polymers called dendrimers, with programs for COVID-19, DEP® drug delivery and VivaGel®. Starpharma has developed VIRALEZE™, an antiviral nasal spray for COVID-19, which is complementary to vaccines and other preventative measures such as distancing and PPE. VIRALEZE™ is registered for sale in the UK/Europe and available for purchase by consumers in the UK through LloydsPharmacy and Europe via Starpharma's www.viraleze.co webstore. SPL7013 is utilised in approved products - the VivaGel® condom and VivaGel® BV. VivaGel® BV has been licensed in >160 countries, is approved in >45 countries and available for sale in the UK, Europe, Japan, South East Asia, Australia and New Zealand. As a leading company in dendrimer-based drug delivery, Starpharma’s proprietary drug delivery platform technology, DEP®, is being used to improve pharmaceuticals, to reduce toxicities and enhance their performance. There are numerous internal and partnered programs underway to develop DEP® versions of existing drugs, particularly in the area of anti-cancer therapies. DEP® partnerships include oncology programs with AstraZeneca, with Merck in the area of Antibody Drug Conjugates (ADCs), with Chase Sun in the area of anti-infectives and other world leading pharmaceutical companies. Starpharma’s partnered DEP® programs have the potential to generate significant future milestones and royalties. Investor Centre →* Latest News
* ASX Announcements
* Annual Report 2020* ESG Report
* Analyst Coverage
* Register for email alerts* In the Media
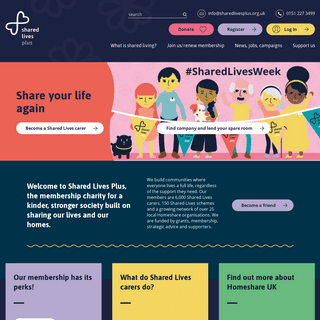
VIRALEZE™ ANTIVIRAL NASAL SPRAY � Starpharma Holdings Limited 2020* Sitemap
* Privacy
* Media
* Disclaimer
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0