Are you over 18 and want to see adult content?
More Annotations

A complete backup of opensourcematters.org
Are you over 18 and want to see adult content?

A complete backup of duwamishtribe.org
Are you over 18 and want to see adult content?

A complete backup of pwelverumandsun.com
Are you over 18 and want to see adult content?

A complete backup of newmarkettoday.ca
Are you over 18 and want to see adult content?

A complete backup of iguazuargentina.com
Are you over 18 and want to see adult content?

A complete backup of actiontrucks.com
Are you over 18 and want to see adult content?
A complete backup of porsche-holding.com
Are you over 18 and want to see adult content?

A complete backup of metropol-moscow.ru
Are you over 18 and want to see adult content?
A complete backup of transcanada-limited.us
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of wolverinesupplies.com
Are you over 18 and want to see adult content?
A complete backup of natcapsolutions.org
Are you over 18 and want to see adult content?
A complete backup of atasteofmylife.fr
Are you over 18 and want to see adult content?
A complete backup of mumonthemove.com
Are you over 18 and want to see adult content?
A complete backup of diencothuanphat.com
Are you over 18 and want to see adult content?
Text
MEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexual FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
FINASTERIDE AND SUICIDE: A POSTMARKETING CASE SERIES Finasteride and Suicide: A Postmarketing Case Series. In this postmarketing case series of 6 former finasteride users who committed suicide, all reported insomnia and persistent sexual dysfunction after medication discontinuation. The most prominent psychiatric symptoms were depression, anxiety, panic attacks, feelings of isolation and“brain
EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
GERMANY JOINS FRENCH DEFIANCE OF FINASTERIDE Germany Joins French Defiance of Finasteride. If PFS awareness is what it takes to unite Germany and France, so be it. Last week brought a slew of media coverage out of Deutschland that closely mirrors what we saw a month earlier in the French Republic. The awareness assault began May 21 with a report on the TV news magazine Markthceck. FINASTERIDE ‘CAUSES SEVERAL ALTERATIONS’ IN THE SECTION OF SOMERSET, N.J., Oct. 1, 2018 – “Finasteride treatment causes several alterations in the hippocampus,” the section of the brain responsible for processing long-term memory and emotional responses, according to a new study conducted at the University of Milano, and the Cajal Institute and Carlos III Health Institute, both in Madrid. REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia orMEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexual FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
FINASTERIDE AND SUICIDE: A POSTMARKETING CASE SERIES Finasteride and Suicide: A Postmarketing Case Series. In this postmarketing case series of 6 former finasteride users who committed suicide, all reported insomnia and persistent sexual dysfunction after medication discontinuation. The most prominent psychiatric symptoms were depression, anxiety, panic attacks, feelings of isolation and“brain
EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
GERMANY JOINS FRENCH DEFIANCE OF FINASTERIDE Germany Joins French Defiance of Finasteride. If PFS awareness is what it takes to unite Germany and France, so be it. Last week brought a slew of media coverage out of Deutschland that closely mirrors what we saw a month earlier in the French Republic. The awareness assault began May 21 with a report on the TV news magazine Markthceck. FINASTERIDE ‘CAUSES SEVERAL ALTERATIONS’ IN THE SECTION OF SOMERSET, N.J., Oct. 1, 2018 – “Finasteride treatment causes several alterations in the hippocampus,” the section of the brain responsible for processing long-term memory and emotional responses, according to a new study conducted at the University of Milano, and the Cajal Institute and Carlos III Health Institute, both in Madrid. REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS andMEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexualPFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. CONTACT THE POST-FINASTERIDE SYNDROME FOUNDATION Post-Finasteride Syndrome Foundation. Philip Roberts. 27 World’s Fair Drive. Somerset, NJ 08873 USA. (856) 425-6046. First name *. Last name. Email address *. Phone number. TOWARDS BETTER PATIENT CARE: DRUGS TO AVOID IN 2021 Prescrire international • February 2021 • Volume 30 n° 223 • Page 51-1 OUTLOOK Towards better patient care: drugs to avoid in 2021 ABSTRACT To make it easier to choose high-quality care, and to prevent disproportionate harm to patients, Prescrire has published its annual update of drugs to avoid. WELCOME - HINDI - THE POST-FINASTERIDE SYNDROME …TRANSLATE THIS PAGE जनवरी 20, 2021 प्रिय मित्रों , जिस राष्ट्र ने हमें महात्मा गांधी, श्रीनिवास रामानुजन और आनंदीबाई जोशी जैसी विद्वान शख़्सियते दीं , अब हुआ थोड़ा और प्रज्ञ 首页 - THE POST-FINASTERIDE SYNDROME FOUNDATIONTRANSLATE THIS PAGE 医护人员应在每次处方非那雄胺之前对患者进行全面评估和风险效益评估。. 作为开处方者,我们的首要职责是不伤害他人。. 医护人员应及时了解这些潜在信号,并在每次处方非那雄胺之前对患者进行全面评估和详细,个性化的风险收益评估。. — Ongoing Concerns FEINBERG SCHOOL OF MEDICINE EPIDEMIOLOGY STUDY SUGGESTS SOMERSET, N.J., March 9, 2017 – More than one percent of young men who took finasteride for 206 days or longer developed persistent erectile dysfunction (PED) that lasted an average of 4.2 years after drug discontinuation, according to a new study published today in PeerJ.. Titled Persistent erectile dysfunction in men exposed to the 5α-reductase inhibitors finasteride or dutasteride, the REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia orMEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexual FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
FINASTERIDE AND SUICIDE: A POSTMARKETING CASE SERIES Finasteride and Suicide: A Postmarketing Case Series. In this postmarketing case series of 6 former finasteride users who committed suicide, all reported insomnia and persistent sexual dysfunction after medication discontinuation. The most prominent psychiatric symptoms were depression, anxiety, panic attacks, feelings of isolation and“brain
EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
GERMANY JOINS FRENCH DEFIANCE OF FINASTERIDE Germany Joins French Defiance of Finasteride. If PFS awareness is what it takes to unite Germany and France, so be it. Last week brought a slew of media coverage out of Deutschland that closely mirrors what we saw a month earlier in the French Republic. The awareness assault began May 21 with a report on the TV news magazine Markthceck. FINASTERIDE ‘CAUSES SEVERAL ALTERATIONS’ IN THE SECTION OF SOMERSET, N.J., Oct. 1, 2018 – “Finasteride treatment causes several alterations in the hippocampus,” the section of the brain responsible for processing long-term memory and emotional responses, according to a new study conducted at the University of Milano, and the Cajal Institute and Carlos III Health Institute, both in Madrid. REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia orMEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexual FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
FINASTERIDE AND SUICIDE: A POSTMARKETING CASE SERIES Finasteride and Suicide: A Postmarketing Case Series. In this postmarketing case series of 6 former finasteride users who committed suicide, all reported insomnia and persistent sexual dysfunction after medication discontinuation. The most prominent psychiatric symptoms were depression, anxiety, panic attacks, feelings of isolation and“brain
EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
GERMANY JOINS FRENCH DEFIANCE OF FINASTERIDE Germany Joins French Defiance of Finasteride. If PFS awareness is what it takes to unite Germany and France, so be it. Last week brought a slew of media coverage out of Deutschland that closely mirrors what we saw a month earlier in the French Republic. The awareness assault began May 21 with a report on the TV news magazine Markthceck. FINASTERIDE ‘CAUSES SEVERAL ALTERATIONS’ IN THE SECTION OF SOMERSET, N.J., Oct. 1, 2018 – “Finasteride treatment causes several alterations in the hippocampus,” the section of the brain responsible for processing long-term memory and emotional responses, according to a new study conducted at the University of Milano, and the Cajal Institute and Carlos III Health Institute, both in Madrid. REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS andMEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexualPFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. CONTACT THE POST-FINASTERIDE SYNDROME FOUNDATION Post-Finasteride Syndrome Foundation. Philip Roberts. 27 World’s Fair Drive. Somerset, NJ 08873 USA. (856) 425-6046. First name *. Last name. Email address *. Phone number. TOWARDS BETTER PATIENT CARE: DRUGS TO AVOID IN 2021 Prescrire international • February 2021 • Volume 30 n° 223 • Page 51-1 OUTLOOK Towards better patient care: drugs to avoid in 2021 ABSTRACT To make it easier to choose high-quality care, and to prevent disproportionate harm to patients, Prescrire has published its annual update of drugs to avoid. WELCOME - HINDI - THE POST-FINASTERIDE SYNDROME …TRANSLATE THIS PAGE जनवरी 20, 2021 प्रिय मित्रों , जिस राष्ट्र ने हमें महात्मा गांधी, श्रीनिवास रामानुजन और आनंदीबाई जोशी जैसी विद्वान शख़्सियते दीं , अब हुआ थोड़ा और प्रज्ञ 首页 - THE POST-FINASTERIDE SYNDROME FOUNDATIONTRANSLATE THIS PAGE 医护人员应在每次处方非那雄胺之前对患者进行全面评估和风险效益评估。. 作为开处方者,我们的首要职责是不伤害他人。. 医护人员应及时了解这些潜在信号,并在每次处方非那雄胺之前对患者进行全面评估和详细,个性化的风险收益评估。. — Ongoing Concerns FEINBERG SCHOOL OF MEDICINE EPIDEMIOLOGY STUDY SUGGESTS SOMERSET, N.J., March 9, 2017 – More than one percent of young men who took finasteride for 206 days or longer developed persistent erectile dysfunction (PED) that lasted an average of 4.2 years after drug discontinuation, according to a new study published today in PeerJ.. Titled Persistent erectile dysfunction in men exposed to the 5α-reductase inhibitors finasteride or dutasteride, the REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia or ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS and FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
PFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
TOWARDS BETTER PATIENT CARE: DRUGS TO AVOID IN 2021 Prescrire international • February 2021 • Volume 30 n° 223 • Page 51-1 OUTLOOK Towards better patient care: drugs to avoid in 2021 ABSTRACT To make it easier to choose high-quality care, and to prevent disproportionate harm to patients, Prescrire has published its annual update of drugs to avoid. EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
GERMANY JOINS FRENCH DEFIANCE OF FINASTERIDE Germany Joins French Defiance of Finasteride. If PFS awareness is what it takes to unite Germany and France, so be it. Last week brought a slew of media coverage out of Deutschland that closely mirrors what we saw a month earlier in the French Republic. The awareness assault began May 21 with a report on the TV news magazine Markthceck. HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia or ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS and FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
PFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
TOWARDS BETTER PATIENT CARE: DRUGS TO AVOID IN 2021 Prescrire international • February 2021 • Volume 30 n° 223 • Page 51-1 OUTLOOK Towards better patient care: drugs to avoid in 2021 ABSTRACT To make it easier to choose high-quality care, and to prevent disproportionate harm to patients, Prescrire has published its annual update of drugs to avoid. EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
GERMANY JOINS FRENCH DEFIANCE OF FINASTERIDE Germany Joins French Defiance of Finasteride. If PFS awareness is what it takes to unite Germany and France, so be it. Last week brought a slew of media coverage out of Deutschland that closely mirrors what we saw a month earlier in the French Republic. The awareness assault began May 21 with a report on the TV news magazine Markthceck. ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS andMEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexualPFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that ONLINE PATIENT SUPPORT PFS patients and family members who wish to share their experiences about the impact the condition has had on their lives are welcome to do so via #MeTooPropecia, a Twitter page edited by PFS patient Michael Adams of Connecticut. You can contact Michael at #MeTooPropecia or metoopropecia@protonmail.com. Aide aux Victimes du Finastéride(France)
PFS CLINICAL STUDIES RECRUITING PATIENTS Research Goals. Determining the underlying biologic mechanisms of PFS, and in turn developing effective therapies for finasteride patients afflicted with the condition, requires ongoing clinical, basic-science and statistical research. Current research is focused on determining underlying biologic mechanisms for PFS at a molecular level, as PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. CONTACT THE POST-FINASTERIDE SYNDROME FOUNDATION Post-Finasteride Syndrome Foundation. Philip Roberts. 27 World’s Fair Drive. Somerset, NJ 08873 USA. (856) 425-6046. First name *. Last name. Email address *. Phone number.PFS FOUNDATION NEWS
April 13, 2021 Dear Friends: Phase V of the PFS Foundation-sponsored research at the University of Milano (UniMi) has found that finasteride, a 5-alpha reductase (5α-R) inhibitor, also inhibits phenylethanolamine N-methyltransferase (PNMT), an enzyme found primarily in the adrenal gland, which in turn may play a role in producing some of the condition’s most common FINASTERIDE AND SUICIDE: A POSTMARKETING CASE SERIES Finasteride and Suicide: A Postmarketing Case Series. In this postmarketing case series of 6 former finasteride users who committed suicide, all reported insomnia and persistent sexual dysfunction after medication discontinuation. The most prominent psychiatric symptoms were depression, anxiety, panic attacks, feelings of isolation and“brain
REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia or ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS and FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
PFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
TOWARDS BETTER PATIENT CARE: DRUGS TO AVOID IN 2021 Prescrire international • February 2021 • Volume 30 n° 223 • Page 51-1 OUTLOOK Towards better patient care: drugs to avoid in 2021 ABSTRACT To make it easier to choose high-quality care, and to prevent disproportionate harm to patients, Prescrire has published its annual update of drugs to avoid. EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
GERMANY JOINS FRENCH DEFIANCE OF FINASTERIDE Germany Joins French Defiance of Finasteride. If PFS awareness is what it takes to unite Germany and France, so be it. Last week brought a slew of media coverage out of Deutschland that closely mirrors what we saw a month earlier in the French Republic. The awareness assault began May 21 with a report on the TV news magazine Markthceck. HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia or ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS and FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
PFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
TOWARDS BETTER PATIENT CARE: DRUGS TO AVOID IN 2021 Prescrire international • February 2021 • Volume 30 n° 223 • Page 51-1 OUTLOOK Towards better patient care: drugs to avoid in 2021 ABSTRACT To make it easier to choose high-quality care, and to prevent disproportionate harm to patients, Prescrire has published its annual update of drugs to avoid. EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
GERMANY JOINS FRENCH DEFIANCE OF FINASTERIDE Germany Joins French Defiance of Finasteride. If PFS awareness is what it takes to unite Germany and France, so be it. Last week brought a slew of media coverage out of Deutschland that closely mirrors what we saw a month earlier in the French Republic. The awareness assault began May 21 with a report on the TV news magazine Markthceck. ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS andMEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexualPFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that ONLINE PATIENT SUPPORT PFS patients and family members who wish to share their experiences about the impact the condition has had on their lives are welcome to do so via #MeTooPropecia, a Twitter page edited by PFS patient Michael Adams of Connecticut. You can contact Michael at #MeTooPropecia or metoopropecia@protonmail.com. Aide aux Victimes du Finastéride(France)
PFS CLINICAL STUDIES RECRUITING PATIENTS Research Goals. Determining the underlying biologic mechanisms of PFS, and in turn developing effective therapies for finasteride patients afflicted with the condition, requires ongoing clinical, basic-science and statistical research. Current research is focused on determining underlying biologic mechanisms for PFS at a molecular level, as PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. CONTACT THE POST-FINASTERIDE SYNDROME FOUNDATION Post-Finasteride Syndrome Foundation. Philip Roberts. 27 World’s Fair Drive. Somerset, NJ 08873 USA. (856) 425-6046. First name *. Last name. Email address *. Phone number.PFS FOUNDATION NEWS
April 13, 2021 Dear Friends: Phase V of the PFS Foundation-sponsored research at the University of Milano (UniMi) has found that finasteride, a 5-alpha reductase (5α-R) inhibitor, also inhibits phenylethanolamine N-methyltransferase (PNMT), an enzyme found primarily in the adrenal gland, which in turn may play a role in producing some of the condition’s most common FINASTERIDE AND SUICIDE: A POSTMARKETING CASE SERIES Finasteride and Suicide: A Postmarketing Case Series. In this postmarketing case series of 6 former finasteride users who committed suicide, all reported insomnia and persistent sexual dysfunction after medication discontinuation. The most prominent psychiatric symptoms were depression, anxiety, panic attacks, feelings of isolation and“brain
REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia or ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS andPFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
ONLINE PATIENT SUPPORT PFS patients and family members who wish to share their experiences about the impact the condition has had on their lives are welcome to do so via #MeTooPropecia, a Twitter page edited by PFS patient Michael Adams of Connecticut. You can contact Michael at #MeTooPropecia or metoopropecia@protonmail.com. Aide aux Victimes du Finastéride(France)
REPORT YOUR SIDE EFFECTS Regulatory Agencies. Anyone suffering from persistent finasteride side effects is encouraged to report them to the appropriate agency. If you are living in the United DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
FINASTERIDE AND SUICIDE: A POSTMARKETING CASE SERIES Finasteride and Suicide: A Postmarketing Case Series. In this postmarketing case series of 6 former finasteride users who committed suicide, all reported insomnia and persistent sexual dysfunction after medication discontinuation. The most prominent psychiatric symptoms were depression, anxiety, panic attacks, feelings of isolation and“brain
HOME - WELCOME TO THE POST-FINASTERIDE FOUNDATION - THERESEARCH INITIATIVESABOUT USCONTACT USDONATE NOWMEDICAL PROFESSIONALS The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS ABOUT POST-FINASTERIDE SYNDROME Overview. Post-finasteride syndrome (PFS) describes persistent sexual, neurological, physical and mental adverse reactions in patients who have taken finasteride, a 5-alpha reductase type II enzyme inhibitor used to treat hair loss (under the brand name Propecia or ABOUT THE POST-FINASTERIDE SYNDROME FOUNDATION Overview. The Post-Finasteride Syndrome Foundation, which is listed in the National Institutes of Health’s Genetic and Rare Diseases Information Center, is dedicated to raising funds for scientific and clinical research into post-finasteride syndrome, a condition with no known cure and few, if any, effective treatments.The organization also focuses on generating global awareness of PFS andPFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, that FREQUENTLY ASKED QUESTIONS Reported symptoms include loss of libido, erectile dysfunction, depression, suicidal ideation, anxiety, panic attacks, Peyronie’s disease, penile shrinkage, chronic testicular pain, gynecomastia, muscle atrophy, cognitive impairment, insomnia and dry skin. (For a complete list of reported PFS symptoms, see our About Post-FinasterideSyndrome
ONLINE PATIENT SUPPORT PFS patients and family members who wish to share their experiences about the impact the condition has had on their lives are welcome to do so via #MeTooPropecia, a Twitter page edited by PFS patient Michael Adams of Connecticut. You can contact Michael at #MeTooPropecia or metoopropecia@protonmail.com. Aide aux Victimes du Finastéride(France)
REPORT YOUR SIDE EFFECTS Regulatory Agencies. Anyone suffering from persistent finasteride side effects is encouraged to report them to the appropriate agency. If you are living in the United DOCTORS & RESEARCHERS SPEAKING OUT Importantly, adverse effects associated with finasteride can persist even after discontinuing its use, leading to what has been termed post-finasteride syndrome, including sexual, physical, and psychiatric symptoms, including depression, anxiety, cognitive impairment, and suicidal ideation. These observations encouraged the presentsystematic
EPIGENETIC MODIFICATIONS DO OCCUR IN PFS PATIENTS, NEW July 20, 2019. Dear Friends: Phase III of the PFS Foundation-sponsored research at the University of Milano (UniMi), which was designed to “study whether epigenetic modifications occur in PFS patients,” has successfully demonstrated epigenetic modifications in PFSpatients.
FINASTERIDE AND SUICIDE: A POSTMARKETING CASE SERIES Finasteride and Suicide: A Postmarketing Case Series. In this postmarketing case series of 6 former finasteride users who committed suicide, all reported insomnia and persistent sexual dysfunction after medication discontinuation. The most prominent psychiatric symptoms were depression, anxiety, panic attacks, feelings of isolation and“brain
PFS BY THE NUMBERS
The PFS Foundation tracks global media coverage of post-finasteride syndrome on an ongoing basis. We then house significant media reports about the condition on our PFS Media Awareness page. To date, we have aggregated 277 national media reports and 52 local and/or trade media reports. If you know of any PFS media reports, in any language, thatMEDICAL LITERATURE
Data presented here indicate that the 5α-R inhibitor FIN is also able to interact with PNMT. This concept is supported by 3D proteome wide-scale screening, by docking and MD simulations, by an in vitro biochemical assay, and in vivo analysis. We believe that the present findings may help in explaining the various side effects reported by FIN users, in particular those related to sexual REPORT YOUR SIDE EFFECTS Regulatory Agencies. Anyone suffering from persistent finasteride side effects is encouraged to report them to the appropriate agency. If you are living in the United PFS CLINICAL STUDIES RECRUITING PATIENTS Research Goals. Determining the underlying biologic mechanisms of PFS, and in turn developing effective therapies for finasteride patients afflicted with the condition, requires ongoing clinical, basic-science and statistical research. Current research is focused on determining underlying biologic mechanisms for PFS at a molecular level, as CONTACT THE POST-FINASTERIDE SYNDROME FOUNDATION Post-Finasteride Syndrome Foundation. Philip Roberts. 27 World’s Fair Drive. Somerset, NJ 08873 USA. (856) 425-6046. First name *. Last name. Email address *. Phone number. PATIENT HEALTH SUPPORT The Post-Finasteride Syndrome Foundation is not a referral service for these or any other health professionals, and itself does not claim to have any effective therapies for PFS. If you would like to suggest adding a medical professional to this list, please email patient manager Philip Roberts at proberts@pfsfoundation.org. PFS FOUNDATION FILES SUPPLEMENTS TO FDA CITIZEN PETITION The PFS Foundation has filed supplements to its FDA Citizen Petition requesting that the agency “immediately require withdrawal of marketing approval for Propeciabecause the risk of serious injury from the drug outweighs its limited benefits.”. The two supplements were filed Dec. 1, 2020 and posted publicly late last month on Regulations.gov, a US Federal government website that serves CLINICAL STUDY OF POST-FINASTERIDE SYNDROME LAUNCHED AT SOMERSET, N.J., Aug. 21, 2013 – The Post-Finasteride Syndrome Foundation today announced the funding of a second major clinical study on post-finasteride syndrome (PFS), this one at Baylor College of Medicine (BCM) in Houston, Texas.. Titled “Genetic and Epigenetic Studies on Post-Finasteride Syndrome Patients,” the research is being led by Mohit Khera (right), MD, MBA, MPH, Assistant REGULATORY UPDATE: FRANCE’S FDA-EQUIVALENT AGENCY REISSUES February 2, 2019. Dear Friends: Prompted by an increasing number of adverse drug-reaction reports to finasteride, France’s FDA-equivalent agency yesterday issued a letter to 100,000 health professionals reminding them that patients treated with the drug are at risk for developing psychiatric disorders and persistent sexualdisorders.
2017 PFS FOUNDATION ANNUAL ADDRESS 2017 PFS Foundation Annual Address. Aug. 4, 2017. Dear Friends: Recent events in Europe and Asia have given me hope that, before this decade is out, we’ll see the day when no man on earth is prescribed finasteride without ample warning of its many potential dangers. On May 24, the UK’s equivalent of the US Food and Drug Administration,the
Skip to content
*
*
*
*
*
* Menu
* Home
* What Is PFS
* What Is PFS?
* FAQs
* PFS by the Numbers* Resources
* Medical Professionals * Support Program Connects PFS Patients Worldwide * Medical Literature * Report Your Side Effects * Online Patient Support* News
* Foundation News
* PFS Media Awareness * Doctors & Researchers Speaking Out * Patients & Loved Ones Crying Out * Research Initiatives* About Us
* Contact Us
* Donate Now
* Menu
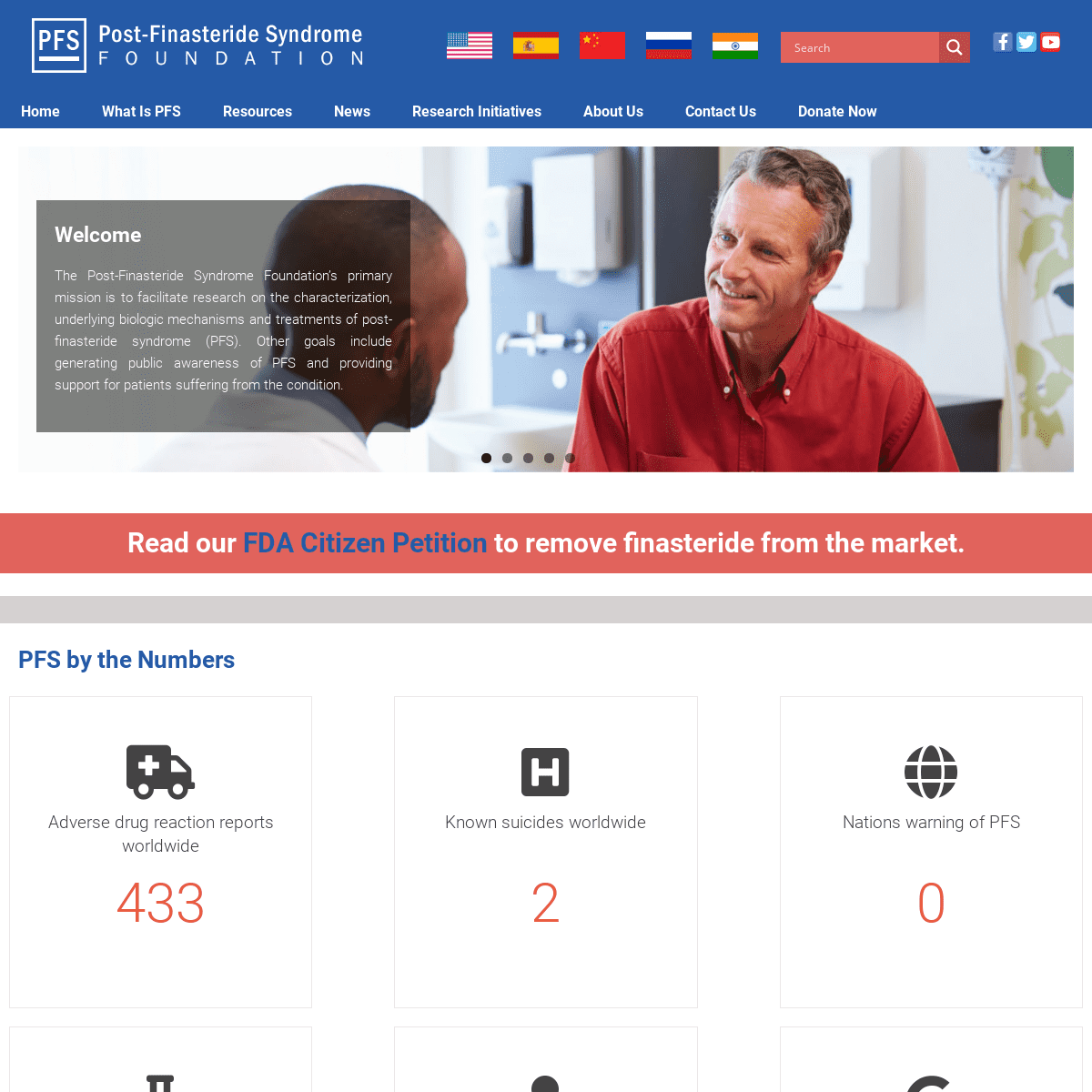
WELCOME
The Post-Finasteride Syndrome Foundation’s primary mission is to facilitate research on the characterization, underlying biologic mechanisms and treatments of post-finasteride syndrome (PFS). Other goals include generating public awareness of PFS and providing support for patients suffering from the condition.*
*
*
*
*
* 1
* 2
* 3
* 4
* 5
READ OUR FDA CITIZEN PETITIONTO REMOVE
FINASTERIDE FROM THE MARKET.PFS BY THE NUMBERS
ADVERSE DRUG REACTION REPORTS WORLDWIDE15,689
PFS RESEARCH STUDIES PUBLISHED47
KNOWN SUICIDES WORLDWIDE78
DOCTORS & RESEARCHERS SPEAKING OUT61
NATIONS WARNING OF PFS0
NATIONAL MEDIA REPORTS WORLDWIDE245
Sources
PATIENT SERVICES
CONNECT WITH OTHER PFS PATIENTS CONSULT KNOWLEDGEABLE DOCTORS REPORT YOUR SIDE EFFECTS READ THE LATEST RESEARCHNEWS
IN ADDITION TO BLOCKING 5Α-R, FINASTERIDE INHIBITS ADRENALINE PRODUCTION, POSSIBLY INDUCING SEXUAL AND PSYCHOLOGICAL SIDE EFFECTS, NEW RESEARCH SUGGESTS April 13, 2021 Dear Friends: Phase V of the PFS Foundation-sponsored research at the University of Milano (UniMi) has found that finasteride, a 5-alpha reductase (5α-R) inhibitor, also inhibits phenylethanolamine N-methyltransferase (PNMT), an enzyme found primarily in the adrenal gland, which in turn may play a role in producing some of the condition’s most common PFS FOUNDATION FILES SUPPLEMENTS TO FDA CITIZEN PETITION SEEKING FINASTERIDE’S REMOVAL FROM THE MARKET February 1, 2021 Dear Friends: The PFS Foundation has filed supplements to its FDA Citizen Petition requesting that the agency “immediately require withdrawal of marketing approval for Propecia…because the risk of serious injury from the drug outweighs its limited benefits.” The two supplements were filed Dec. 1, 2020 and posted publicly late last month on NEW HINDI-LANGUAGE SITE ADDS 615 MILLION POTENTIAL READERS TO THE PFSAWARENESS MOVEMENT
January 20, 2021 Dear Friends: The nation that gave us such wise figures as Mahatma Gandhi, Srinivasa Ramanujan and Anandibai Joshi just got a bit wiser. With respect to PFS, that is. Now, speakers of the world’s third most popular language can read all about post-finasteride syndrome (aka “पोस्ट-फ़ाइस्टरसाइड सिंड्रोम”) in their native tongue. To YOUNG MEN USING FINASTERIDE FOR ALOPECIA MAY BE MORE SUICIDE-PRONE THAN THE GENERAL POPULATION, SAYS NEW PHARMACOVIGILANCE RESEARCH Nov. 12, 2020 Dear Friends: Men 45 years old and younger who use finasteride for hair loss are three times more likely to experience suicidal ideation, plan their suicide or attempt suicide, according to a new pharmacovigilance investigation. Risk of such suicidality was 63 percent higher than among men using other alopecia medications,including minoxidil
GUT MICROBIOTA POPULATION IS ALTERED IN PFS PATIENTS, NEW RESEARCHDEMONSTRATES
Sept. 28, 2020 Dear Friends: Phase IV of the PFS Foundation-sponsored research at the University of Milano (UniMi), designed to investigate the presence of altered gut microbiota in PFS patients, has successfully demonstrated the presence of altered gut microbiota in PFS patients. Titled Alterations of gut microbiota composition in post-finasteride patients: a pilot study, thePOST NAVIGATION
1 2 3
4
5
6
… 15
Next »
More
DOCTORS & RESEARCHERS SPEAKING OUT MEN UNDER THE AGE OF 40 WHO USE FINASTERIDE FOR ALOPECIA ARE AT RISK FOR SUICIDE IF THEY DEVELOP PERSISTENT SEXUAL ADVERSE EFFECTS ANDINSOMNIA.
An important pattern of symptoms was common among all cases who committed suicide in the setting of finasteride use – insomnia and persistent sexual dysfunction after medication discontinuation. Insomnia and fatigue/tiredness were some of the most debilitating symptoms… Men under the age of 40 who use finasteride for alopecia are at risk for suicide if they develop persistent sexual adverse effects and insomnia. —Finasteride and Suicide: A Postmarketing Case Series : Dermatology, January14, 2020
Michael S. Irwig, MD, Attending Endocrinologist Beth Israel Deaconess Medical Center TWO SUBJECTS—EIGHT PERCENT—COMMITTED SUICIDE DURING OR AFTER THESTUDY.
While the sexual side effects of 5ARIs are well known, there may be persistent genitourinary, physical, psycho-cognitive, anti-androgenic and penile vascular changes after 5ARI discontinuation. Use of 5ARIs for treatment of AGA may lead to persistent sexual, genitourinary, physical, psycho-cognitive, and anti-androgenic sequelae even after cessation of 5ARI therapy… Two subjects (8%) committed suicide during or after the study. —Penile vascular abnormalities in young men with persistent side effects after finasteride use for the treatment of androgenic alopecia:
Translational Andrology and Urology, April 2020 Mohit Khera, MD, Director, Laboratory for Andrology Research Baylor College of Medicine HEALTH CARE PROFESSIONALS SHOULD…CONDUCT A FULL EVALUATION AND A…RISK-BENEFIT ASSESSMENT FOR PATIENTS BEFORE EACH PRESCRIPTION OFFINASTERIDE.
As prescribers, our primary duty is to do no harm. Health care professionals should keep themselves abreast of these potential signals and, accordingly, conduct a full evaluation and a detailed, personalized risk-benefit assessment for patients before each prescription of finasteride. —Ongoing Concerns Regarding Finasteride for the Treatment of Male-Pattern Androgenetic Alopecia:
JAMA Dermatology, November 2020 Roger S. Ho, MD, Associate Professor of DermatologyNYU Langone Health
THIS DRUG FOR YOUNG MEN MUST BE TAKEN OFF THE MARKET. As of 2018, 15,000 cases of adverse reactions to finasteride were reported to the WHO—which is a huge number to begin with. And the interesting thing is that the average age of this group was 32 to 34 years old… This drug for young men must be taken off the market. —Documentary on German network NDR titled Side Effects of Finasteride Are Underestimated: January 25, 2021
Hartmut Porst, MD
Founder, European Institute for Sexual HealthMore
PATIENTS & LOVED ONES CRYING OUTMOTHER OF PATIENT
HE WAS A HAPPY, EASY GOING, KIND AND CARING TEENAGER AND PROPECIA HAS MADE HIM MENTALLY DISABLED. My son, 31, had a mild form of BDD and was worried about his hair thinning and took Propecia in 2013. He was taking it for 4 years during which his personality changed beyond recognition and I suspect he developed PFS. He experienced severe side effects: insomnia, loss of libido, obsessive thoughts, anxiety, depression and suicidal ideation. These deteriorated since he stopped taking Propecia in 2017. He was no longer able to go to work, lost the ability to sleep well, was unable to care for his 2 daughters and is feeling increasingly suicidal. He had to go to the emergency department twice this year and now is in a mental hospital in New York. Vulnerable young people should not be prescribed Propecia so easily and drug producers should provide a clear warning about such dangerous side effects. I believe Propecia ruined his health, career and life. He was a happy, easy going, kind and caring teenager and Propecia has made him mentally disabled. I’m very upset and do not know what to do. GL, 62, OXFORD, ENGLAND: AUG. 3, 2018 CONDITION UPDATE: SEPTEMBER 2020NO IMPROVEMENT
MOTHER OF PATIENT
HE’D BEEN USING TOPICAL MEDICATIONS FOR 5 YEARS AND DECIDED TO TRY FINESTERIDE. THAT WAS THE BEGINNING OF A NIGHTMARE. My son was a healthy, smart ambitious 28-year-old man who was going back to school at Suffolk University with a 4.0. He also worked and was in fantastic health, climbing mt Washington in 4 hours for fun. He was, however, battling losing his hair. He’d been using topical medications for 5 years and decided to try Finesteride. That was the beginning of a nightmare. His sister was getting married Alugust 18th. 4 days before the wedding it started…muscle spasms, nonstop and severe anxiety. He had only been on it for 3 months. He immediately stopped taking it but it has continued for the last 5 months. Now though, he is in pain. He has been seeing a neurologist, and is convinced he must have ALS, although so far the tests do not point to that. He started seeing a therapist also hoping it was stress. But the pain and twitching and insomnia are real. He does not currently have any erectile issues thankfully but the physical and mental stress he experiences is horrible. He continues to search foir answers but i am convinced it is from the Finesteride. LM, LOWELL, MA: JAN. 6, 2019 CONDITION UPDATE: AUGUST 2020SLIGHTLY WORSE
PFS PATIENT (MALE)
I AM LIVING NOW FOR SEVEN YEARS WITH MY PARENTS IN THEIR TINY APARTMENT AND DO NOT KNOW WHAT TO DO. I am 35 years old and have this Syndrome since I have been 24 years old. My Life was smashed and destroyed after I started with Finasteride. I can’t stop thinking about this one moment I decided to go to this special Doctor who treats hairloss. I was 24 years young and I had full beautiful hair. This Doctor made some tests and scared the shit out of me my prognosing a full bald me in a few years. I trusted him and took the pills he gave me. Safe pills he said. No side-effects. Soon after I started the pills my life made a u-turn. I was finishing university and had already found a new job in a new city. That’s when I start to enter hell. I lost my mind and had the most disgusting mental health issues. I had to go to the hospital and started with suicide thoughts. It was an absolute nightmare! Fast Forward. I am living now for seven years with my parents in their tiny apartment and do not know what to do. I feel a bit better because I take a lot of different medications but I can not go outside or even read a book because I feel sedated. I really do not know what to dowith my life.
HH, 35, STUTTGART, GERMANY: FEB. 3, 2019 CONDITION UPDATE: SEPTEMBER 2020NO IMPROVEMENT
More
JOIN OUR MAILING LIST* Email*
* Are you a...*
* Health Care Professional * Media Professional * PFS Patient/Friend/Family Member * Regulatory Agency Employee* Comments
This field is for validation purposes and should be left unchanged. The Post-Finasteride Syndrome Foundation © 2021Notifications
Generic selectors
Exact matches only
Exact matches only
Search in title
Search in title
Search in content
Search in content
Search in excerpt
Hidden
Hidden
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0