Are you over 18 and want to see adult content?
More Annotations

A complete backup of historyofrecording.com
Are you over 18 and want to see adult content?

A complete backup of adscendmedia.com
Are you over 18 and want to see adult content?

A complete backup of weatherpeace.blogspot.com
Are you over 18 and want to see adult content?

A complete backup of soelleroed-kro.dk
Are you over 18 and want to see adult content?

A complete backup of sniperstores.com
Are you over 18 and want to see adult content?
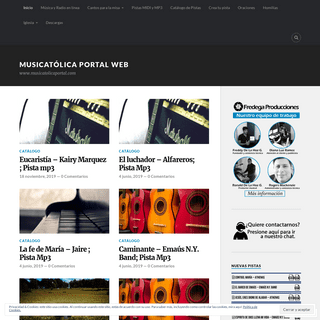
A complete backup of musicatolicaportal.wordpress.com
Are you over 18 and want to see adult content?
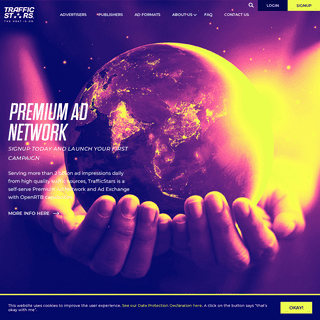
A complete backup of trafficstars.com
Are you over 18 and want to see adult content?

A complete backup of penziletu.blogspot.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of redevaluacionformativa.wordpress.com
Are you over 18 and want to see adult content?

A complete backup of emprendepyme.net
Are you over 18 and want to see adult content?

A complete backup of ereferencedesk.com
Are you over 18 and want to see adult content?

A complete backup of betaprofiles.com
Are you over 18 and want to see adult content?

A complete backup of investigandohistoriaygeografia902.blogspot.com
Are you over 18 and want to see adult content?

A complete backup of securitymanager.gr
Are you over 18 and want to see adult content?
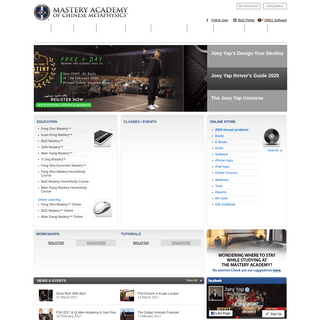
A complete backup of masteryacademy.com
Are you over 18 and want to see adult content?
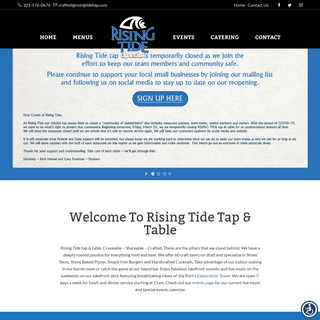
A complete backup of risingtidetap.com
Are you over 18 and want to see adult content?
Text
PROGRAM OVERVIEW
Each year Pfizer selects accomplished new and recent graduate degree-level candidates from top universities for the Pfizer Parenteral R&D Professional Development Program (PDP).. PDP Scientists engage in 4 rotations that encompass every aspect of parenteral drug development. This work begins in Pipeline Development, where new product concepts are identified. PFIZER PARENTERAL R&D The Pfizer Parenteral R&D Professional Development Program is an intensive, two-year rotational program designed to provide recent graduates with diverse and challenging cross-functional R&D assignments that prepare them to assume significant roles in driving Pfizer’s future in the hospital business. The program is designed toaccelerate
APPLY NOW | PFIZER PARENTERAL R&D A PhD or Thesis Masters in Pharmaceutical Sciences, Chemistry, Biomedical/Chemical Engineering or an equivalent discipline. Strong interpersonal skills with demonstrated leadership ability. Relevant research or work experience, internships, etc. A passion for learningand pursuing a
FAQS | PFIZER PARENTERAL R&D The PDP is based in Lake Forest, IL, a northern suburb of Chicago. In exceptional cases, business needs have allowed PDP Scientists to perform a rotation at another R&D facility. How does the selection process work? Once we receive all of the applications from a university, an initial screen is performed. We phone screen the mostpromising
EXPERIENCE BEST-IN-CLASS You achieved your graduate degree, now it’s time to apply your knowledge to develop specialty parenteral products that positively impact patient care. The Pfizer Parenteral R&D Professional Development Program is an intensive, two-year rotational programdesigned
PROGRAM OVERVIEW
Each year Pfizer selects accomplished new and recent graduate degree-level candidates from top universities for the Pfizer Parenteral R&D Professional Development Program (PDP).. PDP Scientists engage in 4 rotations that encompass every aspect of parenteral drug development. This work begins in Pipeline Development, where new product concepts are identified. PFIZER PARENTERAL R&D The Pfizer Parenteral R&D Professional Development Program is an intensive, two-year rotational program designed to provide recent graduates with diverse and challenging cross-functional R&D assignments that prepare them to assume significant roles in driving Pfizer’s future in the hospital business. The program is designed toaccelerate
APPLY NOW | PFIZER PARENTERAL R&D A PhD or Thesis Masters in Pharmaceutical Sciences, Chemistry, Biomedical/Chemical Engineering or an equivalent discipline. Strong interpersonal skills with demonstrated leadership ability. Relevant research or work experience, internships, etc. A passion for learningand pursuing a
FAQS | PFIZER PARENTERAL R&D The PDP is based in Lake Forest, IL, a northern suburb of Chicago. In exceptional cases, business needs have allowed PDP Scientists to perform a rotation at another R&D facility. How does the selection process work? Once we receive all of the applications from a university, an initial screen is performed. We phone screen the mostpromising
EXPERIENCE BEST-IN-CLASS You achieved your graduate degree, now it’s time to apply your knowledge to develop specialty parenteral products that positively impact patient care. The Pfizer Parenteral R&D Professional Development Program is an intensive, two-year rotational programdesigned
PROGRAM OVERVIEW
Each year Pfizer selects accomplished new and recent graduate degree-level candidates from top universities for the Pfizer Parenteral R&D Professional Development Program (PDP).. PDP Scientists engage in 4 rotations that encompass every aspect of parenteral drug development. This work begins in Pipeline Development, where new product concepts are identified. PFIZER PARENTERAL R&D The Pfizer Parenteral R&D Professional Development Program is an intensive, two-year rotational program designed to provide recent graduates with diverse and challenging cross-functional R&D assignments that prepare them to assume significant roles in driving Pfizer’s future in the hospital business. The program is designed toaccelerate
FAQS | PFIZER PARENTERAL R&D The PDP is based in Lake Forest, IL, a northern suburb of Chicago. In exceptional cases, business needs have allowed PDP Scientists to perform a rotation at another R&D facility. How does the selection process work? Once we receive all of the applications from a university, an initial screen is performed. We phone screen the mostpromising
APPLY NOW | PFIZER PARENTERAL R&D A PhD or Thesis Masters in Pharmaceutical Sciences, Chemistry, Biomedical/Chemical Engineering or an equivalent discipline. Strong interpersonal skills with demonstrated leadership ability. Relevant research or work experience, internships, etc. A passion for learningand pursuing a
PFIZER PARENTERAL R&D You achieved your graduate degree, now it’s time to apply your knowledge to develop specialty parenteral products that positively impact patient care.PROGRAM OVERVIEW
Each year Pfizer selects accomplished new and recent graduate degree-level candidates from top universities for the Pfizer Parenteral R&D Professional Development Program (PDP).. PDP Scientists engage in 4 rotations that encompass every aspect of parenteral drug development. This work begins in Pipeline Development, where new product concepts are identified. APPLY NOW | PFIZER PARENTERAL R&DPFIZER DEVELOPMENT PROGRAMPFIZER DIGITAL ROTATIONAL PROGRAMPFIZER MBA INTERNSHIPPFIZER PATIENT ASSISTANCE APPLICATION PDFPFIZER SUMMER INTERNSHIP Qualifications & Skills A PhD in Pharmaceutical Sciences, Chemistry, Biomedical/Chemical Engineering or an equivalent discipline Strong interpersonal skills with demonstrated leadership ability Relevant research or work experience, internships, etc. FAQS | PFIZER PARENTERAL R&DPFIZER DEVELOPMENT PROGRAMPFIZER DIGITAL ROTATIONAL PROGRAMPFIZER MBA INTERNSHIPPFIZER PATIENT ASSISTANCE APPLICATION PDFPFIZER SUMMER INTERNSHIP Our recruitment cycle takes place from August to November, annually. Applications are due by a specific date for each university we visit (see the Apply Now page for more details). EXPERIENCE BEST-IN-CLASS You achieved your graduate degree, now it’s time to apply your knowledge to develop specialty parenteral products that positively impact patient care. The Pfizer Parenteral R&D Professional Development Program is an intensive, two-year rotational programdesigned
PFIZER PARENTERAL R&D You achieved your graduate degree, now it’s time to apply your knowledge to develop specialty parenteral products that positively impact patient care.PROGRAM OVERVIEW
Each year Pfizer selects accomplished new and recent graduate degree-level candidates from top universities for the Pfizer Parenteral R&D Professional Development Program (PDP).. PDP Scientists engage in 4 rotations that encompass every aspect of parenteral drug development. This work begins in Pipeline Development, where new product concepts are identified. APPLY NOW | PFIZER PARENTERAL R&DPFIZER DEVELOPMENT PROGRAMPFIZER DIGITAL ROTATIONAL PROGRAMPFIZER MBA INTERNSHIPPFIZER PATIENT ASSISTANCE APPLICATION PDFPFIZER SUMMER INTERNSHIP Qualifications & Skills A PhD in Pharmaceutical Sciences, Chemistry, Biomedical/Chemical Engineering or an equivalent discipline Strong interpersonal skills with demonstrated leadership ability Relevant research or work experience, internships, etc. FAQS | PFIZER PARENTERAL R&DPFIZER DEVELOPMENT PROGRAMPFIZER DIGITAL ROTATIONAL PROGRAMPFIZER MBA INTERNSHIPPFIZER PATIENT ASSISTANCE APPLICATION PDFPFIZER SUMMER INTERNSHIP Our recruitment cycle takes place from August to November, annually. Applications are due by a specific date for each university we visit (see the Apply Now page for more details). EXPERIENCE BEST-IN-CLASS You achieved your graduate degree, now it’s time to apply your knowledge to develop specialty parenteral products that positively impact patient care. The Pfizer Parenteral R&D Professional Development Program is an intensive, two-year rotational programdesigned
PFIZER PARENTERAL R&D You achieved your graduate degree, now it’s time to apply your knowledge to develop specialty parenteral products that positively impact patient care.PROGRAM OVERVIEW
Each year Pfizer selects accomplished new and recent graduate degree-level candidates from top universities for the Pfizer Parenteral R&D Professional Development Program (PDP).. PDP Scientists engage in 4 rotations that encompass every aspect of parenteral drug development. This work begins in Pipeline Development, where new product concepts are identified. APPLY NOW | PFIZER PARENTERAL R&D Qualifications & Skills A PhD in Pharmaceutical Sciences, Chemistry, Biomedical/Chemical Engineering or an equivalent discipline Strong interpersonal skills with demonstrated leadership ability Relevant research or work experience, internships, etc. FAQS | PFIZER PARENTERAL R&D Our recruitment cycle takes place from August to November, annually. Applications are due by a specific date for each university we visit (see the Apply Now page for more details). Skip to main contentHEADER TOP MENU
*
Careers
*
Investors
*
News
*
Press Release Archive*
Press Statements
*
Podcasts
*
The Antigen Podcast
*
Diverse Perspectives Podcast*
menopause: unmuted
*
Hemcast
*
Contact Media Relations*
Press Kits & Downloads*
Company Press Kit
*
Pfizer Oncology Press Kit*
PSA Narrative
*
RA NarRAtive
*
Rare Disease Gene Therapy Press Kit*
ATTR-Amyloidosis Press Kit*
UC Narrative
*
Eczema Inside Out Press Kit*
Partners
*
Discovery to Pre-candidate*
Focus Areas
*
Therapeutic Areas
*
Internal Medicine
*
Inflammation and Immunology*
Oncology
*
Rare Disease
*
Vaccines
*
Therapeutic Modalities and Technology Platforms*
Drug Safety
*
Gene Therapy
*
Target Sciences
*
Precision Medicine
*
Medicinal Sciences
*
Biologics Product & Process Development*
Biotherapeutics Discovery*
Medicine Design
*
Small Molecule Product & Process Development*
Emerging Science
*
DNA Damage Response
*
Repeat Expansion
*
Senescence
*
Partnering Models
*
Candidate Through Commercialization*
Focus Areas
*
Internal Medicine
*
Inflammation and Immunology*
Oncology
*
Rare Disease
*
Vaccines
*
Hospital
*
Pfizer CentreOne
*
Pfizer Ventures
*
Our portfolio
HEALTHCARE PROFESSIONALS*
Healthcare ProfessionalsCONNECT WITH US
*
Connect with Us
*
*
FAQ
*
Privacy Policy (NEW)*
YouTube
*
Twitter(link is external)*
Facebook(link is external)*
LinkedIn(link is external)*
Instagram(link is external)En
Current country/region and language options:United States
English (En)
*
Argentina - Español*
Australia - English
*
Austria - Deutsch
*
Belarus - Belarusian - English*
Belgium - English - Nederlands- Français
*
Brazil - Português
*
Bulgaria - Български*
Canada - English - Français*
China - ä¸æ–‡ - English*
Colombia - Español
*
Croatia - Croatian
*
Czech Republic - Čeština*
Denmark - Dansk
*
Ecuador - Español
*
Estonia - Eesti - English*
Finland - Suomalainen*
France - Français
*
Germany - Deutsch
*
Greece - ελληνικά*
Hong Kong - 繁體中文 - English*
Hungary - Magyar
*
India - English
*
Indonesia - Indonesian*
Ireland - English
*
Israel - עברית
*
Italy - Italiano
*
Japan - 日本の
*
Korea - 한국의
*
Latvia - Latvian - English*
Lithuania - Lithuanian - English*
Malaysia - English
*
Mexico - Español
*
Netherlands - Nederlands*
New Zealand - English*
Norway - Norsk
*
Pakistan - English
*
Peru - Español
*
Philippines - English*
Poland - Polski
*
Portugal - Português*
Russia - Русский*
Serbia - Serbian
*
Singapore - English
*
Slovenia - Slovenian*
Slovakia - Slovenský*
South Africa - English*
Spain - Español
*
Sweden - Svenska
*
Switzerland - Deutsch - Français*
Taiwan - Taiwan
*
Thailand - ภาษาไทย*
Tunisia - Français
*
Turkey - Türk
*
United Kingdom - English*
United States - English*
Ukraine - Ukrainian
*
Vietnam - Tiếng Việt - English*
Venezuela - Español Apply Toggle navigation*
Your Health
*
Healthy Living
*
Brain & Nervous System*
Cardiovascular Health*
Cold & Flu
*
Digestive Health
*
Healthier Aging
*
Living Tobacco Free
*
Men’s Health
*
Mental Health
*
Nutrition & Fitness
*
Pediatric Health
*
Respiratory Health
*
Skin Health
*
Vaccines & Prevention*
Women’s Health
*
Disease & Condition
*
Cancer
*
Cardiovascular Diseases & Conditions*
Diabetes & Metabolic Disorders*
Immune System Diseases & Conditions*
Infections & Infectious Diseases*
Neurological Diseases & Conditions*
Menopause
*
Rare Disease
*
Vaccine-Related Conditions*
Skin Diseases & Conditions*
Atopic Dermatitis
*
Cardiovascular Disease*
Severe Hypertriglyceridemia*
Coronavirus disease (COVID-19) Resources*
Managing your health*
Caregiving
*
Evaluating Health Information*
Tracking Your Health*
Your Medicines
*
Health Literacy
*
Wellness
Hot Topics
MAINTAINING EMOTIONAL WELL-BEING DURING COVID-19*
Our Science
*
Driven to Discover the cure*
Focus Areas
*
Internal Medicine
*
Inflammation & Immunology*
Oncology
*
Rare Disease
*
Vaccines
*
Anti Infectives
*
Product Pipeline
*
Clinical Trials
*
Guide to Clinical Trials*
Find a Trial
*
Expanded Access & Compassionate Use*
Clinical Trials in Children*
Partnering with Pfizer*
Trial Data & Results*
Plain Language Study Results Summaries*
Research Integrity & Transparency*
Diversity in Clinical Trials*
R&D Platforms
*
Biosimilars
*
Gene Therapy
*
Medicinal Sciences
*
Precision Medicine
*
Maternal Immunization*
Collaboration
*
Partner with Pfizer
*
Research sites
*
Coronavirus disease (COVID-19) Resources*
All COVID-19 Updates*
Vaccine Efforts
*
Antiviral Efforts
*
Partnerships & Anti-Infective Efforts*
Supporting the Fight Against COVID-19HOT TOPICS
MRNA VS. CONVENTIONAL VACCINES*
Our People
*
Meet The Experts
*
Scientists
*
Internal Medicine – Metabolic Disease*
Inflammation & Immunology*
Oncology
*
Rare Disease
*
Vaccines
*
Medical Professionals*
The Journey
*
Leadership
*
Executives
*
Board Members
*
Company Fact Sheet
*
Diverse Perspectives*
Diversity & Inclusion*
Supplier Diversity
HOT TOPICS
MEET OUR NEW CHIEF PATIENT OFFICER, DR. DARA RICHARDSON-HERON WORKING TOGETHER TO EMPOWER WOMEN REDUCING HEALTH DISPARITIES IN LATINO COMMUNITIES*
Hot Topics
*
Our Purpose
*
Commitment to Global Health*
Expanding Access
*
Strengthening Health Systems*
Providing Disaster & Humanitarian Relief*
Unleashing the Power of Our Colleagues*
Highlighting Individual Voices*
Protecting People and the Environment*
Pfizer's Green Journey*
Product Stewardship
*
EHS Governance
*
EHS Supply Chain
*
Key Performance Indicators*
Health & Safety
*
Human Rights
*
Positions on Health Policies*
Ready for Cures
*
How does Pfizer price medicines?*
Value of Medicines
*
Policy Positions
*
Creating Cures Through Facility Investments*
IP PACT
*
Transparency
*
Working with Health Care Professionals*
Transparency in Grants*
Code of Conduct
*
Corporate Compliance*
Sales and Marketing Compliance*
Independent Grants
*
Competitive Grants Program*
Independent Medical Education*
Investigator Sponsored Research*
Quality Improvement
*
COVID-19 Vaccine Grants*
Contributions & Partnerships*
Healthcare Charitable Contributions*
Lobbying & Political ContributionsHOT TOPICS
PFIZER WOMEN’S RESOURCE GROUP*
Hot Topics
*
Our Products
*
Product listing
*
How drugs are made
*
Branded vs. Generic
*
Biologics & Biosimilars*
Commitment to Quality*
Distributors
*
Pfizer Distributors
*
Greenstone Distributors*
Medicine Safety
*
Reporting Adverse Events*
Making Good Treatment Choices*
Partnering With Patients*
Health Literacy
*
Medicine Safety Tips for Patients*
Counterfeiting
*
Unused Medicines
*
Safety Data Sheets
*
Medical Information Contacts*
Pfizer Global SupplyHOT TOPICS
UNIVERSAL VS. SINGLE PAYER HEALTHCARE*
Careers
*
Working Here
*
Mission & Purpose
*
Culture
*
Diversity & Inclusion*
Career Growth & Colleague Development*
Compensation & Benefits*
Ensuring Corporate Responsibility*
Search Jobs(link is external)*
Student & Early Career*
Postdoctoral Program*
US Summer Student Worker Program*
Opportunities for Veterans*
Pfizer365.com(link is external)*
Our Hiring & Recruiting Process*
Frequently Asked Questions*
Tips for Interviewing at Pfizer*
Connect With Us
*
Investors
*
Investors Overview
*
Why Invest / Our Story*
Prospective Investors*
Fact Sheet
*
Events & Presentations*
Financials
*
Quarterly Reports
*
Annual Reports
*
SEC Filings
*
Interactive Data Filings*
Investor News
*
Stock Information
*
Historical Price Lookup*
Investment Calculator*
Historical Stock Chart & Data*
Dividend & Split History*
Stock FAQs
*
Governance
*
Board Committees and Charters*
The Pfizer Board Policies*
Corporate Governance FAQs*
Contact Our Directors*
Shareholder Services*
Stock FAQs
*
Advisory Information*
Cost Basis Calculator*
News
*
Contact Media Relations*
Podcasts
*
Press Kits & Downloads*
Press Release Archive*
Press Statements
*
Partners
*
" sc:linkname="menu:home:footer links">Footer links*
" sc:linkname="menu:home:footer aside section1 ">Footer Aside Section1*
CAREER-SEEKERS
*
INVESTORS
*
News & Media
*
Partners
*
" sc:linkname="menu:home:footer aside section 2 ">Footer Aside Section2
*
Healthcare Professionals*
Business To Business*
Change Region
*
Sitemap
*
" sc:linkname="menu:home:footer aside section3 ">Footer Aside Section3*
Twitter(link is external)*
Facebook(link is external)*
Linkedin(link is external)*
Instagram(link is external)*
Youtube
*
" sc:linkname="menu:home:footer aside section4 ">Footer Aside Section4*
Contact Us
*
Terms of Use
*
Privacy Policy
Our Purpose
Our Purpose
Supporting India in Its Time of Need We are mobilizing the largest humanitarian relief effort in our company’s history to provide urgent and immediate support to patients being treated in public hospitals. Learn more about our response VISIT OUR COVID-19 HUBYour Health
VISIT OUR COVID-19 HUB Sharing resources to stay safe and updates on our efforts we hope could bring an end to the global health crisis.Learn More
THE FACTS ABOUT PFIZER AND BIONTECH’S COVID-19 VACCINEOur Science
THE FACTS ABOUT PFIZER AND BIONTECH’S COVID-19 VACCINE The U.S. FDA authorization of the Pfizer-BioNTech COVID-19 vaccine for emergency use is a significant step forward in our fight against thispandemic.
Learn More
COVID-19 SCIENTIFIC RESOURCESOur Science
COVID-19 SCIENTIFIC RESOURCES Current information on our scientific progress to bring forward a safe and effective vaccine to help protect against the novel coronavirus.More Here
IN HIS SHOES (OR HIKING BOOTS)Your Health
IN HIS SHOES (OR HIKING BOOTS) When it comes to making the world a better place for people living with hemophilia—a rare disease characterized by inadequate blood clotting—William Addison doesn’t just talk the talk. Learn more about WilliamSEARCH PRODUCTS
Submit
Search products, A–Z Need help paying for Pfizer medicines?FIND A TRIAL
Search for Clinical Trials by condition, keyword or trial number. Share your location or enter your city or zip code to find studiesnear you.
Condition, Keyword, or NCT NumberLocation
Search
Advanced Search
GETTING TO KNOW PFIZER’S NEW CHIEF MEDICAL OFFICER AIDA HABTEZIONOur People
GETTING TO KNOW PFIZER’S NEW CHIEF MEDICAL OFFICER AIDA HABTEZION Changing careers in the middle of a pandemic is no small endeavor. But when Aida Habtezion, M.D., learned about the opportunity to serve as Pfizer’s new Chief Medical Officer, she couldn’t pass it up.Meet Aida
PFIZER’S COMMITMENT TO PATIENTS REFLECTED IN NEW IP PACTOur Purpose
PFIZER’S COMMITMENT TO PATIENTS REFLECTED IN NEW IP PACT Understand the way we use IP and why it matters for what we do. See how we are living up to that commitmentLATEST NEWS
PFIZER AND BIONTECH RECEIVE FIRST AUTHORIZATION… NEW YORK and MAINZ, GERMANY, May 28, 2021 — Pfizer Inc. (NYSE: PFE) and BioNTech SE (Nasdaq: BNTX) today announced that the Conditional Marketing Authorization (CMA) for COMIRNATY® in the Euro...Read more
FOOTER MENU
*
Your Health
*
Healthy Living
*
Disease Conditions
*
Manage Your Health
*
Our Science
*
Therapeutic Areas
*
Product Pipeline
*
Clinical Trials
*
R&D Platforms
*
Collaboration
*
Research SItes
*
Our People
*
Meet the Experts
*
The Journey
*
Leadership
*
Diverse Perspectives*
Our Purpose
*
Commitment To Global Health*
Positions on Health Policies*
Transparency
*
Contributions & Partnerships*
Our Products
*
Product Listing
*
How Drugs are Made
*
Distributors
*
Global Product Information ContactsFOOTER ASIDE
*
" sc:linkname="global footer:navigation:footer aside section1 ">FooterAside Section1
*
CAREER-SEEKERS
*
INVESTORS
*
News & Media
*
Partners
*
" sc:linkname="global footer:navigation:footer aside section 2 ">Footer Aside Section 2*
Healthcare Professionals*
Business To Business*
Change Region
*
Sitemap
*
" sc:linkname="global footer:navigation:footer aside section3 ">FooterAside Section3
*
Twitter(link is external)*
Facebook(link is external)*
Linkedin(link is external)*
Instagram(link is external)*
Youtube
*
" sc:linkname="global footer:navigation:footer aside section4 ">FooterAside Section4
*
Contact Us
*
Terms of Use
*
Privacy Policy
Copyright © 2002-2021 Pfizer Inc. All rights reserved. This information—including product information—is intended only for residents of the United States. The products discussed herein may have different labeling in differentcountries.
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0