Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://militarycampgrounds.us
Are you over 18 and want to see adult content?

A complete backup of https://wascoskylights.com
Are you over 18 and want to see adult content?

A complete backup of https://careerattraction.com
Are you over 18 and want to see adult content?
A complete backup of https://skepsis.no
Are you over 18 and want to see adult content?

A complete backup of https://servimedia.es
Are you over 18 and want to see adult content?
A complete backup of https://streetscooter.com
Are you over 18 and want to see adult content?

A complete backup of https://sekolahnesia.com
Are you over 18 and want to see adult content?

A complete backup of https://revell.com
Are you over 18 and want to see adult content?
A complete backup of https://photoback.jp
Are you over 18 and want to see adult content?

A complete backup of https://softwearautomation.com
Are you over 18 and want to see adult content?
A complete backup of https://uebermaps.com
Are you over 18 and want to see adult content?

A complete backup of https://socialcooling.com
Are you over 18 and want to see adult content?
Favourite Annotations
A complete backup of onemanandhismustang.com
Are you over 18 and want to see adult content?

A complete backup of iran-efshagari.com
Are you over 18 and want to see adult content?
A complete backup of kjstarr.weebly.com
Are you over 18 and want to see adult content?
A complete backup of aboutthemafia.com
Are you over 18 and want to see adult content?
A complete backup of folkeautomaten.com
Are you over 18 and want to see adult content?
Text
OUR STORY | MC10
MC10 products are thin and flexible, and built to stretch, bend and twist seamlessly with our bodies and the world around us. MC10’s software platform consists of a complete end-to-end system with mobile interfaces, cloud storage and analytical tools. Lean, agile software development allows us to create robust systems to support the high RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PUBLICATIONS
To objectively demonstrate the capabilities of MC10’s biometric sensors, we have collected the following articles. We invite you to browse these resources to learn more about the power of MC10’s groundbreaking hardware and software systems as well as the future of healthcare analytics in general. BioStamp ® Technologies. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641HELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help. REAL WORLD EVIDENCE AND ITS IMPACT ON HEALTHCARE Real World Evidence and Its Impact on Healthcare. Real world evidence, or RWE for short, is the term made famous by the 21st Century Cures Act, a law enacted by the United States Congress in December 2016. Section 3022 of the bill calls for the FDA to establish a program and framework for evaluating the use of RWE, defined as “data regardingSYSTEM - MC10
1 The BioStampRC System is an end-to-end research tool designed for targeted, accurate, and seamless physiological data collection from across the human body. USING DIGITAL ENDPOINTS AND DIGITAL BIOMARKERS IN CLINICAL Using Digital Endpoints and Digital Biomarkers in Clinical Trials. While traditional endpoints and biomarkers are still necessary for FDA submission and regulation, the advent of digital tools has presented the opportunity to revolutionize the data capture method — specifically, the ability to collect more specific, relevant datapoints or
WEARABLE HEALTHCARE TECHNOLOGY & DEVICES MC10’s proprietary BioStamp® system wearable health tech creates comfortable, discreet sensors that can be applied anywhere on the body for targeted data collection. Our wearable sensors are wireless and rechargeable via the included Link Hub. A dedicated mobile phone plus Link Application designed for remote data collection guides users WEARABLE HEALTH SYSTEMS BioStamp nPoint is an FDA 510 (k) cleared medical device designed to collect medical grade, clinical quality bio-metric, physiological and eCOA data in a clinical trial setting. The system collects and processes raw data into recognizable clinical metrics including vital signs, activity/actigraphy and posture classification, sEMG, and sleepOUR STORY | MC10
MC10 products are thin and flexible, and built to stretch, bend and twist seamlessly with our bodies and the world around us. MC10’s software platform consists of a complete end-to-end system with mobile interfaces, cloud storage and analytical tools. Lean, agile software development allows us to create robust systems to support the high RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PUBLICATIONS
To objectively demonstrate the capabilities of MC10’s biometric sensors, we have collected the following articles. We invite you to browse these resources to learn more about the power of MC10’s groundbreaking hardware and software systems as well as the future of healthcare analytics in general. BioStamp ® Technologies. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641HELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help. REAL WORLD EVIDENCE AND ITS IMPACT ON HEALTHCARE Real World Evidence and Its Impact on Healthcare. Real world evidence, or RWE for short, is the term made famous by the 21st Century Cures Act, a law enacted by the United States Congress in December 2016. Section 3022 of the bill calls for the FDA to establish a program and framework for evaluating the use of RWE, defined as “data regardingSYSTEM - MC10
1 The BioStampRC System is an end-to-end research tool designed for targeted, accurate, and seamless physiological data collection from across the human body. USING DIGITAL ENDPOINTS AND DIGITAL BIOMARKERS IN CLINICAL Using Digital Endpoints and Digital Biomarkers in Clinical Trials. While traditional endpoints and biomarkers are still necessary for FDA submission and regulation, the advent of digital tools has presented the opportunity to revolutionize the data capture method — specifically, the ability to collect more specific, relevant datapoints or
NEWS | MC10
MC10 products and collaborations are making waves in digital health and tech news. See the latest media coverage and MC10 in the press. RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641CAREERS | MC10
MC10 has given me the rare opportunity to wear many hats, allowing me to take on roles I otherwise may not have been lucky enough to experience. ”. “It’s definitely a huge culture shift in a good way for me, from a strict corporate environment to a fun, collaborative workplace. Everyone works together really well.”.CONTACT US | MC10
MC10, Inc. 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421 Phone: (857) 214-5600 Fax: (781) 538-6641 MAP IT. Press Contact press@mc10inc.com. Sales sales@mc10inc.comEVENTS | MC10
10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PRESS & MEDIA
MC10, Inc. Announces Collaboration with Northwestern University and Ann & Robert H. Lurie Children’s Hospital of Chicago: Innovating Precision Medicine Approaches to Improve Maternal-Infant Outcomes. Lexington, MA – MC10, Inc., the company that developed the BioStamp nPoint® System, Northwestern University’s Center for Bio-IntegratedTESTIMONIALS
The sensor is unobtrusive, very comfortable to wear, and it reliably collects data for extensive periods. Charging the sensor’s battery and checking the quality of the data being collected is simple. The system appears to be an ideal platform for many of our clinical research projects.”.QUALITY POLICY
We will achieve this by maintaining an effective quality system that complies with regulatory requirements, applies customer focused continuous improvement, and ensures MC10 is committed to achieving established quality objectives. MC10, Inc. has been granted ISO 13485:2003 certification for the design, development, manufacture, and SOFTWARE ENGINEER IN TEST Software Engineer in Test Job Description / Intro. We're looking for someone to help us write and deploy quality software across the MC10 platform with a focus on MC10, INC. AND ABBVIE (NYSE:ABBV) ANNOUNCE CLINICAL TRIALS MC10, Inc. and AbbVie (NYSE: ABBV) announced today that the companies are working together on clinical trials designed to explore a range of outcome measures in patients with multiple sclerosis (MS) using the BioStamp nPoint® system. WEARABLE HEALTHCARE TECHNOLOGY & DEVICES MC10’s proprietary BioStamp® system wearable health tech creates comfortable, discreet sensors that can be applied anywhere on the body for targeted data collection. Our wearable sensors are wireless and rechargeable via the included Link Hub. A dedicated mobile phone plus Link Application designed for remote data collection guides users WEARABLE HEALTH SYSTEMS BioStamp nPoint is an FDA 510 (k) cleared medical device designed to collect medical grade, clinical quality bio-metric, physiological and eCOA data in a clinical trial setting. The system collects and processes raw data into recognizable clinical metrics including vital signs, activity/actigraphy and posture classification, sEMG, and sleepOUR STORY | MC10
MC10 products are thin and flexible, and built to stretch, bend and twist seamlessly with our bodies and the world around us. MC10’s software platform consists of a complete end-to-end system with mobile interfaces, cloud storage and analytical tools. Lean, agile software development allows us to create robust systems to support the high RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PUBLICATIONS
To objectively demonstrate the capabilities of MC10’s biometric sensors, we have collected the following articles. We invite you to browse these resources to learn more about the power of MC10’s groundbreaking hardware and software systems as well as the future of healthcare analytics in general. BioStamp ® Technologies.HELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641 REAL WORLD EVIDENCE AND ITS IMPACT ON HEALTHCARE Real World Evidence and Its Impact on Healthcare. Real world evidence, or RWE for short, is the term made famous by the 21st Century Cures Act, a law enacted by the United States Congress in December 2016. Section 3022 of the bill calls for the FDA to establish a program and framework for evaluating the use of RWE, defined as “data regardingSYSTEM - MC10
1 The BioStampRC System is an end-to-end research tool designed for targeted, accurate, and seamless physiological data collection from across the human body. 5 UNIVERSITIES EXPLORING THE POTENTIAL OF DIGITAL HEALTH 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641 WEARABLE HEALTHCARE TECHNOLOGY & DEVICES MC10’s proprietary BioStamp® system wearable health tech creates comfortable, discreet sensors that can be applied anywhere on the body for targeted data collection. Our wearable sensors are wireless and rechargeable via the included Link Hub. A dedicated mobile phone plus Link Application designed for remote data collection guides users WEARABLE HEALTH SYSTEMS BioStamp nPoint is an FDA 510 (k) cleared medical device designed to collect medical grade, clinical quality bio-metric, physiological and eCOA data in a clinical trial setting. The system collects and processes raw data into recognizable clinical metrics including vital signs, activity/actigraphy and posture classification, sEMG, and sleepOUR STORY | MC10
MC10 products are thin and flexible, and built to stretch, bend and twist seamlessly with our bodies and the world around us. MC10’s software platform consists of a complete end-to-end system with mobile interfaces, cloud storage and analytical tools. Lean, agile software development allows us to create robust systems to support the high RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PUBLICATIONS
To objectively demonstrate the capabilities of MC10’s biometric sensors, we have collected the following articles. We invite you to browse these resources to learn more about the power of MC10’s groundbreaking hardware and software systems as well as the future of healthcare analytics in general. BioStamp ® Technologies.HELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641 REAL WORLD EVIDENCE AND ITS IMPACT ON HEALTHCARE Real World Evidence and Its Impact on Healthcare. Real world evidence, or RWE for short, is the term made famous by the 21st Century Cures Act, a law enacted by the United States Congress in December 2016. Section 3022 of the bill calls for the FDA to establish a program and framework for evaluating the use of RWE, defined as “data regardingSYSTEM - MC10
1 The BioStampRC System is an end-to-end research tool designed for targeted, accurate, and seamless physiological data collection from across the human body. 5 UNIVERSITIES EXPLORING THE POTENTIAL OF DIGITAL HEALTH 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641NEWS | MC10
MC10 products and collaborations are making waves in digital health and tech news. See the latest media coverage and MC10 in the press. RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641CONTACT US | MC10
MC10, Inc. 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421 Phone: (857) 214-5600 Fax: (781) 538-6641 MAP IT. Press Contact press@mc10inc.com. Sales sales@mc10inc.comEVENTS | MC10
10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641CAREERS | MC10
MC10 has given me the rare opportunity to wear many hats, allowing me to take on roles I otherwise may not have been lucky enough to experience. ”. “It’s definitely a huge culture shift in a good way for me, from a strict corporate environment to a fun, collaborative workplace. Everyone works together really well.”. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PRESS & MEDIA
MC10, Inc. Announces Collaboration with Northwestern University and Ann & Robert H. Lurie Children’s Hospital of Chicago: Innovating Precision Medicine Approaches to Improve Maternal-Infant Outcomes. Lexington, MA – MC10, Inc., the company that developed the BioStamp nPoint® System, Northwestern University’s Center for Bio-IntegratedTESTIMONIALS
The sensor is unobtrusive, very comfortable to wear, and it reliably collects data for extensive periods. Charging the sensor’s battery and checking the quality of the data being collected is simple. The system appears to be an ideal platform for many of our clinical research projects.”.QUALITY POLICY
We will achieve this by maintaining an effective quality system that complies with regulatory requirements, applies customer focused continuous improvement, and ensures MC10 is committed to achieving established quality objectives. MC10, Inc. has been granted ISO 13485:2003 certification for the design, development, manufacture, and SOFTWARE ENGINEER IN TEST Software Engineer in Test Job Description / Intro. We're looking for someone to help us write and deploy quality software across the MC10 platform with a focus on WEARABLE HEALTHCARE TECHNOLOGY & DEVICES MC10’s proprietary BioStamp® system wearable health tech creates comfortable, discreet sensors that can be applied anywhere on the body for targeted data collection. Our wearable sensors are wireless and rechargeable via the included Link Hub. A dedicated mobile phone plus Link Application designed for remote data collection guides users WEARABLE HEALTH SYSTEMS BioStamp nPoint is an FDA 510 (k) cleared medical device designed to collect medical grade, clinical quality bio-metric, physiological and eCOA data in a clinical trial setting. The system collects and processes raw data into recognizable clinical metrics including vital signs, activity/actigraphy and posture classification, sEMG, and sleepOUR STORY | MC10
MC10 products are thin and flexible, and built to stretch, bend and twist seamlessly with our bodies and the world around us. MC10’s software platform consists of a complete end-to-end system with mobile interfaces, cloud storage and analytical tools. Lean, agile software development allows us to create robust systems to support the high RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PUBLICATIONS
To objectively demonstrate the capabilities of MC10’s biometric sensors, we have collected the following articles. We invite you to browse these resources to learn more about the power of MC10’s groundbreaking hardware and software systems as well as the future of healthcare analytics in general. BioStamp ® Technologies.HELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641 REAL WORLD EVIDENCE AND ITS IMPACT ON HEALTHCARE Real World Evidence and Its Impact on Healthcare. Real world evidence, or RWE for short, is the term made famous by the 21st Century Cures Act, a law enacted by the United States Congress in December 2016. Section 3022 of the bill calls for the FDA to establish a program and framework for evaluating the use of RWE, defined as “data regardingSYSTEM - MC10
1 The BioStampRC System is an end-to-end research tool designed for targeted, accurate, and seamless physiological data collection from across the human body. USING DIGITAL ENDPOINTS AND DIGITAL BIOMARKERS IN CLINICAL Using Digital Endpoints and Digital Biomarkers in Clinical Trials. While traditional endpoints and biomarkers are still necessary for FDA submission and regulation, the advent of digital tools has presented the opportunity to revolutionize the data capture method — specifically, the ability to collect more specific, relevant datapoints or
WEARABLE HEALTHCARE TECHNOLOGY & DEVICES MC10’s proprietary BioStamp® system wearable health tech creates comfortable, discreet sensors that can be applied anywhere on the body for targeted data collection. Our wearable sensors are wireless and rechargeable via the included Link Hub. A dedicated mobile phone plus Link Application designed for remote data collection guides users WEARABLE HEALTH SYSTEMS BioStamp nPoint is an FDA 510 (k) cleared medical device designed to collect medical grade, clinical quality bio-metric, physiological and eCOA data in a clinical trial setting. The system collects and processes raw data into recognizable clinical metrics including vital signs, activity/actigraphy and posture classification, sEMG, and sleepOUR STORY | MC10
MC10 products are thin and flexible, and built to stretch, bend and twist seamlessly with our bodies and the world around us. MC10’s software platform consists of a complete end-to-end system with mobile interfaces, cloud storage and analytical tools. Lean, agile software development allows us to create robust systems to support the high RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PUBLICATIONS
To objectively demonstrate the capabilities of MC10’s biometric sensors, we have collected the following articles. We invite you to browse these resources to learn more about the power of MC10’s groundbreaking hardware and software systems as well as the future of healthcare analytics in general. BioStamp ® Technologies.HELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641 REAL WORLD EVIDENCE AND ITS IMPACT ON HEALTHCARE Real World Evidence and Its Impact on Healthcare. Real world evidence, or RWE for short, is the term made famous by the 21st Century Cures Act, a law enacted by the United States Congress in December 2016. Section 3022 of the bill calls for the FDA to establish a program and framework for evaluating the use of RWE, defined as “data regardingSYSTEM - MC10
1 The BioStampRC System is an end-to-end research tool designed for targeted, accurate, and seamless physiological data collection from across the human body. USING DIGITAL ENDPOINTS AND DIGITAL BIOMARKERS IN CLINICAL Using Digital Endpoints and Digital Biomarkers in Clinical Trials. While traditional endpoints and biomarkers are still necessary for FDA submission and regulation, the advent of digital tools has presented the opportunity to revolutionize the data capture method — specifically, the ability to collect more specific, relevant datapoints or
NEWS | MC10
MC10 products and collaborations are making waves in digital health and tech news. See the latest media coverage and MC10 in the press. RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641CONTACT US | MC10
MC10, Inc. 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421 Phone: (857) 214-5600 Fax: (781) 538-6641 MAP IT. Press Contact press@mc10inc.com. Sales sales@mc10inc.comEVENTS | MC10
10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641CAREERS | MC10
MC10 has given me the rare opportunity to wear many hats, allowing me to take on roles I otherwise may not have been lucky enough to experience. ”. “It’s definitely a huge culture shift in a good way for me, from a strict corporate environment to a fun, collaborative workplace. Everyone works together really well.”. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PRESS & MEDIA
MC10, Inc. Announces Collaboration with Northwestern University and Ann & Robert H. Lurie Children’s Hospital of Chicago: Innovating Precision Medicine Approaches to Improve Maternal-Infant Outcomes. Lexington, MA – MC10, Inc., the company that developed the BioStamp nPoint® System, Northwestern University’s Center for Bio-IntegratedTESTIMONIALS
The sensor is unobtrusive, very comfortable to wear, and it reliably collects data for extensive periods. Charging the sensor’s battery and checking the quality of the data being collected is simple. The system appears to be an ideal platform for many of our clinical research projects.”.QUALITY POLICY
We will achieve this by maintaining an effective quality system that complies with regulatory requirements, applies customer focused continuous improvement, and ensures MC10 is committed to achieving established quality objectives. MC10, Inc. has been granted ISO 13485:2003 certification for the design, development, manufacture, and SOFTWARE ENGINEER IN TEST Software Engineer in Test Job Description / Intro. We're looking for someone to help us write and deploy quality software across the MC10 platform with a focus on WEARABLE HEALTHCARE TECHNOLOGY & DEVICES MC10’s proprietary BioStamp® system wearable health tech creates comfortable, discreet sensors that can be applied anywhere on the body for targeted data collection. Our wearable sensors are wireless and rechargeable via the included Link Hub. A dedicated mobile phone plus Link Application designed for remote data collection guides users WEARABLE HEALTH SYSTEMS BioStamp nPoint is an FDA 510 (k) cleared medical device designed to collect medical grade, clinical quality bio-metric, physiological and eCOA data in a clinical trial setting. The system collects and processes raw data into recognizable clinical metrics including vital signs, activity/actigraphy and posture classification, sEMG, and sleepOUR STORY | MC10
MC10 products are thin and flexible, and built to stretch, bend and twist seamlessly with our bodies and the world around us. MC10’s software platform consists of a complete end-to-end system with mobile interfaces, cloud storage and analytical tools. Lean, agile software development allows us to create robust systems to support the high RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641EVENTS | MC10
10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PUBLICATIONS
To objectively demonstrate the capabilities of MC10’s biometric sensors, we have collected the following articles. We invite you to browse these resources to learn more about the power of MC10’s groundbreaking hardware and software systems as well as the future of healthcare analytics in general. BioStamp ® Technologies.HELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641 REAL WORLD EVIDENCE AND ITS IMPACT ON HEALTHCARE Real World Evidence and Its Impact on Healthcare. Real world evidence, or RWE for short, is the term made famous by the 21st Century Cures Act, a law enacted by the United States Congress in December 2016. Section 3022 of the bill calls for the FDA to establish a program and framework for evaluating the use of RWE, defined as “data regarding 5 UNIVERSITIES EXPLORING THE POTENTIAL OF DIGITAL HEALTH 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641 WEARABLE HEALTHCARE TECHNOLOGY & DEVICES MC10’s proprietary BioStamp® system wearable health tech creates comfortable, discreet sensors that can be applied anywhere on the body for targeted data collection. Our wearable sensors are wireless and rechargeable via the included Link Hub. A dedicated mobile phone plus Link Application designed for remote data collection guides users WEARABLE HEALTH SYSTEMS BioStamp nPoint is an FDA 510 (k) cleared medical device designed to collect medical grade, clinical quality bio-metric, physiological and eCOA data in a clinical trial setting. The system collects and processes raw data into recognizable clinical metrics including vital signs, activity/actigraphy and posture classification, sEMG, and sleepOUR STORY | MC10
MC10 products are thin and flexible, and built to stretch, bend and twist seamlessly with our bodies and the world around us. MC10’s software platform consists of a complete end-to-end system with mobile interfaces, cloud storage and analytical tools. Lean, agile software development allows us to create robust systems to support the high RESOURCES | WEARABLE HEALTHCARE TECHNOLOGY | MC10 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641EVENTS | MC10
10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641PUBLICATIONS
To objectively demonstrate the capabilities of MC10’s biometric sensors, we have collected the following articles. We invite you to browse these resources to learn more about the power of MC10’s groundbreaking hardware and software systems as well as the future of healthcare analytics in general. BioStamp ® Technologies.HELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641 REAL WORLD EVIDENCE AND ITS IMPACT ON HEALTHCARE Real World Evidence and Its Impact on Healthcare. Real world evidence, or RWE for short, is the term made famous by the 21st Century Cures Act, a law enacted by the United States Congress in December 2016. Section 3022 of the bill calls for the FDA to establish a program and framework for evaluating the use of RWE, defined as “data regarding 5 UNIVERSITIES EXPLORING THE POTENTIAL OF DIGITAL HEALTH 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641NEWS | MC10
MC10 products and collaborations are making waves in digital health and tech news. See the latest media coverage and MC10 in the press.CONTACT US | MC10
MC10, Inc. 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421 Phone: (857) 214-5600 Fax: (781) 538-6641 MAP IT. Press Contact press@mc10inc.com. Sales sales@mc10inc.comHELP AND SUPPORT
Help and Support. If you have questions about using an MC10 product, or you are looking for customer support, please send a message to support@mc10inc.com, or give our support team a call at 857-214-5630. We will be happy to help.CAREERS | MC10
MC10 has given me the rare opportunity to wear many hats, allowing me to take on roles I otherwise may not have been lucky enough to experience. ”. “It’s definitely a huge culture shift in a good way for me, from a strict corporate environment to a fun, collaborative workplace. Everyone works together really well.”. INSTRUCTIONS FOR USE 10 Maguire Road Building 3, 1st Floor Lexington, MA 02421. info@mc10inc.com phone: (857) 214-5600 fax: (781) 538-6641MC10 PULSE | MC10
The Clinical Trials Transformation Initiative (CTTI) is comprised of more than 80 organizations united by the mission to develop and drive adoption of practices that will increase the quality and efficiency of clinical trials.. The multi-stakeholder member organizations includes representatives from the FDA, big pharma, smaller biotechs, clinical research organizations (CROs), institutionalFAQS | MC10
Each sensor contains a real-time clock, backed by a quartz oscillator, that is set at the beginning of a recording session. MC10 calibrates the sensors to be accurate within 1 second over a 24-hour period. Raw data samples are time stamped as they are created and stored in the Sensors flash memory.TESTIMONIALS
The sensor is unobtrusive, very comfortable to wear, and it reliably collects data for extensive periods. Charging the sensor’s battery and checking the quality of the data being collected is simple. The system appears to be an ideal platform for many of our clinical research projects.”. MOBILE SOFTWARE DEVELOPER BS/MS degree in Computer Science with 3+ years of mobile software development or related work experience. MC10’s goal is to redefine the interface between electronics and the human body. We extend human capabilities through virtually invisible and conformal electronics. We reshape rigid, high performance electronics into human compatible formSYSTEM - MC10
1 The BioStampRC System is an end-to-end research tool designed for targeted, accurate, and seamless physiological data collection from across the human body.__
* Products
* Resources
__
* Publications
* Events
* News
* Press & Media
* Instructions for Use * Frequently Asked Questions* Support
* Blog
* Contact
*
BioStamp nPoint Login__
* Products
* Resources
__
* Publications
* Events
* News
* Press & Media
* Instructions for Use * Frequently Asked Questions* Support
* Blog
* Contact
*
BioStamp RC Login
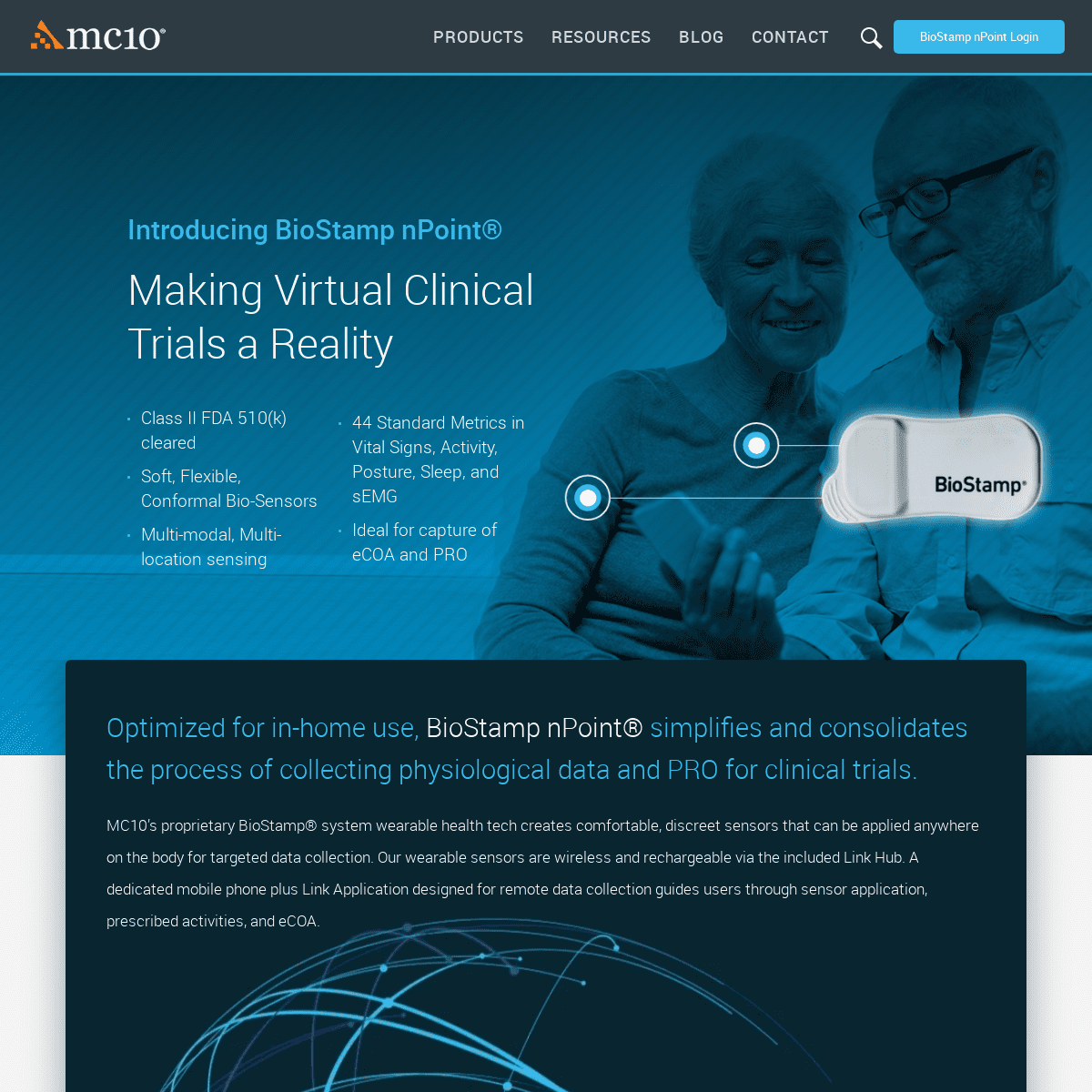
Introducing BioStamp nPoint® MAKING VIRTUAL CLINICAL TRIALS A REALITY * Class II FDA 510(k) cleared * Soft, Flexible, Conformal Bio-Sensors * Multi-modal, Multi-location sensing * 44 Standard Metrics in Vital Signs, Activity, Posture, Sleep, andsEMG
* Ideal for capture of eCOA and PRO OPTIMIZED FOR IN-HOME USE, BIOSTAMP NPOINT® SIMPLIFIES AND CONSOLIDATES THE PROCESS OF COLLECTING PHYSIOLOGICAL DATA AND PRO FORCLINICAL TRIALS.
MC10’s proprietary BioStamp® system wearable health tech creates comfortable, discreet sensors that can be applied anywhere on the body for targeted data collection. Our wearable sensors are wireless and rechargeable via the included Link Hub. A dedicated mobile phone plus Link Application designed for remote data collection guides users through sensor application, prescribed activities, and eCOA. Your browser does not support HTML5 video. FLEXIBILITY BEYOND THE SENSOR * Multiple wear locations * Configurable sampling rate/dynamic range * Synchronized multi-sensor data stream * Processed metrics and raw data * Versatile eCOA editor MUSCLE REGION PLACEMENT With location-specific sensing capabilities, BioStamp sensors are able to retrieve real-world data about muscle activation and movement. Fully integrated hydrogel adhesives enable high fidelity sEMG. BODY REGION PLACEMENT BioStamp sensors are supported by 25 validated wear locations on the body (hand and foot not shown). Multiple sensors can be worn simultaneously to provide more additional data streams for clinical trial endpoints. MUSCLE REGION PLACEMENT With location-specific sensing capabilities, BioStamp sensors are able to retrieve real-world data about muscle activation and movement. Fully integrated hydrogel adhesives enable high fidelity sEMG. BODY REGION PLACEMENT BioStamp sensors are supported by 25 validated wear locations on the body (hand and foot not shown). Multiple sensors can be worn simultaneously to provide more additional data streams for clinical trial endpoints. DELIVERING ACTIONABLE INSIGHTS THROUGH BIOMETRICS BioStamp nPoint's proprietary, clinically validated algorithmic interpretation of raw physiological data delivers clinical insight into vital signs, activity, posture, sleep, and the relationshipsamong them.
SLEEP
Assesses sleep parameters including onset, wakefulness, duration and posture transitions.POSTURE
Quantifies time spent in various body positions including laying down, sitting, standing, or leaning.ACTIVITY
Quantifies activity units, time spent active, step count, and cadence.VITAL SIGNS
Collects continuous HR and HRV (in both time and frequency domains) data and respiration rate during sleep.SLEEP
Assesses sleep parameters including onset, wakefulness, duration and posture transitions.POSTURE
Quantifies time spent in various body positions including laying down, sitting, standing, or leaning.ACTIVITY
Quantifies activity units, time spent active, step count, and cadence.VITAL SIGNS
Collects continuous HR and HRV (in both time and frequency domains) data and respiration rate during sleep. END-TO-END SYSTEM DESIGNED FOR IN-HOME AND IN-CLINIC STUDY EXECUTIONINVESTIGATOR PORTAL
Web portal for researchers to design and configure their study, enroll subjects, monitor trial progress and view / extract raw data andprocessed metrics.
INVESTIGATOR APP
Investigator-facing tablet app to assign sensors to subjects and view streaming data in clinical environments.LINK APP
Subject-facing smartphone app to guide study subjects through sensor application, study activities, eCOA completion and enable communication with the research team.LINK HUB
In-home, wireless charging and data syncing station for the BioStamp Sensors and dedicated mobile phone. RECENT MC10 PUBLICATIONS Using A Soft Conformable Foot Sensor to Measure Changes in Foot Strike Angle During Running Clinical feasibility of a wearable, conformable sensor patch to monitor motor symptoms in Parkinson's disease (2019) A Pivotal Study to Validate the Performance of a Novel Wearable Sensor and System for Biometric Monitoring in Clinical and RemoteEnvironments
Assessment of Postural Sway in Individuals with Multiple Sclerosis Using a Novel Wearable Inertial Sensor LATEST BLOG ARTICLES THURSDAY, DECEMBER 19, 2019 MC10 Researcher Spotlight: Dr. Ryan McGinnis – University ofVermont
THURSDAY, DECEMBER 12, 2019 Benefits of Digital Health Technology for Seniors MONDAY, MARCH 4, 2019 How Measurement Can Improve Medicine THURSDAY, OCTOBER 5, 2017 University of Rochester Researchers Use BioStampRC® Sensors to Assess Parkinson and Huntington Disease SymptomsIntroducing
Begin unlocking the right data from your subjects home today!10 Maguire Road
Building 3, 1st FloorLexington, MA 02421
info@mc10inc.com
phone: (857) 214-5600fax: (781) 538-6641
FOLLOW US
* __
* __
* __
FDA 510(K) CLEARED CLASS II MEDICAL DEVICEGET PRODUCT UPDATES
� 2021 MC10. All rights reservedDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0