Are you over 18 and want to see adult content?
More Annotations

A complete backup of jumptolisting.com
Are you over 18 and want to see adult content?

A complete backup of coloringpagesabc.com
Are you over 18 and want to see adult content?
A complete backup of truereligion-jeans.org.uk
Are you over 18 and want to see adult content?

A complete backup of tetote-market.jp
Are you over 18 and want to see adult content?

A complete backup of supertechlimited.com
Are you over 18 and want to see adult content?
Favourite Annotations
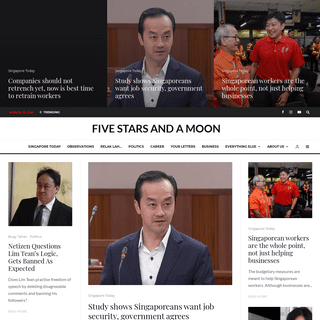
A complete backup of fivestarsandamoon.com
Are you over 18 and want to see adult content?

A complete backup of taiyoakari.tumblr.com
Are you over 18 and want to see adult content?
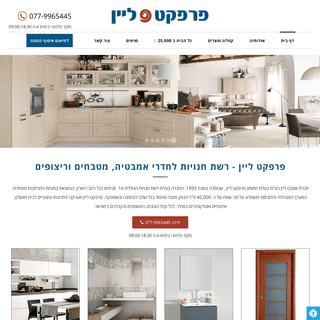
A complete backup of perfectline.co.il
Are you over 18 and want to see adult content?
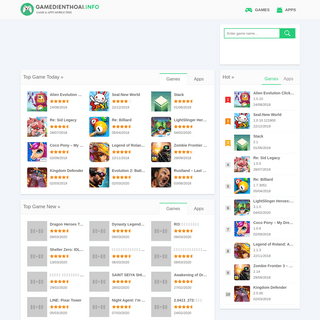
A complete backup of gamedienthoai.info
Are you over 18 and want to see adult content?
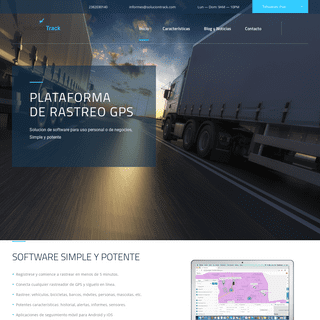
A complete backup of soluciontrack.com
Are you over 18 and want to see adult content?
A complete backup of supershareware.com
Are you over 18 and want to see adult content?
Text
autoimmune disease
LOG IN | IMMUNE TOLERANCE NETWORK The Immune Tolerance Network and is sponsored by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. ABOUT US | IMMUNE TOLERANCE NETWORK About Us. The Immune Tolerance Network (ITN) is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. Our mission is to accelerate the clinical development of immune tolerance therapies. YouTube. IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
Clinical Trials. Building on clinical trial and laboratory research insights from the past decade, the ITN has focused and deliberate strategies to achieving tolerance in each disease area. The research supported by the ITN has unlocked new therapeutic approaches and discovered new biomarkers that promise to change the way physicianstreat
IMMUNE TOLERANCE IN TRANSPLANTATION This means that people taking immunosuppressive drugs are at risk of developing serious infections. The Immune Tolerance Network (ITN) is working to find new therapies that reprogram the immune system in a way that will allow people to maintain the health of the organ without long-term use of immunosuppression, also known as tolerance. IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
VAGINAL MICROBIOME EXPOSURE AND IMMUNE RESPONSES IN C ACTIVATE is a pilot study that will investigate how differences in the microbiome of a baby may protect, or put them at risk, for allergies. The study will measure whether wiping babies born by C-section with their mother’s vaginal fluids (which contains potentially beneficial bacteria; referred to as “vaginal seeding”) right after birth will lower the risk of developing positive allergy CLINICAL TRIAL INFORMATION Thank you for your interest in the RAVE Trial. RAVE is an international Phase II/III double-blind, placebo-controlled trial of the anti-CD20 monoclonal antibody rituximab in ANCA-associated vasculitis (AAV), specifically Wegener’s Granulomatosis or Microscopic Polyangiitis.The trial is being conducted by the Immune Tolerance Network (ITN), with support from the National Institutes ofHealth
WE’RE ADVANCING THE CLINICAL APPLICATION OF IMMUNEABOUT USFOR RESEARCHERSFOR PATIENTSTRIALSHARECONTACTCORE FACILITIES The Immune Tolerance Network is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. The ITN is focused on the development of therapeutic approaches for asthma and allergy, autoimmune diseases, type 1 diabetes and solid organ IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE The Immune Tolerance Network (ITN) is also working to identify biomarkers of immune tolerance that may help to identify the right treatment course for future patients. This section contains a list of ITN's autoimmune clinical trials that are currently enrolling participants. To see all of ITN's active and completed studies inautoimmune disease
LOG IN | IMMUNE TOLERANCE NETWORK The Immune Tolerance Network and is sponsored by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. ABOUT US | IMMUNE TOLERANCE NETWORK About Us. The Immune Tolerance Network (ITN) is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. Our mission is to accelerate the clinical development of immune tolerance therapies. YouTube. IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
Clinical Trials. Building on clinical trial and laboratory research insights from the past decade, the ITN has focused and deliberate strategies to achieving tolerance in each disease area. The research supported by the ITN has unlocked new therapeutic approaches and discovered new biomarkers that promise to change the way physicianstreat
IMMUNE TOLERANCE IN TRANSPLANTATION This means that people taking immunosuppressive drugs are at risk of developing serious infections. The Immune Tolerance Network (ITN) is working to find new therapies that reprogram the immune system in a way that will allow people to maintain the health of the organ without long-term use of immunosuppression, also known as tolerance. IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
VAGINAL MICROBIOME EXPOSURE AND IMMUNE RESPONSES IN C ACTIVATE is a pilot study that will investigate how differences in the microbiome of a baby may protect, or put them at risk, for allergies. The study will measure whether wiping babies born by C-section with their mother’s vaginal fluids (which contains potentially beneficial bacteria; referred to as “vaginal seeding”) right after birth will lower the risk of developing positive allergy CLINICAL TRIAL INFORMATION Thank you for your interest in the RAVE Trial. RAVE is an international Phase II/III double-blind, placebo-controlled trial of the anti-CD20 monoclonal antibody rituximab in ANCA-associated vasculitis (AAV), specifically Wegener’s Granulomatosis or Microscopic Polyangiitis.The trial is being conducted by the Immune Tolerance Network (ITN), with support from the National Institutes ofHealth
LATEST NEWS
The Immune Tolerance Network (ITN) has opened the BEAT-MS clinical trial for enrollment. BEAT-MS or “Best Available Therapy vs. Autologous Hematopoietic Stem Cell Transplant (AHSCT) for MS” will investigate high dose immunosuppression followed by AHSCT – a type of transplant using a patient’s own immune stem cells – compared to the best medical treatment currently available for IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
The ITN conducts clinical trials with the goal of achieving immune tolerance for patients. To see a list of ITN's clinical trials that are currently enrolling participants, please select a disease area: Allergy & Asthma Autoimmune Disease Transplantation Type 1 Diabetes. To see all of ITN's active and completed studies, please visit ForResearchers.
MISSION & VALUES
Mission & Values. The Immune Tolerance Network (ITN) is an organization born from the collective vision of the National Institute of Allergy and Infectious Disease (of the National Institutes of Health) and the tolerance research community. As such, our vision and the principles upon which we were founded are a reflection of thecommunity itself.
IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Among the different autoimmune disorders within the Immune Tolerance Network's (ITN’s) portfolio, several studies focus on early intervention of type 1 diabetes (T1D), also called autoimmune diabetes, juvenile diabetes or insulin-dependent diabetes. This chronic inflammatory diseaseprimarily affects
PROPOSAL GUIDELINES
The ITN application process is a two-stage process designed to fast-track applications from proposal to protocol development. Applicants submit a short (~5-page) Concept Proposal containing a basic outline and justification of the proposed research. Applicants whose proposals are of interest to the ITN will then be asked tosubmit a more
LIVER TRANSPLANTATION WITH TREGS AT MGH AND UCSF (LITTMUS LITTMUS is a clinical research study testing a new approach to achieve transplant tolerance using the liver transplant recipient’s own T regulatory cells (Tregs). Tregs are normally present in very low numbers and act as the brakes on the immune system. In this study, we will take cells from the recipient’s blood and cells from the living AUTOIMMUNITY-BLOCKING ANTIBODY FOR TOLERANCE IN RECENTLY Objective: The objective of this proposal is to study the immunologic effects of humanized FcR non-binding anti-CD3 mAb on immune responses associated with Type 1 diabetes (T1DM), and develop this therapy to prevent the immune destruction leading to beta cell loss. Basis/Rationale: Studies of the natural history of T1DM indicate that 100% of individuals with the disease still make detectable ABATACEPT FOR LUPUS NEPHRITIS (THE ACCESS STUDY) The results of the ACCESS study, “Abatacept and Cyclophosphamide Combination Therapy for Lupus Nephritis” sponsored by the Immune Tolerance Network (ITN) and led by Drs. Betty Diamond (The Feinstein Institute) and David Wofsy (University of California, San Francisco), were published this week in Arthritis and Rheumatology. Abatacept (ORENCIA ®) in combination with low-dose WE’RE ADVANCING THE CLINICAL APPLICATION OF IMMUNEABOUT USFOR RESEARCHERSFOR PATIENTSTRIALSHARECONTACTCORE FACILITIES The Immune Tolerance Network is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. The ITN is focused on the development of therapeutic approaches for asthma and allergy, autoimmune diseases, type 1 diabetes and solid organ IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE The Immune Tolerance Network (ITN) is also working to identify biomarkers of immune tolerance that may help to identify the right treatment course for future patients. This section contains a list of ITN's autoimmune clinical trials that are currently enrolling participants. To see all of ITN's active and completed studies inautoimmune disease
LOG IN | IMMUNE TOLERANCE NETWORK The Immune Tolerance Network and is sponsored by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. ABOUT US | IMMUNE TOLERANCE NETWORK About Us. The Immune Tolerance Network (ITN) is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. Our mission is to accelerate the clinical development of immune tolerance therapies. YouTube. IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
Clinical Trials. Building on clinical trial and laboratory research insights from the past decade, the ITN has focused and deliberate strategies to achieving tolerance in each disease area. The research supported by the ITN has unlocked new therapeutic approaches and discovered new biomarkers that promise to change the way physicianstreat
IMMUNE TOLERANCE IN TRANSPLANTATION This means that people taking immunosuppressive drugs are at risk of developing serious infections. The Immune Tolerance Network (ITN) is working to find new therapies that reprogram the immune system in a way that will allow people to maintain the health of the organ without long-term use of immunosuppression, also known as tolerance. IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
VAGINAL MICROBIOME EXPOSURE AND IMMUNE RESPONSES IN C ACTIVATE is a pilot study that will investigate how differences in the microbiome of a baby may protect, or put them at risk, for allergies. The study will measure whether wiping babies born by C-section with their mother’s vaginal fluids (which contains potentially beneficial bacteria; referred to as “vaginal seeding”) right after birth will lower the risk of developing positive allergy CLINICAL TRIAL INFORMATION Thank you for your interest in the RAVE Trial. RAVE is an international Phase II/III double-blind, placebo-controlled trial of the anti-CD20 monoclonal antibody rituximab in ANCA-associated vasculitis (AAV), specifically Wegener’s Granulomatosis or Microscopic Polyangiitis.The trial is being conducted by the Immune Tolerance Network (ITN), with support from the National Institutes ofHealth
WE’RE ADVANCING THE CLINICAL APPLICATION OF IMMUNEABOUT USFOR RESEARCHERSFOR PATIENTSTRIALSHARECONTACTCORE FACILITIES The Immune Tolerance Network is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. The ITN is focused on the development of therapeutic approaches for asthma and allergy, autoimmune diseases, type 1 diabetes and solid organ IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE The Immune Tolerance Network (ITN) is also working to identify biomarkers of immune tolerance that may help to identify the right treatment course for future patients. This section contains a list of ITN's autoimmune clinical trials that are currently enrolling participants. To see all of ITN's active and completed studies inautoimmune disease
LOG IN | IMMUNE TOLERANCE NETWORK The Immune Tolerance Network and is sponsored by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. ABOUT US | IMMUNE TOLERANCE NETWORK About Us. The Immune Tolerance Network (ITN) is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. Our mission is to accelerate the clinical development of immune tolerance therapies. YouTube. IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
Clinical Trials. Building on clinical trial and laboratory research insights from the past decade, the ITN has focused and deliberate strategies to achieving tolerance in each disease area. The research supported by the ITN has unlocked new therapeutic approaches and discovered new biomarkers that promise to change the way physicianstreat
IMMUNE TOLERANCE IN TRANSPLANTATION This means that people taking immunosuppressive drugs are at risk of developing serious infections. The Immune Tolerance Network (ITN) is working to find new therapies that reprogram the immune system in a way that will allow people to maintain the health of the organ without long-term use of immunosuppression, also known as tolerance. IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
VAGINAL MICROBIOME EXPOSURE AND IMMUNE RESPONSES IN C ACTIVATE is a pilot study that will investigate how differences in the microbiome of a baby may protect, or put them at risk, for allergies. The study will measure whether wiping babies born by C-section with their mother’s vaginal fluids (which contains potentially beneficial bacteria; referred to as “vaginal seeding”) right after birth will lower the risk of developing positive allergy CLINICAL TRIAL INFORMATION Thank you for your interest in the RAVE Trial. RAVE is an international Phase II/III double-blind, placebo-controlled trial of the anti-CD20 monoclonal antibody rituximab in ANCA-associated vasculitis (AAV), specifically Wegener’s Granulomatosis or Microscopic Polyangiitis.The trial is being conducted by the Immune Tolerance Network (ITN), with support from the National Institutes ofHealth
LATEST NEWS
The Immune Tolerance Network (ITN) has opened the BEAT-MS clinical trial for enrollment. BEAT-MS or “Best Available Therapy vs. Autologous Hematopoietic Stem Cell Transplant (AHSCT) for MS” will investigate high dose immunosuppression followed by AHSCT – a type of transplant using a patient’s own immune stem cells – compared to the best medical treatment currently available for IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
The ITN conducts clinical trials with the goal of achieving immune tolerance for patients. To see a list of ITN's clinical trials that are currently enrolling participants, please select a disease area: Allergy & Asthma Autoimmune Disease Transplantation Type 1 Diabetes. To see all of ITN's active and completed studies, please visit ForResearchers.
IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
MISSION & VALUES
Mission & Values. The Immune Tolerance Network (ITN) is an organization born from the collective vision of the National Institute of Allergy and Infectious Disease (of the National Institutes of Health) and the tolerance research community. As such, our vision and the principles upon which we were founded are a reflection of thecommunity itself.
IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Among the different autoimmune disorders within the Immune Tolerance Network's (ITN’s) portfolio, several studies focus on early intervention of type 1 diabetes (T1D), also called autoimmune diabetes, juvenile diabetes or insulin-dependent diabetes. This chronic inflammatory diseaseprimarily affects
PROPOSAL GUIDELINES
The ITN application process is a two-stage process designed to fast-track applications from proposal to protocol development. Applicants submit a short (~5-page) Concept Proposal containing a basic outline and justification of the proposed research. Applicants whose proposals are of interest to the ITN will then be asked tosubmit a more
LIVER TRANSPLANTATION WITH TREGS AT MGH AND UCSF (LITTMUS LITTMUS is a clinical research study testing a new approach to achieve transplant tolerance using the liver transplant recipient’s own T regulatory cells (Tregs). Tregs are normally present in very low numbers and act as the brakes on the immune system. In this study, we will take cells from the recipient’s blood and cells from the living AUTOIMMUNITY-BLOCKING ANTIBODY FOR TOLERANCE IN RECENTLY Linsley PS, Greenbaum CJ, Rosasco M, Presnell S, Herold KC, Dufort MJ. (2019) Elevated T cell levels in peripheral blood predict poor clinical response following rituximab treatment in new-onset type 1 diabetes. Genes Immun, 20 (4), 293-307. DOI: 10.1038/s41435-018-0032-1 PMID: 29925930 PMCID: PMC6477779 CRACKING THE PEANUT ALLERGY PROBLEM In peanut allergy, a protein (or combination of proteins) in the peanuts triggers the immune system to create an antibody called IgE (immunoglobulin E). In turn, IgE triggers allergic reactions that can range from hives and sneezing to vomiting, diarrhea, choking, gagging, and occasionally anaphylaxis, a severe allergic reaction that can befatal.
WE’RE ADVANCING THE CLINICAL APPLICATION OF IMMUNEABOUT USFOR RESEARCHERSFOR PATIENTSTRIALSHARECONTACTCORE FACILITIES The Immune Tolerance Network is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. The ITN is focused on the development of therapeutic approaches for asthma and allergy, autoimmune diseases, type 1 diabetes and solid organ IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE The Immune Tolerance Network (ITN) is also working to identify biomarkers of immune tolerance that may help to identify the right treatment course for future patients. This section contains a list of ITN's autoimmune clinical trials that are currently enrolling participants. To see all of ITN's active and completed studies inautoimmune disease
ABOUT US | IMMUNE TOLERANCE NETWORK About Us. The Immune Tolerance Network (ITN) is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. Our mission is to accelerate the clinical development of immune tolerance therapies. YouTube. LOG IN | IMMUNE TOLERANCE NETWORK The Immune Tolerance Network and is sponsored by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
The ITN conducts clinical trials with the goal of achieving immune tolerance for patients. To see a list of ITN's clinical trials that are currently enrolling participants, please select a disease area: Allergy & Asthma Autoimmune Disease Transplantation Type 1 Diabetes. To see all of ITN's active and completed studies, please visit ForResearchers.
IMMUNE TOLERANCE IN TRANSPLANTATION This means that people taking immunosuppressive drugs are at risk of developing serious infections. The Immune Tolerance Network (ITN) is working to find new therapies that reprogram the immune system in a way that will allow people to maintain the health of the organ without long-term use of immunosuppression, also known as tolerance. IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
VAGINAL MICROBIOME EXPOSURE AND IMMUNE RESPONSES IN C ACTIVATE is a pilot study that will investigate how differences in the microbiome of a baby may protect, or put them at risk, for allergies. The study will measure whether wiping babies born by C-section with their mother’s vaginal fluids (which contains potentially beneficial bacteria; referred to as “vaginal seeding”) right after birth will lower the risk of developing positive allergy CLINICAL TRIAL INFORMATION Thank you for your interest in the RAVE Trial. RAVE is an international Phase II/III double-blind, placebo-controlled trial of the anti-CD20 monoclonal antibody rituximab in ANCA-associated vasculitis (AAV), specifically Wegener’s Granulomatosis or Microscopic Polyangiitis.The trial is being conducted by the Immune Tolerance Network (ITN), with support from the National Institutes ofHealth
WE’RE ADVANCING THE CLINICAL APPLICATION OF IMMUNEABOUT USFOR RESEARCHERSFOR PATIENTSTRIALSHARECONTACTCORE FACILITIES The Immune Tolerance Network is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. The ITN is focused on the development of therapeutic approaches for asthma and allergy, autoimmune diseases, type 1 diabetes and solid organ IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE The Immune Tolerance Network (ITN) is also working to identify biomarkers of immune tolerance that may help to identify the right treatment course for future patients. This section contains a list of ITN's autoimmune clinical trials that are currently enrolling participants. To see all of ITN's active and completed studies inautoimmune disease
ABOUT US | IMMUNE TOLERANCE NETWORK About Us. The Immune Tolerance Network (ITN) is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. Our mission is to accelerate the clinical development of immune tolerance therapies. YouTube. LOG IN | IMMUNE TOLERANCE NETWORK The Immune Tolerance Network and is sponsored by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
The ITN conducts clinical trials with the goal of achieving immune tolerance for patients. To see a list of ITN's clinical trials that are currently enrolling participants, please select a disease area: Allergy & Asthma Autoimmune Disease Transplantation Type 1 Diabetes. To see all of ITN's active and completed studies, please visit ForResearchers.
IMMUNE TOLERANCE IN TRANSPLANTATION This means that people taking immunosuppressive drugs are at risk of developing serious infections. The Immune Tolerance Network (ITN) is working to find new therapies that reprogram the immune system in a way that will allow people to maintain the health of the organ without long-term use of immunosuppression, also known as tolerance. IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
VAGINAL MICROBIOME EXPOSURE AND IMMUNE RESPONSES IN C ACTIVATE is a pilot study that will investigate how differences in the microbiome of a baby may protect, or put them at risk, for allergies. The study will measure whether wiping babies born by C-section with their mother’s vaginal fluids (which contains potentially beneficial bacteria; referred to as “vaginal seeding”) right after birth will lower the risk of developing positive allergy CLINICAL TRIAL INFORMATION Thank you for your interest in the RAVE Trial. RAVE is an international Phase II/III double-blind, placebo-controlled trial of the anti-CD20 monoclonal antibody rituximab in ANCA-associated vasculitis (AAV), specifically Wegener’s Granulomatosis or Microscopic Polyangiitis.The trial is being conducted by the Immune Tolerance Network (ITN), with support from the National Institutes ofHealth
LATEST NEWS
The Immune Tolerance Network (ITN) has opened the BEAT-MS clinical trial for enrollment. BEAT-MS or “Best Available Therapy vs. Autologous Hematopoietic Stem Cell Transplant (AHSCT) for MS” will investigate high dose immunosuppression followed by AHSCT – a type of transplant using a patient’s own immune stem cells – compared to the best medical treatment currently available for IMMUNE TOLERANCE IN AUTOIMMUNE DISEASE Immune tolerance therapies are designed to stop, or even prevent, the autoimmune disease while leaving the body's disease-fighting abilities intact. These tolerance therapies essentially reprogram the immune system, so that a short course of treatment will have long-lasting, perhaps lifelong effects. While immune tolerance therapies are mainlyCLINICAL TRIALS
Clinical Trials. Building on clinical trial and laboratory research insights from the past decade, the ITN has focused and deliberate strategies to achieving tolerance in each disease area. The research supported by the ITN has unlocked new therapeutic approaches and discovered new biomarkers that promise to change the way physicianstreat
MISSION & VALUES
Mission & Values. The Immune Tolerance Network (ITN) is an organization born from the collective vision of the National Institute of Allergy and Infectious Disease (of the National Institutes of Health) and the tolerance research community. As such, our vision and the principles upon which we were founded are a reflection of thecommunity itself.
IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Type 1 diabetes (T1D) is a chronic inflammatory disease that develops when the body’s immune system mistakenly attacks the cells that make insulin (called ‘beta cells’). Insulin allows the body to use energy from food and helps control blood sugar levels, and without it the body cannot functionproperly.
TRIALSHARE | IMMUNE TOLERANCE NETWORK TrialShare. ITN TrialShare is a clinical trials research portal that was created to promote the sharing of information about ITN's clinical studies and specimen bio-repository with the scientific research community. TrialShare was developed by ITN with the goal of promoting transparency, reproducibility and scientific collaboration. IMMUNE TOLERANCE AND TYPE 1 DIABETES Immune Tolerance and Type 1 Diabetes. Among the different autoimmune disorders within the Immune Tolerance Network's (ITN’s) portfolio, several studies focus on early intervention of type 1 diabetes (T1D), also called autoimmune diabetes, juvenile diabetes or insulin-dependent diabetes. This chronic inflammatory diseaseprimarily affects
PROPOSAL GUIDELINES
The ITN application process is a two-stage process designed to fast-track applications from proposal to protocol development. Applicants submit a short (~5-page) Concept Proposal containing a basic outline and justification of the proposed research. Applicants whose proposals are of interest to the ITN will then be asked tosubmit a more
AUTOIMMUNITY-BLOCKING ANTIBODY FOR TOLERANCE IN RECENTLY Linsley PS, Greenbaum CJ, Rosasco M, Presnell S, Herold KC, Dufort MJ. (2019) Elevated T cell levels in peripheral blood predict poor clinical response following rituximab treatment in new-onset type 1 diabetes. Genes Immun, 20 (4), 293-307. DOI: 10.1038/s41435-018-0032-1 PMID: 29925930 PMCID: PMC6477779 CRACKING THE PEANUT ALLERGY PROBLEM In peanut allergy, a protein (or combination of proteins) in the peanuts triggers the immune system to create an antibody called IgE (immunoglobulin E). In turn, IgE triggers allergic reactions that can range from hives and sneezing to vomiting, diarrhea, choking, gagging, and occasionally anaphylaxis, a severe allergic reaction that can befatal.
Skip to main contentToggle navigation
Search
Search
Primary links
* About Us »
* About Us
* Mission & Values
* Leadership
* News
* Careers
* Partnerships
* Support
* Achievements
* For Researchers » * Clinical Trials »* Allergy & Asthma
* Autoimmune Disease* Transplantation
* Type 1 Diabetes
* Core Facilities
* Lab Protocols »
* ITN Protocols
* T1D Preclinical Consortium* Policies »
* Publications Policy* Proposals »
* Submit Proposal
* Current RFPs
* Past RFPs
* Publications
* TrialShare
* Strategic Plans »* Allergy
* Autoimmune Disease »* Type 1 Diabetes
* Transplantation
* For Patients »
* Allergy & Asthma
* Autoimmune Disease * Transplantation »* Kidney
* Liver
* Type 1 Diabetes
* Clinical Trial Info* TrialShare
* Contact »
* Contact Us »
* BRI - Seattle Office * UCSF - San Francisco Office* Media Inquiries
* About Us »
* About Us
* Mission & Values
* Leadership
* News
* Careers
* Partnerships
* Support
* Achievements
* For Researchers » * Clinical Trials »* Allergy & Asthma
* Autoimmune Disease* Transplantation
* Type 1 Diabetes
* Core Facilities
* Lab Protocols »
* ITN Protocols
* T1D Preclinical Consortium* Policies »
* Publications Policy* Proposals »
* Submit Proposal
* Current RFPs
* Past RFPs
* Publications
* TrialShare
* Strategic Plans »* Allergy
* Autoimmune Disease »* Type 1 Diabetes
* Transplantation
* For Patients »
* Allergy & Asthma
* Autoimmune Disease * Transplantation »* Kidney
* Liver
* Type 1 Diabetes
* Clinical Trial Info* TrialShare
* Contact »
* Contact Us »
* BRI - Seattle Office * UCSF - San Francisco Office* Media Inquiries
SECONDARY LINKS
* " class="is-active">Home* TrialShare
* Member Portal
Search
Search
* About Us
* About Us
* Mission & Values
* Leadership
* News
* Careers
* Partnerships
* Support
* Achievements
* For Researchers
* Clinical Trials
* Core Facilities
* Lab Protocols
* Policies
* Proposals
* Publications
* TrialShare
* Strategic Plans
* For Patients
* Allergy & Asthma
* Autoimmune Disease* Transplantation
* Type 1 Diabetes
* Clinical Trial Info* TrialShare
* Contact
* Contact Us
* Media Inquiries
Developing, funding & conducting clinical trials in immune tolerance Learn about our research ENROLL IN A CLINICAL TRIALAutoimmune Disease
Autoimmune Disease
ITN is identifying therapies to achieve durable clinical remission ofautoimmune disease
Allergy & Asthma
Allergy & Asthma
ITN is focused on achieving durable desensitization to allergenexposure
Transplantation
Transplantation
ITN's goal is to induce tolerance of transplanted organs without lifelong immunosuppression WE'RE ADVANCING THE CLINICAL APPLICATION OF IMMUNE TOLERANCE. Immune Tolerance NetworkSTRATEGIC PLANS
REQUEST FOR PROPOSAL Clinical trials of desensitization in solid organ transplantationTRIALSHARE
Access raw data, analysis and clinical specimensFEATURED
TEACH: Investigating a treatment to allow kidney transplant recipients to stop immunosuppressive medications. The ACTIVATE Study will look at how the type of delivery may protect, or put a baby at risk, for allergies. BRAVOS is a clincial trial evaluating Brentuximab Vendotin treatment for Diffuse Cutaneous Systemic Sclerosis. The Immune Tolerance Network is a collaborative network for clinical research, funded by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. The ITN is focused on the development of therapeutic approaches for asthma and allergy, autoimmune diseases, type 1 diabetes and solid organ transplantation that lead to immune tolerance. More EXPLORE OUR CLINICAL TRIALS: Allergy & Asthma |Autoimmune Disease
| Transplantation | Type 1 Diabetes | All Clinical TrialsNEWS
Allergy, Asthma and COVID-19 Susceptibility ITN’s CATEEC Article Selected as Editor’s Choice Feature by JACI ITN’s REBOOT Study for Primary Membranous Nephropathy Has FirstPatient Visit
BEAT-MS, a Clinical Trial of Autologous Hematopoietic Stem Cell Transplantation versus Best Available Therapy for Multiple Sclerosis,Begins Enrollment
View All
The Immune Tolerance Network and is sponsored by the National Institute of Allergy and Infectious Diseases , part of the National Institutes ofHealth.
FOOTER
* " class="is-active">Home* Sponsors
* Careers
* Contact
* Search
� 2021 Immune Tolerance Network* Privacy
* Terms of Use
* Accessibility
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0