Are you over 18 and want to see adult content?
More Annotations
A complete backup of matesinconstruction.org.au
Are you over 18 and want to see adult content?

A complete backup of tastybooktours.com
Are you over 18 and want to see adult content?

A complete backup of starrynighteducation.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of https://world-of-photonics-china.com.cn
Are you over 18 and want to see adult content?
A complete backup of https://mcdonalds.pl
Are you over 18 and want to see adult content?
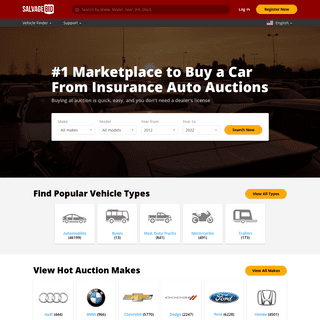
A complete backup of https://salvagebid.com
Are you over 18 and want to see adult content?
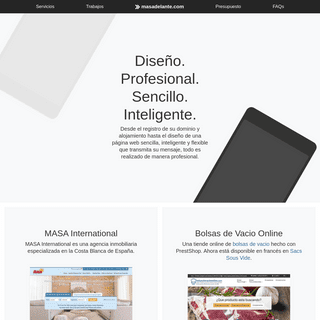
A complete backup of https://masadelante.com
Are you over 18 and want to see adult content?

A complete backup of https://xnview.com
Are you over 18 and want to see adult content?

A complete backup of https://heemskerk.nl
Are you over 18 and want to see adult content?

A complete backup of https://transportesaso.com
Are you over 18 and want to see adult content?

A complete backup of https://heistbrewery.com
Are you over 18 and want to see adult content?
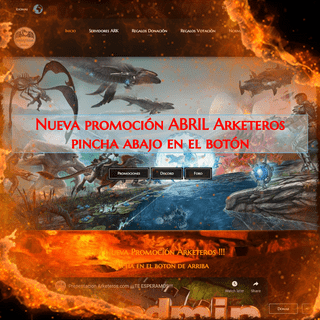
A complete backup of https://arketeros.com
Are you over 18 and want to see adult content?
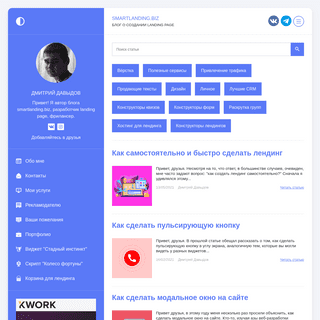
A complete backup of https://smartlanding.biz
Are you over 18 and want to see adult content?

A complete backup of https://fcr.co.jp
Are you over 18 and want to see adult content?

A complete backup of https://richardsonpioneer.ca
Are you over 18 and want to see adult content?
Text
CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in SHINGRIX VACCINATION DURING THE COVID-19 PANDEMIC DRAT �2021 GSK or licensor. SGXLBND210012 January 2021 Produced in USA. 0002-0010-92 Trademarks are owned by or licensed to the GSK group ofcompanies.
FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE Adverse Reaction KINRIX INFANRIX + IPV Localb n = 3,121-3,128 n = 1,039-1,043 Pain, any c57 53 Pain, Grade 2 or 3d 14 12 Pain, Grade 3d 2c 1 Redness, any 37 37 Redness, ≥50 mm 18 20 GSKPRO MY | PRODUCT INFORMATION FOR HEALTHCARE PROFESSIONALS For Medical/Healthcare Professionals only. Before prescribing, please refer to the full prescribing information. GlaxoSmithKline Pharmaceutical Sdn Bhd (3277-U). Level 6, Quill 9, 112, Jalan Semangat, 46300 Petaling Jaya, Selangor Darul Ehsan, Malaysia. Tel:(603)-7495 2600.
HIGHLIGHTS OF PRESCRIBING INFORMATION 2 . FULL PRESCRIBING INFORMATION . 1 INDICATIONS AND USAGE . FLUARIX QUADRIVALENT is indicated for active immunization for the preventionof disease caused
MENVEO® [MENINGOCOCCAL (GROUPS A, C, Y AND W-135 2 . FULL PRESCRIBING INFORMATION . 1 INDICATIONS AND USAGE . MENVEO is a vaccine indicated for active immunization to prevent invasivemeningococcal
2021 NO.4 包装変更のお知らせ Title: 包装変更のお知らせ オーグメンチン配合錠250RS「2021年No.4」 Author: グラクソ・スミスクライン株式会社 GSKPRO GB | PRODUCT INFORMATION FOR HEALTHCARE PROFESSIONALS The Remote Reviews Resource Hub offers the tools and resources you need to confidently review your respiratory patients and manage their treatment remotely. Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/. 帯状疱疹ワクチン (シングリックス筋注用) 接種説明書・予診票 sgnpqst190001-p1908n 作成年月2019年8月 製造販売元(輸入) 帯状疱疹ワクチン (シングリックス筋注用) 接種説明書・予診票 GSKPRO FOR HEALTHCARE PROFESSIONALSLOGINPRODUCTSGSKPRO CAGSKPRO GBPATIENT AND HCP RESOURCESGSK PATIENT ASSISTANCE Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on ourCLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in SHINGRIX VACCINATION DURING THE COVID-19 PANDEMIC DRAT �2021 GSK or licensor. SGXLBND210012 January 2021 Produced in USA. 0002-0010-92 Trademarks are owned by or licensed to the GSK group ofcompanies.
FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE Adverse Reaction KINRIX INFANRIX + IPV Localb n = 3,121-3,128 n = 1,039-1,043 Pain, any c57 53 Pain, Grade 2 or 3d 14 12 Pain, Grade 3d 2c 1 Redness, any 37 37 Redness, ≥50 mm 18 20 GSKPRO MY | PRODUCT INFORMATION FOR HEALTHCARE PROFESSIONALS For Medical/Healthcare Professionals only. Before prescribing, please refer to the full prescribing information. GlaxoSmithKline Pharmaceutical Sdn Bhd (3277-U). Level 6, Quill 9, 112, Jalan Semangat, 46300 Petaling Jaya, Selangor Darul Ehsan, Malaysia. Tel:(603)-7495 2600.
HIGHLIGHTS OF PRESCRIBING INFORMATION 2 . FULL PRESCRIBING INFORMATION . 1 INDICATIONS AND USAGE . FLUARIX QUADRIVALENT is indicated for active immunization for the preventionof disease caused
MENVEO® [MENINGOCOCCAL (GROUPS A, C, Y AND W-135 2 . FULL PRESCRIBING INFORMATION . 1 INDICATIONS AND USAGE . MENVEO is a vaccine indicated for active immunization to prevent invasivemeningococcal
2021 NO.4 包装変更のお知らせ Title: 包装変更のお知らせ オーグメンチン配合錠250RS「2021年No.4」 Author: グラクソ・スミスクライン株式会社 GSKPRO GB | PRODUCT INFORMATION FOR HEALTHCARE PROFESSIONALS The Remote Reviews Resource Hub offers the tools and resources you need to confidently review your respiratory patients and manage their treatment remotely. Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/. 帯状疱疹ワクチン (シングリックス筋注用) 接種説明書・予診票 sgnpqst190001-p1908n 作成年月2019年8月 製造販売元(輸入) 帯状疱疹ワクチン (シングリックス筋注用) 接種説明書・予診票CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in ZEJULA (NIRAPARIB) CAPSULES 3 . swallowing. ZEJULA may be taken with or without food. Bedtime administration may be a potential method for managing nausea. In the case of a missed dose of ZEJULA, instruct patients to take their next dose at its regularly GSK PATIENT ASSISTANCE Event Registration successful. You now have sucesfully register for the event and you can also access the latest updates, events and resources. In case you would like to receive SMS-reminders for the event you have registered, please scroll down to provide your mobilenumber.
DOSING AND ADMINISTRATION Event Registration successful. You now have sucesfully register for the event and you can also access the latest updates, events and resources. In case you would like to receive SMS-reminders for the event you have registered, please scroll down to provide your mobilenumber.
RECONSTITUTION & ADMINISTRATION TIPS FOR SHINGRIX Reconstitution & Administration. Please refer to the full Prescribing Information for SHINGRIX for full details. 1. Refrigerate between 2º and 8ºC (36º and 46ºF). Protect vials from light. DO NOT FREEZE (discard if frozen) Reconstitute and use immediately. Reconstituted vaccine is stable for 6 hours refrigerated between 2º and 8ºC (36º SHINGRIX MECHANISM OF ACTION IMMUNE RESPONSEWITH SHINGRIX. The antigen gE and Adjuvant System AS01 B enhance VZV-specific immune response in a declining immune system 1-3. VZV=varicella zoster virus. *In a descriptive analysis of the pivotal clinical trials, the efficacy in preventing herpes zoster in adults ≥50 and ≥70 years of age was 93% and 85% respectively inthe
FULL PRESCRIBING INFORMATION: CONTENTS* EPIVIR Scored Tablets EPIVIR scored tablets contain 150 mg of lamivudine. The tablets are white, diamond-shaped, scored, film-coated tablets debossed with “GX CJ7” on both sides.RESOURCES | GSKPRO
Nucala (mepolizumab) Homecare patient support booklet. Designed to support patients prescribed Nucala pre-filled pen or pre-filled syringe in a home setting. Item added. Add to Basket. Order now. Download here. NP-GB-MPL-WCNT-210001. January 2021. PrescribingInformation.
HIGHLIGHTS OF PRESCRIBING INFORMATION • THESE HIGHLIGHTS If certain UGT1A or CYP3A inducers are coadministered, then adjust the weight-based dose of TIVICAY to twice daily. (2.4, 2.5, 7.2, 7.3) FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE Solicited Adverse Reactions In 7 clinical studies, detailed safety information was collected by parents/guardians for 8 consecutive days following vaccination with GSKPRO FOR HEALTHCARE PROFESSIONALSLOGINPRODUCTSGSKPRO CAGSKPRO GBPATIENT AND HCP RESOURCESGSK PATIENT ASSISTANCE Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our SHINGRIX VACCINATION DURING THE COVID-19 PANDEMIC DRAT �2021 GSK or licensor. SGXLBND210012 January 2021 Produced in USA. 0002-0010-92 Trademarks are owned by or licensed to the GSK group ofcompanies.
CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in ZEJULA (NIRAPARIB) CAPSULES 3 . swallowing. ZEJULA may be taken with or without food. Bedtime administration may be a potential method for managing nausea. In the case of a missed dose of ZEJULA, instruct patients to take their next dose at its regularly DOSING AND ADMINISTRATION * The need for, and timing of further doses has not yet been determined. BEXSERO should be given by deep intramuscular injection, preferably in the anterolateral aspect of the thigh in infants or in the deltoid muscle region of the upper arm in older subjects. The vaccine must not be mixed with other vaccines or medicinal products inthe same syringe.
RECONSTITUTION & ADMINISTRATION TIPS FOR SHINGRIX Reconstitution & Administration. Please refer to the full Prescribing Information for SHINGRIX for full details. 1. Refrigerate between 2º and 8ºC (36º and 46ºF). Protect vials from light. DO NOT FREEZE (discard if frozen) Reconstitute and use immediately. Reconstituted vaccine is stable for 6 hours refrigerated between 2º and 8ºC (36º SHINGRIX MECHANISM OF ACTION IMMUNE RESPONSEWITH SHINGRIX. The antigen gE and Adjuvant System AS01 B enhance VZV-specific immune response in a declining immune system 1-3. VZV=varicella zoster virus. *In a descriptive analysis of the pivotal clinical trials, the efficacy in preventing herpes zoster in adults ≥50 and ≥70 years of age was 93% and 85% respectively inthe
FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE 3 intramuscular injection is the deltoid region of the upper arm. 2.3 Dose and Schedule . Two doses (0.5 mL each) administered intramuscularly according to the following schedule: A JULUCA (DOLUTEGRAVIR AND RILPIVIRINE) TABLETS 2 12.4 Microbiology 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Clinical Trials in Adult Subjects Switching to JULUCA HIGHLIGHTS OF PRESCRIBING INFORMATION • THESE HIGHLIGHTS If certain UGT1A or CYP3A inducers are coadministered, then adjust the weight-based dose of TIVICAY to twice daily. (2.4, 2.5, 7.2, 7.3) GSKPRO FOR HEALTHCARE PROFESSIONALSLOGINPRODUCTSGSKPRO CAGSKPRO GBPATIENT AND HCP RESOURCESGSK PATIENT ASSISTANCE Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our SHINGRIX VACCINATION DURING THE COVID-19 PANDEMIC DRAT �2021 GSK or licensor. SGXLBND210012 January 2021 Produced in USA. 0002-0010-92 Trademarks are owned by or licensed to the GSK group ofcompanies.
CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in ZEJULA (NIRAPARIB) CAPSULES 3 . swallowing. ZEJULA may be taken with or without food. Bedtime administration may be a potential method for managing nausea. In the case of a missed dose of ZEJULA, instruct patients to take their next dose at its regularly DOSING AND ADMINISTRATION * The need for, and timing of further doses has not yet been determined. BEXSERO should be given by deep intramuscular injection, preferably in the anterolateral aspect of the thigh in infants or in the deltoid muscle region of the upper arm in older subjects. The vaccine must not be mixed with other vaccines or medicinal products inthe same syringe.
RECONSTITUTION & ADMINISTRATION TIPS FOR SHINGRIX Reconstitution & Administration. Please refer to the full Prescribing Information for SHINGRIX for full details. 1. Refrigerate between 2º and 8ºC (36º and 46ºF). Protect vials from light. DO NOT FREEZE (discard if frozen) Reconstitute and use immediately. Reconstituted vaccine is stable for 6 hours refrigerated between 2º and 8ºC (36º SHINGRIX MECHANISM OF ACTION IMMUNE RESPONSEWITH SHINGRIX. The antigen gE and Adjuvant System AS01 B enhance VZV-specific immune response in a declining immune system 1-3. VZV=varicella zoster virus. *In a descriptive analysis of the pivotal clinical trials, the efficacy in preventing herpes zoster in adults ≥50 and ≥70 years of age was 93% and 85% respectively inthe
FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE 3 intramuscular injection is the deltoid region of the upper arm. 2.3 Dose and Schedule . Two doses (0.5 mL each) administered intramuscularly according to the following schedule: A JULUCA (DOLUTEGRAVIR AND RILPIVIRINE) TABLETS 2 12.4 Microbiology 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Clinical Trials in Adult Subjects Switching to JULUCA HIGHLIGHTS OF PRESCRIBING INFORMATION • THESE HIGHLIGHTS If certain UGT1A or CYP3A inducers are coadministered, then adjust the weight-based dose of TIVICAY to twice daily. (2.4, 2.5, 7.2, 7.3) DOSING AND ADMINISTRATION * The need for, and timing of further doses has not yet been determined. BEXSERO should be given by deep intramuscular injection, preferably in the anterolateral aspect of the thigh in infants or in the deltoid muscle region of the upper arm in older subjects. The vaccine must not be mixed with other vaccines or medicinal products inthe same syringe.
GSK SAMPLES AND SAVINGS OFFERS Only practitioners who are licensed to prescribe can request available samples & savings offers. Prescription drug samples and savings offers may be provided, in accordance with State laws, PDMA guidelines, medical specialty, and at the discretion of GSK.RECONSTITUTION
SHINGRIX. Filter redirection page URL: events.html. Please note: This site is intended for Canadian healthcare professionals only and is not intended as a means for reporting an adverse event (side effect) or complaint for any GlaxoSmithKline product. To report an adverse event or product complaint, please call 1-800-387-7374. JULUCA (DOLUTEGRAVIR AND RILPIVIRINE) TABLETS 2 12.4 Microbiology 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Clinical Trials in Adult Subjects Switching to JULUCA HAVRIX VACCINE FOR HEPATITIS A Vaccine Patient Assistance. Coding for GSK Vaccines. HAVRIX is a vaccine indicated for active immunization against disease caused by hepatitis A virus (HAV). HAVRIX is approved for use in persons 12 months of age and older. Primary immunization should be administered at least 2 weeks prior to expected exposure to HAV.PEDIARIX SCHEDULE
PEDIARIX as a primary series: 3 doses of 0.5 mL each, by intramuscular injection, at 2, 4, and 6 months of age (at intervals of 6 to 8 weeks, preferably 8 weeks) 1 According to the CDC, administration of a total of 4 doses of hepatitis B vaccine is permitted when a combination vaccine containing hepatitis B is administered after the birth dose 2; A 3-dose series of PEDIARIX may be administered HAVRIX SCHEDULE & DOSING INFORMATION HAVRIX is a vaccine indicated for active immunization against disease caused by hepatitis A virus (HAV). HAVRIX is approved for use in persons 12 months of age and older. Primary immunization should be administered at least 2 weeks prior to expected exposure to HAV. SERVICE PROVISION FOR THE COPD PATIENT; FROM COVID TO THE Promotional webinar organised and funded by GlaxoSmithKline. PM-GB-NA-LBND-200002 July 2020. Has attended a GSK Webinar. Service Provision for the COPD Patient; from COVID to the Future.HOME | GSKPRO IE
Webinars on demand. Catch up on Asthma & COPD video webinars anytime from the comfort of your own office. Order resources to support your patients including, samples, demo devices, leaflets plus more. Join your peers and healthcare experts at live webinars run by GSK. 2021 NO.4 包装変更のお知らせ Title: 包装変更のお知らせ オーグメンチン配合錠250RS「2021年No.4」 Author: グラクソ・スミスクライン株式会社 GSKPRO FOR HEALTHCARE PROFESSIONALSLOGINPRODUCTSGSKPRO CAGSKPRO GBPATIENT AND HCP RESOURCESGSK PATIENT ASSISTANCE Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our SHINGRIX VACCINATION DURING THE COVID-19 PANDEMIC DRAT �2021 GSK or licensor. SGXLBND210012 January 2021 Produced in USA. 0002-0010-92 Trademarks are owned by or licensed to the GSK group ofcompanies.
CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in ZEJULA (NIRAPARIB) CAPSULES 3 . swallowing. ZEJULA may be taken with or without food. Bedtime administration may be a potential method for managing nausea. In the case of a missed dose of ZEJULA, instruct patients to take their next dose at its regularly DOSING AND ADMINISTRATION * The need for, and timing of further doses has not yet been determined. BEXSERO should be given by deep intramuscular injection, preferably in the anterolateral aspect of the thigh in infants or in the deltoid muscle region of the upper arm in older subjects. The vaccine must not be mixed with other vaccines or medicinal products inthe same syringe.
RECONSTITUTION & ADMINISTRATION TIPS FOR SHINGRIX Reconstitution & Administration. Please refer to the full Prescribing Information for SHINGRIX for full details. 1. Refrigerate between 2º and 8ºC (36º and 46ºF). Protect vials from light. DO NOT FREEZE (discard if frozen) Reconstitute and use immediately. Reconstituted vaccine is stable for 6 hours refrigerated between 2º and 8ºC (36º SHINGRIX MECHANISM OF ACTION IMMUNE RESPONSEWITH SHINGRIX. The antigen gE and Adjuvant System AS01 B enhance VZV-specific immune response in a declining immune system 1-3. VZV=varicella zoster virus. *In a descriptive analysis of the pivotal clinical trials, the efficacy in preventing herpes zoster in adults ≥50 and ≥70 years of age was 93% and 85% respectively inthe
FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE 3 intramuscular injection is the deltoid region of the upper arm. 2.3 Dose and Schedule . Two doses (0.5 mL each) administered intramuscularly according to the following schedule: A JULUCA (DOLUTEGRAVIR AND RILPIVIRINE) TABLETS 2 12.4 Microbiology 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Clinical Trials in Adult Subjects Switching to JULUCA HIGHLIGHTS OF PRESCRIBING INFORMATION • THESE HIGHLIGHTS If certain UGT1A or CYP3A inducers are coadministered, then adjust the weight-based dose of TIVICAY to twice daily. (2.4, 2.5, 7.2, 7.3) GSKPRO FOR HEALTHCARE PROFESSIONALSLOGINPRODUCTSGSKPRO CAGSKPRO GBPATIENT AND HCP RESOURCESGSK PATIENT ASSISTANCE Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our SHINGRIX VACCINATION DURING THE COVID-19 PANDEMIC DRAT �2021 GSK or licensor. SGXLBND210012 January 2021 Produced in USA. 0002-0010-92 Trademarks are owned by or licensed to the GSK group ofcompanies.
CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in ZEJULA (NIRAPARIB) CAPSULES 3 . swallowing. ZEJULA may be taken with or without food. Bedtime administration may be a potential method for managing nausea. In the case of a missed dose of ZEJULA, instruct patients to take their next dose at its regularly DOSING AND ADMINISTRATION * The need for, and timing of further doses has not yet been determined. BEXSERO should be given by deep intramuscular injection, preferably in the anterolateral aspect of the thigh in infants or in the deltoid muscle region of the upper arm in older subjects. The vaccine must not be mixed with other vaccines or medicinal products inthe same syringe.
RECONSTITUTION & ADMINISTRATION TIPS FOR SHINGRIX Reconstitution & Administration. Please refer to the full Prescribing Information for SHINGRIX for full details. 1. Refrigerate between 2º and 8ºC (36º and 46ºF). Protect vials from light. DO NOT FREEZE (discard if frozen) Reconstitute and use immediately. Reconstituted vaccine is stable for 6 hours refrigerated between 2º and 8ºC (36º SHINGRIX MECHANISM OF ACTION IMMUNE RESPONSEWITH SHINGRIX. The antigen gE and Adjuvant System AS01 B enhance VZV-specific immune response in a declining immune system 1-3. VZV=varicella zoster virus. *In a descriptive analysis of the pivotal clinical trials, the efficacy in preventing herpes zoster in adults ≥50 and ≥70 years of age was 93% and 85% respectively inthe
FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE 3 intramuscular injection is the deltoid region of the upper arm. 2.3 Dose and Schedule . Two doses (0.5 mL each) administered intramuscularly according to the following schedule: A JULUCA (DOLUTEGRAVIR AND RILPIVIRINE) TABLETS 2 12.4 Microbiology 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Clinical Trials in Adult Subjects Switching to JULUCA HIGHLIGHTS OF PRESCRIBING INFORMATION • THESE HIGHLIGHTS If certain UGT1A or CYP3A inducers are coadministered, then adjust the weight-based dose of TIVICAY to twice daily. (2.4, 2.5, 7.2, 7.3) DOSING AND ADMINISTRATION * The need for, and timing of further doses has not yet been determined. BEXSERO should be given by deep intramuscular injection, preferably in the anterolateral aspect of the thigh in infants or in the deltoid muscle region of the upper arm in older subjects. The vaccine must not be mixed with other vaccines or medicinal products inthe same syringe.
GSK SAMPLES AND SAVINGS OFFERS Only practitioners who are licensed to prescribe can request available samples & savings offers. Prescription drug samples and savings offers may be provided, in accordance with State laws, PDMA guidelines, medical specialty, and at the discretion of GSK.RECONSTITUTION
SHINGRIX. Filter redirection page URL: events.html. Please note: This site is intended for Canadian healthcare professionals only and is not intended as a means for reporting an adverse event (side effect) or complaint for any GlaxoSmithKline product. To report an adverse event or product complaint, please call 1-800-387-7374. JULUCA (DOLUTEGRAVIR AND RILPIVIRINE) TABLETS 2 12.4 Microbiology 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Clinical Trials in Adult Subjects Switching to JULUCA HAVRIX VACCINE FOR HEPATITIS A Vaccine Patient Assistance. Coding for GSK Vaccines. HAVRIX is a vaccine indicated for active immunization against disease caused by hepatitis A virus (HAV). HAVRIX is approved for use in persons 12 months of age and older. Primary immunization should be administered at least 2 weeks prior to expected exposure to HAV.PEDIARIX SCHEDULE
PEDIARIX as a primary series: 3 doses of 0.5 mL each, by intramuscular injection, at 2, 4, and 6 months of age (at intervals of 6 to 8 weeks, preferably 8 weeks) 1 According to the CDC, administration of a total of 4 doses of hepatitis B vaccine is permitted when a combination vaccine containing hepatitis B is administered after the birth dose 2; A 3-dose series of PEDIARIX may be administered HAVRIX SCHEDULE & DOSING INFORMATION HAVRIX is a vaccine indicated for active immunization against disease caused by hepatitis A virus (HAV). HAVRIX is approved for use in persons 12 months of age and older. Primary immunization should be administered at least 2 weeks prior to expected exposure to HAV. SERVICE PROVISION FOR THE COPD PATIENT; FROM COVID TO THE Promotional webinar organised and funded by GlaxoSmithKline. PM-GB-NA-LBND-200002 July 2020. Has attended a GSK Webinar. Service Provision for the COPD Patient; from COVID to the Future.HOME | GSKPRO IE
Webinars on demand. Catch up on Asthma & COPD video webinars anytime from the comfort of your own office. Order resources to support your patients including, samples, demo devices, leaflets plus more. Join your peers and healthcare experts at live webinars run by GSK. 2021 NO.4 包装変更のお知らせ Title: 包装変更のお知らせ オーグメンチン配合錠250RS「2021年No.4」 Author: グラクソ・スミスクライン株式会社 GSKPRO FOR HEALTHCARE PROFESSIONALSLOGINPRODUCTSGSKPRO CAGSKPRO GBPATIENT AND HCP RESOURCESGSK PATIENT ASSISTANCE Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our SHINGRIX VACCINATION DURING THE COVID-19 PANDEMIC DRAT �2021 GSK or licensor. SGXLBND210012 January 2021 Produced in USA. 0002-0010-92 Trademarks are owned by or licensed to the GSK group ofcompanies.
CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in JULUCA (DOLUTEGRAVIR AND RILPIVIRINE) TABLETS 2 12.4 Microbiology 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Clinical Trials in Adult Subjects Switching to JULUCAGSK RESOURCES
Event Registration successful. You now have sucesfully register for the event and you can also access the latest updates, events and resources. In case you would like to receive SMS-reminders for the event you have registered, please scroll down to provide your mobilenumber.
SHINGRIX MECHANISM OF ACTION IMMUNE RESPONSEWITH SHINGRIX. The antigen gE and Adjuvant System AS01 B enhance VZV-specific immune response in a declining immune system 1-3. VZV=varicella zoster virus. *In a descriptive analysis of the pivotal clinical trials, the efficacy in preventing herpes zoster in adults ≥50 and ≥70 years of age was 93% and 85% respectively inthe
RECONSTITUTION & ADMINISTRATION TIPS FOR SHINGRIX Reconstitution & Administration. Please refer to the full Prescribing Information for SHINGRIX for full details. 1. Refrigerate between 2º and 8ºC (36º and 46ºF). Protect vials from light. DO NOT FREEZE (discard if frozen) Reconstitute and use immediately. Reconstituted vaccine is stable for 6 hours refrigerated between 2º and 8ºC (36ºRESOURCES | GSKPRO
Nucala (mepolizumab) Homecare patient support booklet. Designed to support patients prescribed Nucala pre-filled pen or pre-filled syringe in a home setting. Item added. Add to Basket. Order now. Download here. NP-GB-MPL-WCNT-210001. January 2021. PrescribingInformation.
GSK'S MEDICINES AND VACCINES SUPPLY AVAILABILITY FORTUM INJECTION 500mg. 434092. OUT OF STOCK. EARLY JAN 2022. INFANRIX IPV+HIB+2N FS. 3243839. OUT OF STOCK. END OCTOBER 2021. If you have any queries about the availability of a product, please contact yourwholesaler.
GSKPRO GB | PRODUCT INFORMATION FOR HEALTHCARE PROFESSIONALS The Remote Reviews Resource Hub offers the tools and resources you need to confidently review your respiratory patients and manage their treatment remotely. Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/. GSKPRO FOR HEALTHCARE PROFESSIONALSLOGINPRODUCTSGSKPRO CAGSKPRO GBPATIENT AND HCP RESOURCESGSK PATIENT ASSISTANCE Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our SHINGRIX VACCINATION DURING THE COVID-19 PANDEMIC DRAT �2021 GSK or licensor. SGXLBND210012 January 2021 Produced in USA. 0002-0010-92 Trademarks are owned by or licensed to the GSK group ofcompanies.
CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo in JULUCA (DOLUTEGRAVIR AND RILPIVIRINE) TABLETS 2 12.4 Microbiology 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Clinical Trials in Adult Subjects Switching to JULUCAGSK RESOURCES
Event Registration successful. You now have sucesfully register for the event and you can also access the latest updates, events and resources. In case you would like to receive SMS-reminders for the event you have registered, please scroll down to provide your mobilenumber.
SHINGRIX MECHANISM OF ACTION IMMUNE RESPONSEWITH SHINGRIX. The antigen gE and Adjuvant System AS01 B enhance VZV-specific immune response in a declining immune system 1-3. VZV=varicella zoster virus. *In a descriptive analysis of the pivotal clinical trials, the efficacy in preventing herpes zoster in adults ≥50 and ≥70 years of age was 93% and 85% respectively inthe
RECONSTITUTION & ADMINISTRATION TIPS FOR SHINGRIX Reconstitution & Administration. Please refer to the full Prescribing Information for SHINGRIX for full details. 1. Refrigerate between 2º and 8ºC (36º and 46ºF). Protect vials from light. DO NOT FREEZE (discard if frozen) Reconstitute and use immediately. Reconstituted vaccine is stable for 6 hours refrigerated between 2º and 8ºC (36ºRESOURCES | GSKPRO
Nucala (mepolizumab) Homecare patient support booklet. Designed to support patients prescribed Nucala pre-filled pen or pre-filled syringe in a home setting. Item added. Add to Basket. Order now. Download here. NP-GB-MPL-WCNT-210001. January 2021. PrescribingInformation.
GSK'S MEDICINES AND VACCINES SUPPLY AVAILABILITY FORTUM INJECTION 500mg. 434092. OUT OF STOCK. EARLY JAN 2022. INFANRIX IPV+HIB+2N FS. 3243839. OUT OF STOCK. END OCTOBER 2021. If you have any queries about the availability of a product, please contact yourwholesaler.
GSKPRO GB | PRODUCT INFORMATION FOR HEALTHCARE PROFESSIONALS The Remote Reviews Resource Hub offers the tools and resources you need to confidently review your respiratory patients and manage their treatment remotely. Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/.CLINICAL TRIALS
Modified Total Vaccinated Cohort (mTVC): The primary efficacy analysis included all subjects who did not develop a confirmed case of shingles within 1 month after the second dose. 1-3. Primary endpoints: 1-3 ZOE-50: to evaluate the efficacy of SHINGRIX vs. placebo inGSK RESOURCES
Event Registration successful. You now have sucesfully register for the event and you can also access the latest updates, events and resources. In case you would like to receive SMS-reminders for the event you have registered, please scroll down to provide your mobilenumber.
ZEJULA (NIRAPARIB) CAPSULES 3 . swallowing. ZEJULA may be taken with or without food. Bedtime administration may be a potential method for managing nausea. In the case of a missed dose of ZEJULA, instruct patients to take their next dose at its regularly GSK PATIENT ASSISTANCE Event Registration successful. You now have sucesfully register for the event and you can also access the latest updates, events and resources. In case you would like to receive SMS-reminders for the event you have registered, please scroll down to provide your mobilenumber.
GSK VACCINES
Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our GSK SAMPLES AND SAVINGS OFFERS Event registration completed - pending validation. Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on ourPEDIARIX SCHEDULE
PEDIARIX as a primary series: 3 doses of 0.5 mL each, by intramuscular injection, at 2, 4, and 6 months of age (at intervals of 6 to 8 weeks, preferably 8 weeks) 1 According to the CDC, administration of a total of 4 doses of hepatitis B vaccine is permitted when a combination vaccine containing hepatitis B is administered after the birth dose 2; A 3-dose series of PEDIARIX may be administered HIGHLIGHTS OF PRESCRIBING INFORMATION • THESE HIGHLIGHTS If certain UGT1A or CYP3A inducers are coadministered, then adjust the weight-based dose of TIVICAY to twice daily. (2.4, 2.5, 7.2, 7.3) SERVICE PROVISION FOR THE COPD PATIENT; FROM COVID TO THE Promotional webinar organised and funded by GlaxoSmithKline. PM-GB-NA-LBND-200002 July 2020. Has attended a GSK Webinar. Service Provision for the COPD Patient; from COVID to the Future.WWW.GSKPRO.COM
Moved Permanently. The document has moved here. Skip to main content Not a healthcare professional? Visit our public siteENGLISH
English
Français
FOR HEALTHCARE PROFESSIONALS Not a healthcare professional? Visit our public site THIS SITE MAY CONTAIN PROMOTIONAL MATERIAL For Healthcare Professionals Report adverse eventMenu
Close
Close
Login Register Your browser does not support JavaScript. Some components may not bevisible.
* Communication preferencesBasket
User logged in
* Products
* Therapy areas
* Contact us
* Live Chat
* Report adverse event* Basket 0
ANORO ELLIPTA 20 MCG _Added to your cart_Popup
59
_Registration successful_ You now have access to the latest updates, events and resources. Event _Registration successful_ You now have sucesfully register for the event and you can also access the latest updates, events and resources. In case you would like to receive SMS-reminders for the event you have registered, please scroll down to provide your mobile number. _Account activated – pending validation._ Your account has been activated successfully, but we still need to validate you as a healthcare professional. We’ll send you an email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our website _EVENT REGISTRATION COMPLETED - PENDING VALIDATION._ Your account has been activated successfully, but we still need to validate you as a healthcare professional. We'll send you and email with the result of the validation process in the next days. In the meantime, you can enjoy access to all the latest news, events and resources on our website In case you would like to receive sms-reminders for the event you have registered, please update your communication preferences here.
_Account activated - pending validation._ Your account has been activated successfully, but we still need to validate you as a healthcare professional. We’ll send you an email with the result of the validation process in the next days. In the meantime, you can enjoy the public content on our website _Event Account activated - pending validation._ Your account has been activated successfully, but we still need to validate you as a healthcare professional. We’ll send you an email with the result of the validation process in the next days. In the meantime, you can enjoy the public content on our website In case you would like to receive sms-reminders for the event you have registered, please update your communication preferences here.
already registered
X »
YOU ARE NOW LEAVING GSK’S WEBSITE You are now leaving a GSK Website. By clicking this link, you will be taken to a website that is not owned or controlled by GSK, and GSK is not responsible for the content provided on that siteContinue
Go back
Unit for Hours:H
Unit for Minutes: M
Loading text: $loading Filter redirection page URL: events.html Login overlay needed?: true Filter Tags:Products Filter Tags values when filtered by all Tags:All Products OutofBox filter category name:Format Filter Reset all button text:Reset All Filter Apply button text:Apply Events registration success message: Registration successfull Events registration failure message: Registration failure Events un-registration success message: Un-Registration successfull Events un-registration failure message: Un-Registration failure Reset all default value: false Show More Naivagation Values: 10|15|20 Previous Text label: Previous Next Text label: Next Show Text label: Show Seats left Text: spaces left No Seats left Text: No spaces left Text Area Blank copy in Email Template:Not Provided * ASYEVE0001=Event Registration-Portal * ASYEVE0002=Watched General Video * ASYEVE0003=Watched Virtual Rep Video * ASYEVE0004=Watched Patient Video * ASYEVE0005=Watched Expert Video * ASYEVE0006=Watched Webcast * ASYEVE0007=Downloaded Content-Portal * ASYEVE0008=Clicked to App Store * ASYEVE0009=Watched Learning Module Video * ASYEVE0010=Email Sent * ASYEVE0011=Email Read * ASYEVE0012=Email Clicked * ASYEVE0013=Shared a Link * ASYEVE0014=Rep Request * ASYEVE0015=Joined health.gsk * ASYEVE0016=Email not sent GSK'S HEALTHCARE PROFESSIONAL WEBSITE IS HERE, OFFERING INFORMATION AND UPDATES ON GSK PRODUCTS. We believe in putting patients first and staying connected with you. CREATE YOUR GSK PRO ACCOUNT TODAY. Sign up for your personalized account today and discover what GSK Prohas to offer.
Register now
SHINGRIX VACCINE
Learn about how SHINGRIX can help protect your patients against herpes zoster in adults 50 years of age or older.TRELEGY ELLIPTA
Discover TRELEGY ELLIPTABREO
Discover BREO
NUCALA
Explore NUCALA
OUR PRODUCTS
Quick access to GSK prescriptions and vaccines for you and yourpatients.
Explore our productsJOIN GSK PRO TODAY
We share our research and knowledge through live and recorded webinars, live chat with our Medical Information Team and patienttools.
Video Player
Use Left/Right Arrow keys to advance one second, Up/Down arrows to advance ten seconds.00:0000:00 | 00:00
Use Up/Down Arrow keys to increase or decrease volume.NaN
Video player requires JavaScript enabled. You can download this videohere:
https://videos.gskstatic.com/pharma/GSKpro/Canada/en-ca-portal-update-13-may-1020-v1.mp4 Here at GSK, we help you whenever you need it, wherever you need it to better support your patients. Join GSK Pro and discover what it has tooffer.
GSK PRO: FOR YOU _AND_ YOUR PATIENTS.Français
* Public site at gsk.ca* Terms of use
* Privacy statement
Trade marks are owned by or licensed to the GSK group of companies. � 2020 GSK group of companies or its licensor.03427 08/20
Please note: This site is intended for Canadian healthcare professionals only and is not intended as a means for reporting an adverse event (side effect) or complaint for any GlaxoSmithKline product. To report an adverse event or product complaint, please call1-800-387-7374.
SORRY, THIS SITE IS NOT SUPPORTED ON ANDROID V4.3 AND BELOW. Please update your operating system or use a different device.Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0