Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://deltadentalil.com
Are you over 18 and want to see adult content?
A complete backup of https://hbhsfoundation.org
Are you over 18 and want to see adult content?
A complete backup of https://brownsugarkitchen.com
Are you over 18 and want to see adult content?

A complete backup of https://vsba.com
Are you over 18 and want to see adult content?
A complete backup of https://spiceislands.com
Are you over 18 and want to see adult content?
A complete backup of https://mywebisworth.com
Are you over 18 and want to see adult content?
A complete backup of https://mcchrystalgroup.com
Are you over 18 and want to see adult content?
A complete backup of https://borderlandsthegame.com
Are you over 18 and want to see adult content?

A complete backup of https://businessconnectworld.com
Are you over 18 and want to see adult content?
A complete backup of https://shadowkeepzine.org
Are you over 18 and want to see adult content?

A complete backup of https://cheapjerseyssonly.com
Are you over 18 and want to see adult content?

A complete backup of https://sunbasket.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of http://batesturkey.com/wholeturkey.aspx
Are you over 18 and want to see adult content?

A complete backup of https://sattamatkaind.in/
Are you over 18 and want to see adult content?
A complete backup of https://www.comico.in.th/novel/titles/1709
Are you over 18 and want to see adult content?
A complete backup of https://www.shapeways.com/designer/checkerviet
Are you over 18 and want to see adult content?

A complete backup of https://calcularsueldo.com.ar/sueldopordia.html
Are you over 18 and want to see adult content?
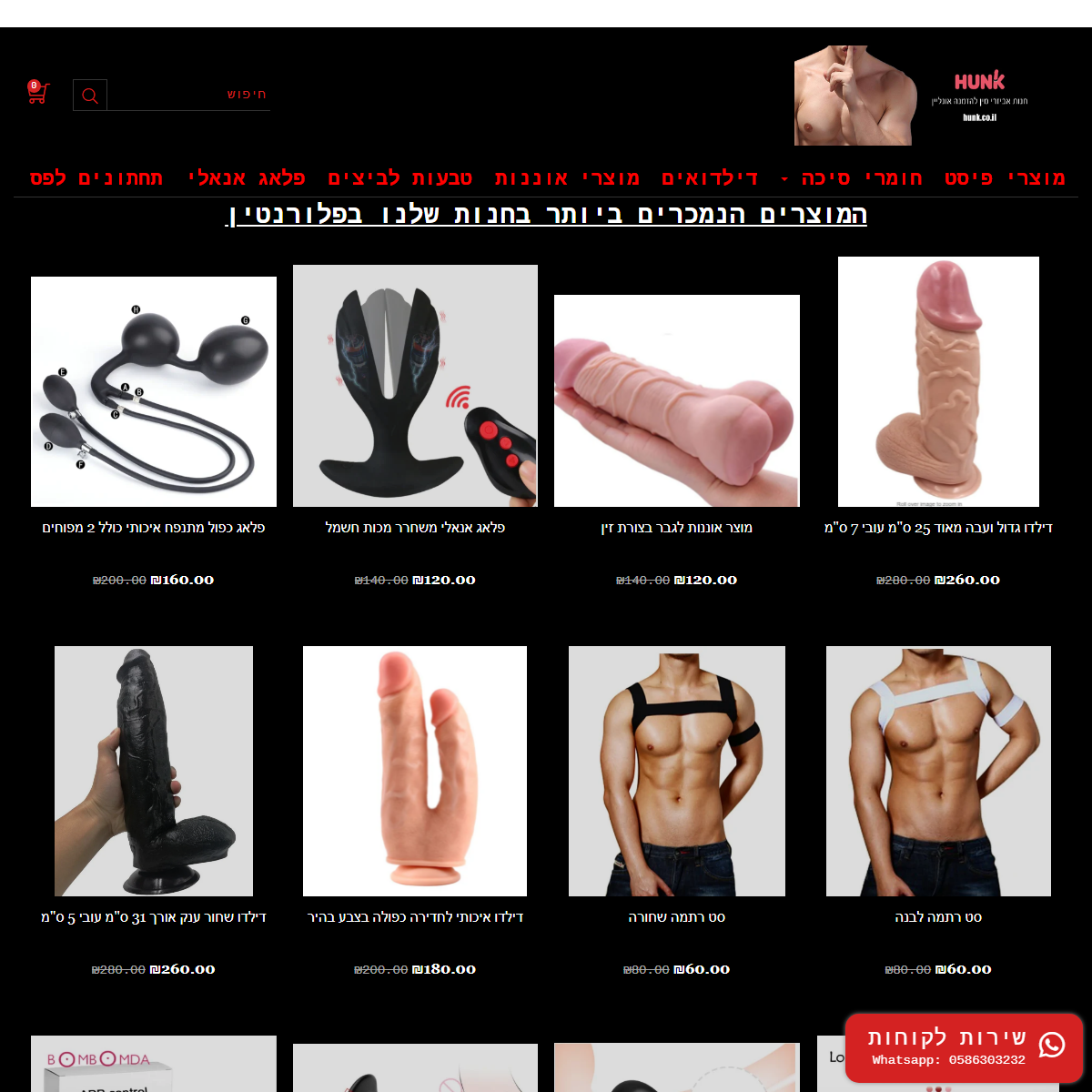
A complete backup of https://www.hunk.co.il/
Are you over 18 and want to see adult content?

A complete backup of https://dpboss.net/satta-matka-fix-game.php
Are you over 18 and want to see adult content?

A complete backup of https://sehuatang.net/thread-453386-1-1.html
Are you over 18 and want to see adult content?
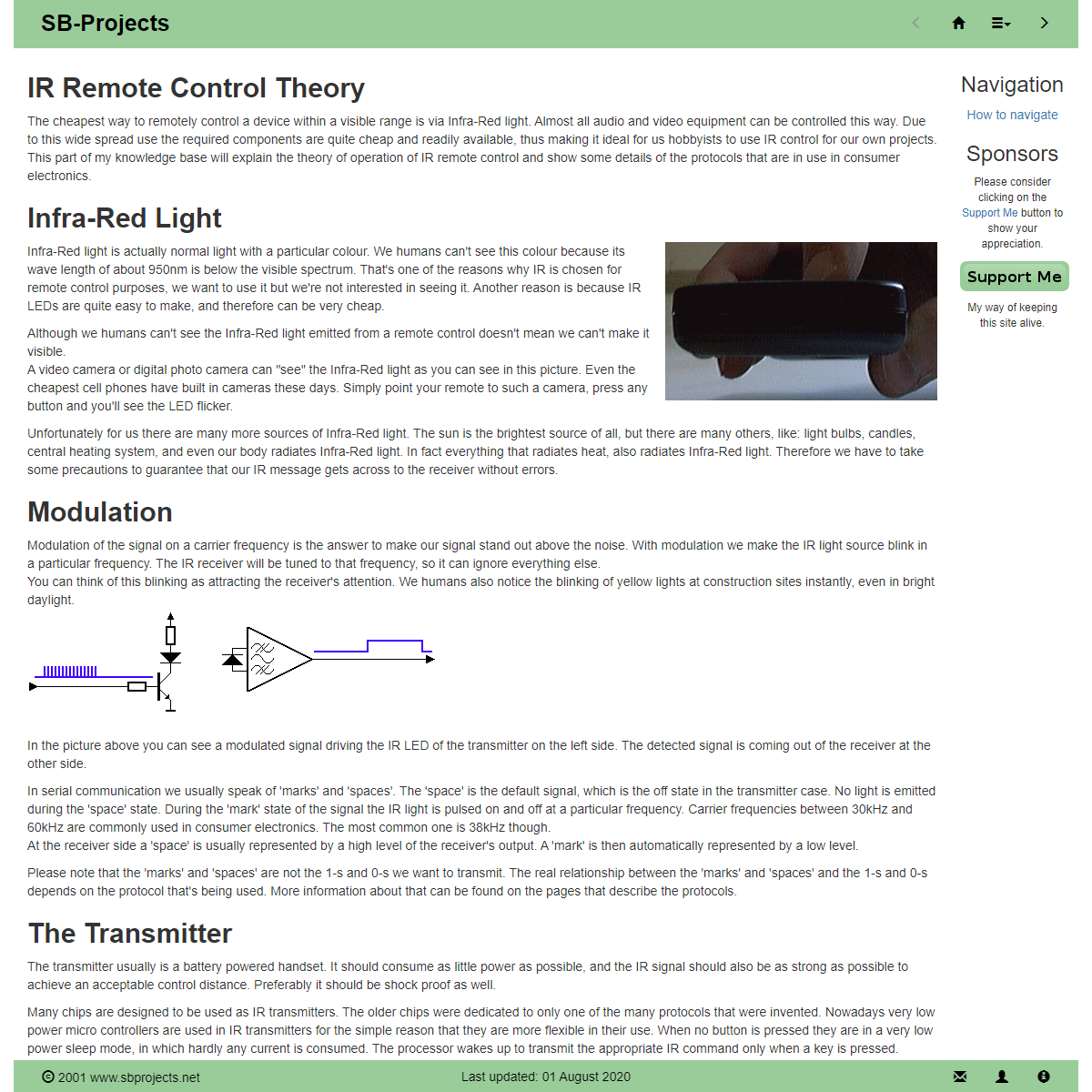
A complete backup of https://www.sbprojects.net/knowledge/ir/index.php
Are you over 18 and want to see adult content?
A complete backup of https://marathisituation.blogspot.com/
Are you over 18 and want to see adult content?
Text
which
ISTENT INJECT®
The iStent inject ® Advantage. The iStent inject ® is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma, or pigmentary glaucoma. Key advantages to using the device include: MINIMAL INVASIVENESS. Implanted through a single-entry point; iStent inject ® is implanted during cataract surgery or as aGLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2021. Key highlights include: Net sales growth of 23% to $68.0 million in Q1 2021, compared to $55.3 million in Q1 2020. ISTENT INJECT® W IMPORTANT SAFETY INFORMATION Speak with a Physician Today. Help is right around the corner. Leading eye professionals across the country offer iStent inject ® W for the treatment and control of eye pressure associated with mild-to-moderate open-angle glaucoma, and are available to answer your questions. Find out if you are a candidate for iStent inject ® W by talking with an iStent inject ® W physician in your area today.HCP RESOURCES
Using Photrexa® Viscous (riboflavin 5’-phosphate in 20% dextran ophthalmic solution), Photrexa® (riboflavin 5’-phosphate ophthalmic solution), and the KXL® System, the iLink™ corneal cross-linking procedure from Glaukos is the only FDA-approved therapeutic treatment for patients with progressive keratoconus and corneal ectasia following refractive surgery. *1GLAUKOS
Glaukos started a revolution in glaucoma surgery and treatment with the development of the first Micro-Invasive Glaucoma Surgery device. To date, trabecular micro-bypass technologies such as iStent ®, iStent inject ®, and iStent inject ® W have been implanted in over 600,000 eyes around the world, with more patients benefitting fromthese
GLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2020. Key highlights include: Q1 2020 net sales of $55.3 million, compared to $54.0 million in Q1 2019.GLAUKOS CORPORATION
Substantial IOP Reduction Observed with Highly Favorable Safety Profile Through 12 Months 76% of Subjects Achieved 20% or Greater Reduction in Month 12 IOP with Same or Lower Medication Burden Data Suggest iStent infinite May Offer Ophthalmic Surgeons a Compelling New Treatment Option in Standalone Procedure for Managing Glaucoma Patients’ IOP and Disease ADVANCING THE TREATMENT OF OPHTHALMIC DISEASES Corporate Overview. Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedurewhich
GLAUKOSTRANSLATE THIS PAGE Glaukosは、世界で失明の主な原因である緑内障に関連する治療に特化し、革新的な治療法と製品を開発し市場への導入を目指す眼科専門企業です。 ADVANCING THE TREATMENT OF OPHTHALMIC DISEASES Corporate Overview. Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedurewhich
ISTENT INJECT®
The iStent inject ® Advantage. The iStent inject ® is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma, or pigmentary glaucoma. Key advantages to using the device include: MINIMAL INVASIVENESS. Implanted through a single-entry point; iStent inject ® is implanted during cataract surgery or as aGLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2021. Key highlights include: Net sales growth of 23% to $68.0 million in Q1 2021, compared to $55.3 million in Q1 2020. ISTENT INJECT® W IMPORTANT SAFETY INFORMATION Speak with a Physician Today. Help is right around the corner. Leading eye professionals across the country offer iStent inject ® W for the treatment and control of eye pressure associated with mild-to-moderate open-angle glaucoma, and are available to answer your questions. Find out if you are a candidate for iStent inject ® W by talking with an iStent inject ® W physician in your area today.HCP RESOURCES
Using Photrexa® Viscous (riboflavin 5’-phosphate in 20% dextran ophthalmic solution), Photrexa® (riboflavin 5’-phosphate ophthalmic solution), and the KXL® System, the iLink™ corneal cross-linking procedure from Glaukos is the only FDA-approved therapeutic treatment for patients with progressive keratoconus and corneal ectasia following refractive surgery. *1GLAUKOS
Glaukos started a revolution in glaucoma surgery and treatment with the development of the first Micro-Invasive Glaucoma Surgery device. To date, trabecular micro-bypass technologies such as iStent ®, iStent inject ®, and iStent inject ® W have been implanted in over 600,000 eyes around the world, with more patients benefitting fromthese
GLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2020. Key highlights include: Q1 2020 net sales of $55.3 million, compared to $54.0 million in Q1 2019.GLAUKOS CORPORATION
Substantial IOP Reduction Observed with Highly Favorable Safety Profile Through 12 Months 76% of Subjects Achieved 20% or Greater Reduction in Month 12 IOP with Same or Lower Medication Burden Data Suggest iStent infinite May Offer Ophthalmic Surgeons a Compelling New Treatment Option in Standalone Procedure for Managing Glaucoma Patients’ IOP and Disease ADVANCING THE TREATMENT OF OPHTHALMIC DISEASES Corporate Overview. Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedurewhich
GLAUKOSTRANSLATE THIS PAGE Glaukosは、世界で失明の主な原因である緑内障に関連する治療に特化し、革新的な治療法と製品を開発し市場への導入を目指す眼科専門企業です。ABOUT US - GLAUKOS
Mission Statement Our mission is to transform the treatment of chronic eye diseases with novel therapies that provide sustainable solutions to important clinical needs. Glaukos Corporate Overview Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal GLAUKOS HISTORY AND LOCATIONS New Glaukos headquarters is established in San Clemente, California. 2017. Glaukos global direct sales operations expands to 17 countries and more than 170 professionals. iStent inject is approved in Brazil, Israel, Malaysia, Saudi Arabia, and Singapore. Glaukos completes PhaseII
GLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced the completion of patient enrollment and randomization in its U.S. Food and Drug Administration (FDA) New Drug Application (NDA) Phase 3 clinical trials for the iDose® TR ISTENT INJECT® W SURGERY Exceptional Elegance. Advanced Innovation. The iStent inject ® W System is engineered to provide an enhanced surgical experience and ensure confident delivery, for every procedure. With a streamlined injector system and next-generation stent design, featuring a wide flange at its base, iStent inject ® W is designed to optimize stent visualization and placement, enhance procedural WHAT TO EXPECT WITH ISTENT INJECT® W SURGERY Speak with a Physician Today. Help is right around the corner. Leading eye professionals across the country offer iStent inject ® W for the treatment and control of eye pressure associated with mild-to-moderate open-angle glaucoma, and are available to answer your questions. Find out if you are a candidate for iStent inject ® W by talking with an iStent inject ® W physician in your area today.GLAUKOS CORPORATION
Substantial IOP Reduction Observed with Highly Favorable Safety Profile Through 12 Months 76% of Subjects Achieved 20% or Greater Reduction in Month 12 IOP with Same or Lower Medication Burden Data Suggest iStent infinite May Offer Ophthalmic Surgeons a Compelling New Treatment Option in Standalone Procedure for Managing Glaucoma Patients’ IOP and DiseaseGLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced that the Therapeutic Goods Administration (TGA) of Australia has granted regulatory approval for the PRESERFLO MicroShunt intended for the reduction of intraocular pressureRIBOFLAVIN FAMILY
CE-marked ParaCel is a 2-step trans-epithelial riboflavin specifically formulated for direct application on the intact epithelium. ParaCel’s higher concentration of riboflavin and proprietary formulation allow for fast penetration and diffusion into the cornealstroma.
INTEGRATING ISTENT INJECT® W INTO YOUR PRACTICE Integrating iStent inject ® W and MIGS into your practice helps you expand care, and provide a wider range of benefits to many of your patients. As a trusted industry leader—and the corporate founder of MIGS—we have the experience, tools, and training to make integration easy. We lead the industry in clinical support and patient education ADVANCING THE TREATMENT OF OPHTHALMIC DISEASES Corporate Overview. Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedurewhich
ADVANCING THE TREATMENT OF OPHTHALMIC DISEASES Corporate Overview. Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedurewhich
ISTENT INJECT®
The iStent inject ® Advantage. The iStent inject ® is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma, or pigmentary glaucoma. Key advantages to using the device include: MINIMAL INVASIVENESS. Implanted through a single-entry point; iStent inject ® is implanted during cataract surgery or as aWORK WITH US
R&D Project Engineer. Kristine oversees the iterative design process at Glaukos, making sure that new products meet regulatory and quality assurance standards, implementing improvements to existing product lines. show more. background image of group of people looking overnotes before a
ISTENT INJECT® W IMPORTANT SAFETY INFORMATION Speak with a Physician Today. Help is right around the corner. Leading eye professionals across the country offer iStent inject ® W for the treatment and control of eye pressure associated with mild-to-moderate open-angle glaucoma, and are available to answer your questions. Find out if you are a candidate for iStent inject ® W by talking with an iStent inject ® W physician in your area today.GLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2021. Key highlights include: Net sales growth of 23% to $68.0 million in Q1 2021, compared to $55.3 million in Q1 2020.GLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2020. Key highlights include: Q1 2020 net sales of $55.3 million, compared to $54.0 million in Q1 2019.GLAUKOS
Glaukos started a revolution in glaucoma surgery and treatment with the development of the first Micro-Invasive Glaucoma Surgery device. To date, trabecular micro-bypass technologies such as iStent ®, iStent inject ®, and iStent inject ® W have been implanted in over 600,000 eyes around the world, with more patients benefitting fromthese
ISTENT INJECT BILLING AND CODING GUIDE iStent inject®: BILLING AND CODING GUIDE The iStent inject Trabecular Micro-Bypass System Model G2-M-IS is indicated for use in conjunction with cataract surgery for the reduction of intraocular pressure (IOP) in adult patients with mild to moderate primary open-angle glaucoma. ADVANCING THE TREATMENT OF OPHTHALMIC DISEASES Corporate Overview. Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedurewhich
GLAUKOSTRANSLATE THIS PAGEGLAUKOS CORPORATIONGLAUKOS NEWSGLAUKOS STOCKGLAUKOS TICKERGLAUKOS CORP STOCKGLAUKOS IPRISM CLIP Glaukosは、世界で失明の主な原因である緑内障に関連する治療に特化し、革新的な治療法と製品を開発し市場への導入を目指す眼科専門企業です。 ADVANCING THE TREATMENT OF OPHTHALMIC DISEASES Corporate Overview. Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedurewhich
ISTENT INJECT®
The iStent inject ® Advantage. The iStent inject ® is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma, or pigmentary glaucoma. Key advantages to using the device include: MINIMAL INVASIVENESS. Implanted through a single-entry point; iStent inject ® is implanted during cataract surgery or as aWORK WITH US
R&D Project Engineer. Kristine oversees the iterative design process at Glaukos, making sure that new products meet regulatory and quality assurance standards, implementing improvements to existing product lines. show more. background image of group of people looking overnotes before a
ISTENT INJECT® W IMPORTANT SAFETY INFORMATION Speak with a Physician Today. Help is right around the corner. Leading eye professionals across the country offer iStent inject ® W for the treatment and control of eye pressure associated with mild-to-moderate open-angle glaucoma, and are available to answer your questions. Find out if you are a candidate for iStent inject ® W by talking with an iStent inject ® W physician in your area today.GLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2021. Key highlights include: Net sales growth of 23% to $68.0 million in Q1 2021, compared to $55.3 million in Q1 2020.GLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2020. Key highlights include: Q1 2020 net sales of $55.3 million, compared to $54.0 million in Q1 2019.GLAUKOS
Glaukos started a revolution in glaucoma surgery and treatment with the development of the first Micro-Invasive Glaucoma Surgery device. To date, trabecular micro-bypass technologies such as iStent ®, iStent inject ®, and iStent inject ® W have been implanted in over 600,000 eyes around the world, with more patients benefitting fromthese
ISTENT INJECT BILLING AND CODING GUIDE iStent inject®: BILLING AND CODING GUIDE The iStent inject Trabecular Micro-Bypass System Model G2-M-IS is indicated for use in conjunction with cataract surgery for the reduction of intraocular pressure (IOP) in adult patients with mild to moderate primary open-angle glaucoma. ADVANCING THE TREATMENT OF OPHTHALMIC DISEASES Corporate Overview. Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedurewhich
GLAUKOSTRANSLATE THIS PAGEGLAUKOS CORPORATIONGLAUKOS NEWSGLAUKOS STOCKGLAUKOS TICKERGLAUKOS CORP STOCKGLAUKOS IPRISM CLIP Glaukosは、世界で失明の主な原因である緑内障に関連する治療に特化し、革新的な治療法と製品を開発し市場への導入を目指す眼科専門企業です。ABOUT US - GLAUKOS
Mission Statement Our mission is to transform the treatment of chronic eye diseases with novel therapies that provide sustainable solutions to important clinical needs. Glaukos Corporate Overview Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinalGLAUCOMA TREATMENT
Implanted during cataract surgery, iStent inject ® W can help reduce your eye pressure, and may reduce your reliance on glaucoma medication, at the discretion of an eye care professional. background image of A penny shows how small the Glaukos iStent inject® device is at just .23 millimeters in width and .36 millimeters in length.GLAUKOS CORPORATION
Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders and retinal diseases, today announced financial results for the first quarter ended March 31, 2021. Key highlights include: Net sales growth of 23% to $68.0 million in Q1 2021, compared to $55.3 million in Q1 2020.HCP RESOURCES
Using Photrexa® Viscous (riboflavin 5’-phosphate in 20% dextran ophthalmic solution), Photrexa® (riboflavin 5’-phosphate ophthalmic solution), and the KXL® System, the iLink™ corneal cross-linking procedure from Glaukos is the only FDA-approved therapeutic treatment for patients with progressive keratoconus and corneal ectasia following refractive surgery. *1 ISTENT INJECT® W IMPORTANT SAFETY INFORMATION Speak with a Physician Today. Help is right around the corner. Leading eye professionals across the country offer iStent inject ® W for the treatment and control of eye pressure associated with mild-to-moderate open-angle glaucoma, and are available to answer your questions. Find out if you are a candidate for iStent inject ® W by talking with an iStent inject ® W physician in your area today. THE ISTENT INJECT® W PROCEDURE FOR GLAUCOMA TREATMENT iStent inject ® W can help to restore your eye’s natural fluid outflow and is designed to reduce pressure inside your eye. background image of A penny shows how small the Glaukos iStent inject® W device is at just .23 millimeters in width and .36 millimeters in length. It is the smallest medical device known to be implanted into humans. INTEGRATING ISTENT INJECT® W INTO YOUR PRACTICE Integrating iStent inject ® W and MIGS into your practice helps you expand care, and provide a wider range of benefits to many of your patients. As a trusted industry leader—and the corporate founder of MIGS—we have the experience, tools, and training to make integration easy. We lead the industry in clinical support and patient educationGLAUKOS CORPORATION
Amendment Adds Presbyopia as a New Investigational Application for Patented, Non-Invasive, Topical Eyelid Drug Delivery Platform Presbyopia Expands Upon Already Established Investigational Applications for Dry Eye Disease, Glaucoma and Other Corneal Disorders Glaukos Corporation (NYSE: GKOS), an ophthalmic medical technology and pharmaceutical company focused GLAUKOSTRANSLATE THIS PAGE Glaukosは、世界で失明の主な原因である緑内障に関連する治療に特化し、革新的な治療法と製品を開発し市場への導入を目指す眼科専門企業です。 ISTENT® - GLAUKOSTRANSLATE THIS PAGE iStent® – 開放隅角緑内障で眼圧(IOP)を下降 させるための最初のMIGSデバイス iStent®は、原発開放隅角緑内障患者の眼圧(IOP)を下降させるためのデバイスです。このデバイスを使用する主なメリットは以下の通りです: 組織への最小限の侵襲 iStent®は、白内障手術のために必要な切開創をToggle navigation
Glaukos Glaukos Logo Glaucoma Corneal HealthRetinal Diseases
* Understanding Glaucoma* About Glaucoma
* Symptoms & Risk Factors* Diagnosis
* World Glaucoma Week* Treatment Options
* Understanding Your Options * Micro-Invasive Glaucoma Surgery (MIGS) * The iStent _inject_® W Procedure * iStent _inject_® W Overview* Innovative Design
* How iStent _inject_® W Works * What To Expect With iStent _inject_®* Find a Physician
* Healthcare Professionals* MIGS
* iStent _inject_® W* Clinical Data
* Integrating iStent _inject_® W Into Your Practice* Reimbursement
* Upcoming Events
* Contact Us
* Understanding Keratoconus * World Keratoconus Day * The iLink™ Procedure * Diagnosing Keratoconus* iDetect KC
* Insurance Coverage * iLink™ Copay Savings Program* HCP Resources
* Patients
* Overview
* Glaucoma
* Corneal Health
* Retinal Diseases
Back
Glaucoma
* Understanding Glaucoma* About Glaucoma
* Symptoms & Risk Factors* Diagnosis
* World Glaucoma Week* Treatment Options
* Understanding Your Options * Micro-Invasive Glaucoma Surgery (MIGS) * The iStent _inject_® W Procedure * iStent _inject_® W Overview* Innovative Design
* How iStent _inject_® W Works * What To Expect With iStent _inject_®* Find a Physician
* Healthcare Professionals* MIGS
* iStent _inject_® W* Clinical Data
* Integrating iStent _inject_® W Into Your Practice* Reimbursement
* Upcoming Events
* Contact Us
Back
Corneal Health
* Understanding Keratoconus * World Keratoconus Day * The iLink™ Procedure * Diagnosing Keratoconus* iDetect KC
* Insurance Coverage * iLink™ Copay Savings Program* HCP Resources
* Patients
Search for:
Search
* News & Events
* Blog
* About Us
* About Glaukos
* History & Locations* Innovation
* Glaukos Charitable Foundation * Awards & Recognition * Careers at Glaukos* Careers
* Life at Glaukos
* Benefits
* Investors
* Donate
* Location: United StatesAustralia & New ZealandBrasilCanadaEspañaFranceDeutschland日本America LatinaUnitedKingdom
* Language: English Français* Search for:
Search
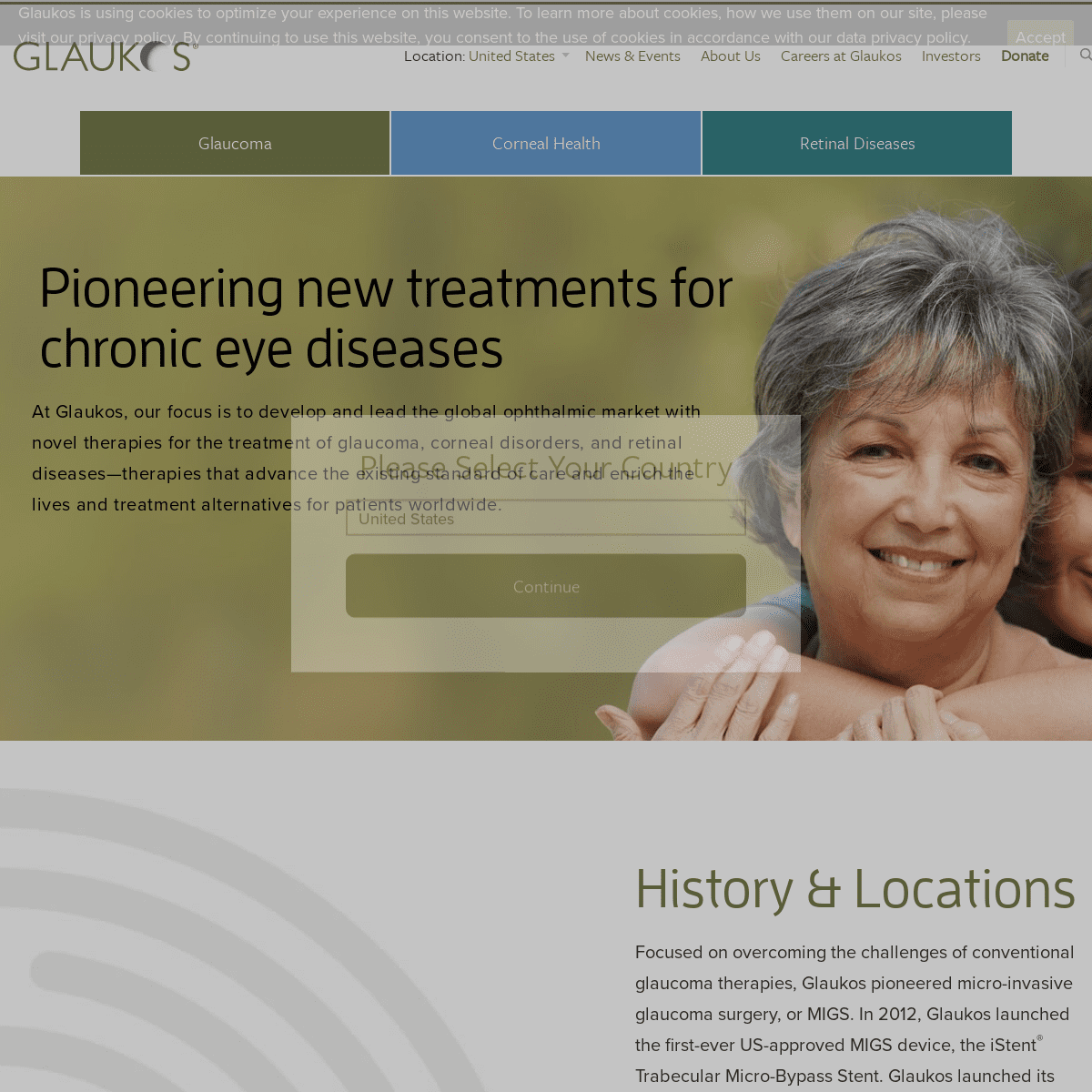
PIONEERING NEW TREATMENTS FOR CHRONIC EYE DISEASES At Glaukos, our focus is to develop and lead the global ophthalmic market with novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases—therapies that advance the existing standard of care and enrich the lives and treatment alternatives forpatients worldwide.
HISTORY & LOCATIONS
Focused on overcoming the challenges of conventional glaucoma therapies, Glaukos pioneered micro-invasive glaucoma surgery, or MIGS. In 2012, Glaukos launched the first-ever US-approved MIGS device, the iStent® Trabecular Micro-Bypass Stent. Glaukos launched its next-generation iStent _inject_® device in the United States in 2018. In addition to an abiding focus on glaucoma, Glaukos is broadening its mission to include new aspirations that seek to improve treatment alternatives and quality of life for people suffering with corneal disorders and retinal diseases. Glaukos is seeking to leverage its platform technologies to build a comprehensive and proprietary portfolio of micro-scale surgical and pharmaceutical therapies in glaucoma, corneal health and retinal disease. Learn more about our history and international presence. More About Our History & Locations OPHTHALMIC INNOVATION Glaukos is committed to investing in the future of ophthalmology to transform standards of care and improve patient lives. See how our dedicated commitment to continuous innovation has resulted in an unrivaled pipeline of novel therapies.Learn More
PHILANTHROPY IS IN OUR BACKBONE From financial contributions to product donations, helping the visually impaired and others in need is deeply ingrained in ourcorporate culture.
Learn About Corporate Giving background image of A large pile of 3D metallic stars AWARDS & RECOGNITION We’re honored to be the recipient of multiple industry awards and recognized by major media publications.Learn More
CORPORATE OVERVIEW
Founded in 1998, Glaukos Corporation is an ophthalmic medical technology and pharmaceutical company focused on novel therapies for the treatment of glaucoma, corneal disorders, and retinal diseases. Our company was the first company to bring to market Micro-Invasive Glaucoma Surgery (MIGS), the micro-invasive procedure which revolutionized the treatment and management of glaucoma. In 2012, we launched our first MIGS device—the iStent®—in the United States, followed by our next-generation iStent _inject_® device in 2018 and the iStent _inject_® W in 2020. In November 2019, Glaukos acquired Avedro, maker of the first and only FDA-approved cross-linking technology for progressive keratoconus. And we continue to seek to leverage our platform technologies to build a comprehensive and proprietary portfolio of micro-scale surgical and pharmaceutical therapies for glaucoma, corneal health, and retinaldiseases.
Our company completed an initial public offering in June of 2015, and our shares are traded on the New York Stock Exchange under the ticker symbol “GKOS”. Our global headquarters is located in San Clemente, California, with additional locations in Waltham and Burlington,Massachusetts.
* About Us
* Mission / Overview* Global Charity
* Management Team
* Board of Directors * Scientific Advisory Board* Newsroom
* Careers at Glaukos * Patient Information* Policies & Info
* Terms of Use
* Privacy Policy
* Declaration of Compliance * Talent Acquisition Privacy Policy * DFU & MRI Information* Patents
* Investors
* Investor Overview
* Stock Information
* Financials
* Corporate Governance* Press Releases
* Events & Presentations * Resources & AlertsContact Us
Sign Up For News & Updates Sign Up For Investor-Related Email AlertsFOLLOW US
logo facebook logo linkedinlogo instagram
logo twitter
Glaukos®, iStent®, iStent _inject_® and iStent _inject_® W are registered trademarks of Glaukos Corporation. iLink™ is a trademark of Glaukos Corporation. Photrexa®, Photrexa® Viscous, and the KXL® system are registered trademarks of Avedro, a Glaukos company. All rights reserved. ©2021. Phone: +1-949-367-9600 Fax: +1-949-367-9984 229 Avenida Fabricante, San Clemente, CA 92672 United States PLEASE SELECT YOUR COUNTRY Please Select Your Country United StatesAustralia & New ZealandBrasilCanadaEspañaFranceDeutschland日本America LatinaUnitedKingdom Continue
By clicking “Yes” you acknowledge and agree to Glaukos’ policy regarding the use and sharing of your personal data. For more information, please see our privacy policy.Yes
×
LEAVING THE GLAUKOS.COM WEBSITE You have just clicked a link that will take you outside of Glaukos.com By clicking "Continue to outside site" you will be taken to: This is a website to which our privacy policy does not apply. You are solely responsible for your interactions on outside websites. Continue to outside site Cancel and stay here WE ARE EXCITED TO ANNOUNCE THAT GLAUKOS HAS ACQUIRED AVEDRO, INC.! You will now be directed to Avedro's homepage. Glaukos is using cookies to optimize your experience on this website. To learn more about cookies, how we use them on our site, please visit our privacy policy. By continuing to use this website, you consent to the use of cookies in accordance with our data privacy policy. PrivacyPolicy Accept
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0