Are you over 18 and want to see adult content?
More Annotations
A complete backup of emporiododireito.com.br
Are you over 18 and want to see adult content?
A complete backup of naturalhealinghome.com
Are you over 18 and want to see adult content?
A complete backup of jamaicainquirer.com
Are you over 18 and want to see adult content?

A complete backup of frenchmorning.com
Are you over 18 and want to see adult content?
A complete backup of autosport.com.ru
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of https://securecafe.com
Are you over 18 and want to see adult content?
A complete backup of https://mybeautifulbride.net
Are you over 18 and want to see adult content?

A complete backup of https://4humm87.com
Are you over 18 and want to see adult content?

A complete backup of https://bantenglive.com
Are you over 18 and want to see adult content?
A complete backup of https://kiotviet.vn
Are you over 18 and want to see adult content?

A complete backup of https://rtvt32i3.online
Are you over 18 and want to see adult content?
A complete backup of https://listbell.com
Are you over 18 and want to see adult content?
A complete backup of https://dailycandidnews.com
Are you over 18 and want to see adult content?
A complete backup of https://hminterriors.com
Are you over 18 and want to see adult content?

A complete backup of https://bighugelabs.com
Are you over 18 and want to see adult content?

A complete backup of https://hist-chron.com
Are you over 18 and want to see adult content?
A complete backup of https://thecipherbrief.com
Are you over 18 and want to see adult content?
Text
Panels. Learn More.
EPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within aHCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive ESENSOR THROMBOPHILIA RISK TEST PACKAGE INSERT PI0971 REV C Page 3 of 20 INTENDED USE The eSensor® Thrombophilia Risk Test is an in vitro diagnostic for the detection and genotyping of Factor II (Prothrombin) G20210A, Factor V (Factor V Leiden) G1691A and MTHFR (human 5, 10 GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
EPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within aHCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive ESENSOR THROMBOPHILIA RISK TEST PACKAGE INSERT PI0971 REV C Page 3 of 20 INTENDED USE The eSensor® Thrombophilia Risk Test is an in vitro diagnostic for the detection and genotyping of Factor II (Prothrombin) G20210A, Factor V (Factor V Leiden) G1691A and MTHFR (human 5, 10 TECHNOLOGY | GENMARK DIAGNOSTICS Technology eSensor® Technology GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is not based on fluorescent or optical detection. As a result, diagnostic tests are CONTACT US | GENMARK DIAGNOSTICS Contact Us GenMark Diagnostics, Inc. 5964 La Place Court Carlsbad, CA 92008 USA Toll-Free: Phone: Fax: Email: 1.800.eSensor (373.6767) +1 760.448.4300 +1 760.448.4301 info@genmarkdx.com Customer Service Phone: Email: Fax: 1.800.eSensor (373.6767), Option 1 CustomerService@genmarkdx.com +1 866.831.2001 Technical Support Phone: Email: 1.800.eSensor RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP)1 and NEW Respiratory Pathogen Panel 2 (RP2)2 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 DETECTION OF VARIANT SARS-COV-2 STRAINS ON THE EPLEX Analysis of more than 150,500 complete genome sequences of SARS-CoV-2 submitted to GISAID between February 1, 2021 and March 10, 2021 identified only 3 sequences in this dataset predicted to be missed by both of the N gene assays resulting in a combined inclusivity of the 2 N gene assays on the ePlex RP2 Panel of 99.99%. SARS-CoV-2 Variant.Also.
INVESTORS OVERVIEW
Investors Overview. GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. Utilizing GenMark’s proprietary eSensor ® detection technology, GenMark’s eSensor XT-8 ® and ePlex ® systems are designed tosupport a
ESENSOR TECHNOLOGY
eSensor Technology. Use Up/Down Arrow keys to increase or decrease volume. GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and isMANAGEMENT TEAM
Mendel earned a B.S. in finance from Indiana University and an M.B.A. from Northwestern University’s Kellogg School of Management. Mr. Ek has served as Chief Financial Officer since February 2019. Previously, Mr. Ek served as GenMark’s VP of Finance & Accounting and Controller from November 2013 until his appointment as Chief Financial Officer. WEBINARS: MOLECULAR DIAGNOSTIC SOLUTIONS DESIGNED FOR THE Webinars Molecular Diagnostic Solutions Designed for the Patient and Optimized for the Laboratory in a Pandemic Presented by: Heba Mostafa, MD, Ph.D, D(ABMM) Dr. Heba Mostafa and her team have taken a non-conventional approach to guide and build a testing algorithm forthe
PACKAGE INSERTS
Package Inserts. Package Inserts for GenMark products are found on the Customer Resource Center, in addition to assay manuals, quick reference guides, brochures, FAQs, Customer Training Presentations, assay worksheets, and Safety Data Sheets (SDS). Products may be subject to restrictions on use as set forth in GenMark’s Terms. EPLEX® RESPIRATORY PATHOGEN (RP) PANELWEB VIEW ePlex® Respiratory Pathogen (RP) Panel. The ePlex RP Panel is a qualitative nucleic acid multiplex in vitro diagnostic test intended for use on the ePlex instrument for simultaneous detection and identification of multiple respiratory viral and bacterial nucleic acids in nasopharyngeal swabs (NPS) obtained from individuals exhibiting signs and symptoms of respiratory tract infection. GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
TECHNOLOGY | GENMARK DIAGNOSTICS Technology eSensor® Technology GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is not based on fluorescent or optical detection. As a result, diagnostic tests areEPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within aHCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
TECHNOLOGY | GENMARK DIAGNOSTICS Technology eSensor® Technology GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is not based on fluorescent or optical detection. As a result, diagnostic tests areEPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within aHCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive TECHNOLOGY | GENMARK DIAGNOSTICS Technology eSensor® Technology GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is not based on fluorescent or optical detection. As a result, diagnostic tests are CONTACT US | GENMARK DIAGNOSTICS Contact Us GenMark Diagnostics, Inc. 5964 La Place Court Carlsbad, CA 92008 USA Toll-Free: Phone: Fax: Email: 1.800.eSensor (373.6767) +1 760.448.4300 +1 760.448.4301 info@genmarkdx.com Customer Service Phone: Email: Fax: 1.800.eSensor (373.6767), Option 1 CustomerService@genmarkdx.com +1 866.831.2001 Technical Support Phone: Email: 1.800.eSensor RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP)1 and NEW Respiratory Pathogen Panel 2 (RP2)2 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 DETECTION OF VARIANT SARS-COV-2 STRAINS ON THE EPLEX Analysis of more than 150,500 complete genome sequences of SARS-CoV-2 submitted to GISAID between February 1, 2021 and March 10, 2021 identified only 3 sequences in this dataset predicted to be missed by both of the N gene assays resulting in a combined inclusivity of the 2 N gene assays on the ePlex RP2 Panel of 99.99%. SARS-CoV-2 Variant.Also.
LOGIN — CUSTOMER RESOURCE CENTER Login to the Customer Resource Center to view SDS Sheets, Package Inserts, Worksheets, Training Presentations and Brochures.INVESTORS OVERVIEW
Investors Overview. GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. Utilizing GenMark’s proprietary eSensor ® detection technology, GenMark’s eSensor XT-8 ® and ePlex ® systems are designed tosupport a
WEBINARS: MOLECULAR DIAGNOSTIC SOLUTIONS DESIGNED FOR THE Webinars Molecular Diagnostic Solutions Designed for the Patient and Optimized for the Laboratory in a Pandemic Presented by: Heba Mostafa, MD, Ph.D, D(ABMM) Dr. Heba Mostafa and her team have taken a non-conventional approach to guide and build a testing algorithm forthe
PACKAGE INSERTS
Package Inserts. Package Inserts for GenMark products are found on the Customer Resource Center, in addition to assay manuals, quick reference guides, brochures, FAQs, Customer Training Presentations, assay worksheets, and Safety Data Sheets (SDS). Products may be subject to restrictions on use as set forth in GenMark’s Terms.OPERATOR MANUAL
PI1066 – Rev. G 1 Operator Manual GenMark Diagnostics Inc. EMERCO EUROP 5964 La Place Court Prinsessegracht 20 EPLEX® RESPIRATORY PATHOGEN (RP) PANELWEB VIEW ePlex® Respiratory Pathogen (RP) Panel. The ePlex RP Panel is a qualitative nucleic acid multiplex in vitro diagnostic test intended for use on the ePlex instrument for simultaneous detection and identification of multiple respiratory viral and bacterial nucleic acids in nasopharyngeal swabs (NPS) obtained from individuals exhibiting signs and symptoms of respiratory tract infection. GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS ePlex Panels. GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels.EPLEX SYSTEM
Designed for the Patient: Reduce Avoidable Medical Errors. Preventable medical errors are now the third leading cause of death in the United States at more than 250,000 per year. 1 Automating information transfer has been shown to be effective in reducing many common errors, including patient identity checking and order transcription. 2 The ePlex system is uniquely designed with patient safetyEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, comparedINVESTORS OVERVIEW
The Investor Relations website contains information about GenMark Diagnostics, Inc. business for stockholders, potential investors, andfinancial analysts.
HCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
ELECTROWETTING TECHNOLOGY Electrowetting is a digital microfluidic technology that greatly simplifies the movement of fluid to enable rapid thermal cycling for fast turnaround time. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS ePlex Panels. GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels.EPLEX SYSTEM
Designed for the Patient: Reduce Avoidable Medical Errors. Preventable medical errors are now the third leading cause of death in the United States at more than 250,000 per year. 1 Automating information transfer has been shown to be effective in reducing many common errors, including patient identity checking and order transcription. 2 The ePlex system is uniquely designed with patient safetyEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, comparedINVESTORS OVERVIEW
The Investor Relations website contains information about GenMark Diagnostics, Inc. business for stockholders, potential investors, andfinancial analysts.
HCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
ELECTROWETTING TECHNOLOGY Electrowetting is a digital microfluidic technology that greatly simplifies the movement of fluid to enable rapid thermal cycling for fast turnaround time. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. TECHNOLOGY | GENMARK DIAGNOSTICS Technology eSensor® Technology GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is not based on fluorescent or optical detection. As a result, diagnostic tests areXT-8 SYSTEMS
XT-8® Systems XT-8 System Benefits The XT-8 system is the pioneer of true multiplex molecular testing, offering post-PCR detection for infectious disease, genetics, and pharmacogenetics targets. DOWNLOAD XT-8 SYSTEM BROCHURE Simple To Use An intuitive touch-screen user interface and easy-to-interpret reports enable seamless integration ofINVESTORS OVERVIEW
The Investor Relations website contains information about GenMark Diagnostics, Inc. business for stockholders, potential investors, andfinancial analysts.
LOGIN — CUSTOMER RESOURCE CENTER Login to the Customer Resource Center to view SDS Sheets, Package Inserts, Worksheets, Training Presentations and Brochures. RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP) and NEW Respiratory Pathogen Panel 2 (RP2)1 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 DETECTION OF VARIANT SARS-COV-2 STRAINS ON THE EPLEX Detection of variant SARS-CoV-2 strains on the ePlex ® RP2 Panel * March 22, 2021. Genetic surveillance of SARS-CoV-2 strains circulating globally has identified multiple variants containing one or more mutations that can potentially impact detection by nucleic acid basedtesting methods.
WEBINARS: MOLECULAR DIAGNOSTIC SOLUTIONS DESIGNED FOR THE Webinars Molecular Diagnostic Solutions Designed for the Patient and Optimized for the Laboratory in a Pandemic Presented by: Heba Mostafa, MD, Ph.D, D(ABMM) Dr. Heba Mostafa and her team have taken a non-conventional approach to guide and build a testing algorithm forthe
ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information.OPERATOR MANUAL
PI1066 – Rev. G 1 Operator Manual GenMark Diagnostics Inc. EMERCO EUROP 5964 La Place Court Prinsessegracht 20 ESENSOR THROMBOPHILIA RISK TEST PACKAGE INSERT PI0971 REV C Page 3 of 20 INTENDED USE The eSensor® Thrombophilia Risk Test is an in vitro diagnostic for the detection and genotyping of Factor II (Prothrombin) G20210A, Factor V (Factor V Leiden) G1691A and MTHFR (human 5, 10 GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
CONTACT US | GENMARK DIAGNOSTICS Contact Us GenMark Diagnostics, Inc. 5964 La Place Court Carlsbad, CA 92008 USA Toll-Free: Phone: Fax: Email: 1.800.eSensor (373.6767) +1 760.448.4300 +1 760.448.4301 info@genmarkdx.com Customer Service Phone: Email: Fax: 1.800.eSensor (373.6767), Option 1 CustomerService@genmarkdx.com +1 866.831.2001 Technical Support Phone: Email: 1.800.eSensorINVESTORS OVERVIEW
Investors Overview. GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. Utilizing GenMark’s proprietary eSensor ® detection technology, GenMark’s eSensor XT-8 ® and ePlex ® systems are designed tosupport a
RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP) and NEW Respiratory Pathogen Panel 2 (RP2)1 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19EPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared GENMARK DIAGNOSTICS’ EPLEX® RESPIRATORY PATHOGEN PANEL 2 Combination test for COVID-19, flu and other common respiratory illnesses helps health care professionals prepare for flu season. RP2 Panel at a Glance. GenMark’s ePlex ® Respiratory Pathogen Panel 2 (RP2 Panel) has received Emergency Use Authorization from the FDA.; The RP2 Panel provides results in less than two hours for more than 20 viruses and bacteria that cause commonPUBLICATIONS
Publications. Opportunities Revealed for Antimicrobial Stewardship and Clinical Practice with Implementation of a Rapid Respiratory Multiplex Assay. Clinical Significance of Upper Airway Virus Detection in Critically Ill Hematology Patients. Drive More Efficient Clinical Action by Streamlining the Interpretation of Test Results.HCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within a GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
CONTACT US | GENMARK DIAGNOSTICS Contact Us GenMark Diagnostics, Inc. 5964 La Place Court Carlsbad, CA 92008 USA Toll-Free: Phone: Fax: Email: 1.800.eSensor (373.6767) +1 760.448.4300 +1 760.448.4301 info@genmarkdx.com Customer Service Phone: Email: Fax: 1.800.eSensor (373.6767), Option 1 CustomerService@genmarkdx.com +1 866.831.2001 Technical Support Phone: Email: 1.800.eSensorINVESTORS OVERVIEW
Investors Overview. GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. Utilizing GenMark’s proprietary eSensor ® detection technology, GenMark’s eSensor XT-8 ® and ePlex ® systems are designed tosupport a
RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP) and NEW Respiratory Pathogen Panel 2 (RP2)1 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19EPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared GENMARK DIAGNOSTICS’ EPLEX® RESPIRATORY PATHOGEN PANEL 2 Combination test for COVID-19, flu and other common respiratory illnesses helps health care professionals prepare for flu season. RP2 Panel at a Glance. GenMark’s ePlex ® Respiratory Pathogen Panel 2 (RP2 Panel) has received Emergency Use Authorization from the FDA.; The RP2 Panel provides results in less than two hours for more than 20 viruses and bacteria that cause commonPUBLICATIONS
Publications. Opportunities Revealed for Antimicrobial Stewardship and Clinical Practice with Implementation of a Rapid Respiratory Multiplex Assay. Clinical Significance of Upper Airway Virus Detection in Critically Ill Hematology Patients. Drive More Efficient Clinical Action by Streamlining the Interpretation of Test Results.HCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within a CONTACT US | GENMARK DIAGNOSTICS Contact Us GenMark Diagnostics, Inc. 5964 La Place Court Carlsbad, CA 92008 USA Toll-Free: Phone: Fax: Email: 1.800.eSensor (373.6767) +1 760.448.4300 +1 760.448.4301 info@genmarkdx.com Customer Service Phone: Email: Fax: 1.800.eSensor (373.6767), Option 1 CustomerService@genmarkdx.com +1 866.831.2001 Technical Support Phone: Email: 1.800.eSensorEPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsXT-8 SYSTEMS
XT-8® Systems XT-8 System Benefits The XT-8 system is the pioneer of true multiplex molecular testing, offering post-PCR detection for infectious disease, genetics, and pharmacogenetics targets. DOWNLOAD XT-8 SYSTEM BROCHURE Simple To Use An intuitive touch-screen user interface and easy-to-interpret reports enable seamless integration of RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP) and NEW Respiratory Pathogen Panel 2 (RP2)1 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 DETECTION OF VARIANT SARS-COV-2 STRAINS ON THE EPLEX Detection of variant SARS-CoV-2 strains on the ePlex ® RP2 Panel * March 22, 2021. Genetic surveillance of SARS-CoV-2 strains circulating globally has identified multiple variants containing one or more mutations that can potentially impact detection by nucleic acid basedtesting methods.
LOGIN — CUSTOMER RESOURCE CENTER Login to the Customer Resource Center to view SDS Sheets, Package Inserts, Worksheets, Training Presentations and Brochures.PRESS RELEASES
GenMark Diagnostics to Report Fourth Quarter and Full Year 2020 Financial Results on February 25, 2021. CARLSBAD, Calif. , Feb. 11, 2021 (GLOBE NEWSWIRE) -- GenMark Diagnostics, Inc. (NASDAQ: GNMK) today announced that it plans to release its fourth quarter and full year earnings results after market close on Thursday, February 25,2021.
RESPIRATORY PATHOGEN PANEL Respiratory Pathogen Panels. Respiratory Pathogen Panel (RP) & NEW Respiratory Pathogen Panel 2 (RP2) GenMark’s ePlex Respiratory Pathogen Panels detect and identify the most common viral and bacterial organisms associated with upper respiratory infection. The new RP2 panel includes SARS-CoV-2, the pathogen that causes COVID-19.PACKAGE INSERTS
Package Inserts. Package Inserts for GenMark products are found on the Customer Resource Center, in addition to assay manuals, quick reference guides, brochures, FAQs, Customer Training Presentations, assay worksheets, and Safety Data Sheets (SDS). Products may be subject to restrictions on use as set forth in GenMark’s Terms. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
CONTACT US | GENMARK DIAGNOSTICS Contact Us GenMark Diagnostics, Inc. 5964 La Place Court Carlsbad, CA 92008 USA Toll-Free: Phone: Fax: Email: 1.800.eSensor (373.6767) +1 760.448.4300 +1 760.448.4301 info@genmarkdx.com Customer Service Phone: Email: Fax: 1.800.eSensor (373.6767), Option 1 CustomerService@genmarkdx.com +1 866.831.2001 Technical Support Phone: Email: 1.800.eSensorINVESTORS OVERVIEW
Investors Overview. GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. Utilizing GenMark’s proprietary eSensor ® detection technology, GenMark’s eSensor XT-8 ® and ePlex ® systems are designed tosupport a
RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP) and NEW Respiratory Pathogen Panel 2 (RP2)1 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19EPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared GENMARK DIAGNOSTICS’ EPLEX® RESPIRATORY PATHOGEN PANEL 2 Combination test for COVID-19, flu and other common respiratory illnesses helps health care professionals prepare for flu season. RP2 Panel at a Glance. GenMark’s ePlex ® Respiratory Pathogen Panel 2 (RP2 Panel) has received Emergency Use Authorization from the FDA.; The RP2 Panel provides results in less than two hours for more than 20 viruses and bacteria that cause commonPUBLICATIONS
Publications. Opportunities Revealed for Antimicrobial Stewardship and Clinical Practice with Implementation of a Rapid Respiratory Multiplex Assay. Clinical Significance of Upper Airway Virus Detection in Critically Ill Hematology Patients. Drive More Efficient Clinical Action by Streamlining the Interpretation of Test Results.HCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within a GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
CONTACT US | GENMARK DIAGNOSTICS Contact Us GenMark Diagnostics, Inc. 5964 La Place Court Carlsbad, CA 92008 USA Toll-Free: Phone: Fax: Email: 1.800.eSensor (373.6767) +1 760.448.4300 +1 760.448.4301 info@genmarkdx.com Customer Service Phone: Email: Fax: 1.800.eSensor (373.6767), Option 1 CustomerService@genmarkdx.com +1 866.831.2001 Technical Support Phone: Email: 1.800.eSensorINVESTORS OVERVIEW
Investors Overview. GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. Utilizing GenMark’s proprietary eSensor ® detection technology, GenMark’s eSensor XT-8 ® and ePlex ® systems are designed tosupport a
RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP) and NEW Respiratory Pathogen Panel 2 (RP2)1 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19EPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared GENMARK DIAGNOSTICS’ EPLEX® RESPIRATORY PATHOGEN PANEL 2 Combination test for COVID-19, flu and other common respiratory illnesses helps health care professionals prepare for flu season. RP2 Panel at a Glance. GenMark’s ePlex ® Respiratory Pathogen Panel 2 (RP2 Panel) has received Emergency Use Authorization from the FDA.; The RP2 Panel provides results in less than two hours for more than 20 viruses and bacteria that cause commonPUBLICATIONS
Publications. Opportunities Revealed for Antimicrobial Stewardship and Clinical Practice with Implementation of a Rapid Respiratory Multiplex Assay. Clinical Significance of Upper Airway Virus Detection in Critically Ill Hematology Patients. Drive More Efficient Clinical Action by Streamlining the Interpretation of Test Results.HCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within a CONTACT US | GENMARK DIAGNOSTICS Contact Us GenMark Diagnostics, Inc. 5964 La Place Court Carlsbad, CA 92008 USA Toll-Free: Phone: Fax: Email: 1.800.eSensor (373.6767) +1 760.448.4300 +1 760.448.4301 info@genmarkdx.com Customer Service Phone: Email: Fax: 1.800.eSensor (373.6767), Option 1 CustomerService@genmarkdx.com +1 866.831.2001 Technical Support Phone: Email: 1.800.eSensorEPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsXT-8 SYSTEMS
XT-8® Systems XT-8 System Benefits The XT-8 system is the pioneer of true multiplex molecular testing, offering post-PCR detection for infectious disease, genetics, and pharmacogenetics targets. DOWNLOAD XT-8 SYSTEM BROCHURE Simple To Use An intuitive touch-screen user interface and easy-to-interpret reports enable seamless integration of RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP) and NEW Respiratory Pathogen Panel 2 (RP2)1 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 DETECTION OF VARIANT SARS-COV-2 STRAINS ON THE EPLEX Detection of variant SARS-CoV-2 strains on the ePlex ® RP2 Panel * March 22, 2021. Genetic surveillance of SARS-CoV-2 strains circulating globally has identified multiple variants containing one or more mutations that can potentially impact detection by nucleic acid basedtesting methods.
LOGIN — CUSTOMER RESOURCE CENTER Login to the Customer Resource Center to view SDS Sheets, Package Inserts, Worksheets, Training Presentations and Brochures.PRESS RELEASES
GenMark Diagnostics to Report Fourth Quarter and Full Year 2020 Financial Results on February 25, 2021. CARLSBAD, Calif. , Feb. 11, 2021 (GLOBE NEWSWIRE) -- GenMark Diagnostics, Inc. (NASDAQ: GNMK) today announced that it plans to release its fourth quarter and full year earnings results after market close on Thursday, February 25,2021.
RESPIRATORY PATHOGEN PANEL Respiratory Pathogen Panels. Respiratory Pathogen Panel (RP) & NEW Respiratory Pathogen Panel 2 (RP2) GenMark’s ePlex Respiratory Pathogen Panels detect and identify the most common viral and bacterial organisms associated with upper respiratory infection. The new RP2 panel includes SARS-CoV-2, the pathogen that causes COVID-19.PACKAGE INSERTS
Package Inserts. Package Inserts for GenMark products are found on the Customer Resource Center, in addition to assay manuals, quick reference guides, brochures, FAQs, Customer Training Presentations, assay worksheets, and Safety Data Sheets (SDS). Products may be subject to restrictions on use as set forth in GenMark’s Terms. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
EPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP)1 and NEW Respiratory Pathogen Panel 2 (RP2)2 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within aHCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
EPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP)1 and NEW Respiratory Pathogen Panel 2 (RP2)2 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within aHCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive TECHNOLOGY | GENMARK DIAGNOSTICS Technology eSensor® Technology GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is not based on fluorescent or optical detection. As a result, diagnostic tests are RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP)1 and NEW Respiratory Pathogen Panel 2 (RP2)2 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19XT-8 SYSTEMS
XT-8® Systems XT-8 System Benefits The XT-8 system is the pioneer of true multiplex molecular testing, offering post-PCR detection for infectious disease, genetics, and pharmacogenetics targets. DOWNLOAD XT-8 SYSTEM BROCHURE Simple To Use An intuitive touch-screen user interface and easy-to-interpret reports enable seamless integration of DETECTION OF VARIANT SARS-COV-2 STRAINS ON THE EPLEX Detection of variant SARS-CoV-2 strains on the ePlex ® RP2 Panel * March 22, 2021. Genetic surveillance of SARS-CoV-2 strains circulating globally has identified multiple variants containing one or more mutations that can potentially impact detection by nucleic acid basedtesting methods.
INVESTORS OVERVIEW
Investors Overview. GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. Utilizing GenMark’s proprietary eSensor ® detection technology, GenMark’s eSensor XT-8 ® and ePlex ® systems are designed tosupport a
LOGIN — CUSTOMER RESOURCE CENTER Login to the Customer Resource Center to view SDS Sheets, Package Inserts, Worksheets, Training Presentations and Brochures. WEBINARS: MOLECULAR DIAGNOSTIC SOLUTIONS DESIGNED FOR THE Webinars Molecular Diagnostic Solutions Designed for the Patient and Optimized for the Laboratory in a Pandemic Presented by: Heba Mostafa, MD, Ph.D, D(ABMM) Dr. Heba Mostafa and her team have taken a non-conventional approach to guide and build a testing algorithm forthe
ESENSOR TECHNOLOGY
eSensor Technology. Use Up/Down Arrow keys to increase or decrease volume. GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information.OPERATOR MANUAL
PI1066 – Rev. G 1 Operator Manual GenMark Diagnostics Inc. EMERCO EUROP 5964 La Place Court Prinsessegracht 20 GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
EPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP)1 and NEW Respiratory Pathogen Panel 2 (RP2)2 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within aHCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive GENMARK DIAGNOSTICSCAREERSCOMPANYRESOURCESSUPPORTSYSTEMSPANELS GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) Panels. Learn More. Respiratory PathogenPanels. Learn More.
EPLEX SYSTEM
The ePlex system is the only sample-to-answer solution on the market that streamlines the diagnostic workflow from physician order entry to the release of the final report. Bi-directional LIS to automate and accelerate order entry and results reporting. Random and continuous access and a modular, scalable design to ensure capacity that meetsEPLEX PANELS
ePlex ® Panels. Infectious diseases, such as lower respiratory infections, bloodstream infections that can lead to sepsis, and diarrhea are among the top causes of death worldwide. 1,2 These and other infectious diseases can be considered syndromes (a group of symptoms that may not point to a specific causative agent) and may be difficult to diagnose using conventional diagnostic testing RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP)1 and NEW Respiratory Pathogen Panel 2 (RP2)2 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19 BLOOD CULTURE IDENTIFICATION (BCID) PANELS Blood Culture Identification (BCID) Panels. The More You Detect, The Better You Protect. GenMark’s ePlex ® Blood Culture Identification (BCID) Panels provide broad coverage of organisms that can lead to sepsis along with their resistance genes. This broad coverage means that about 95% of currently identified bloodstream infections can be detected early with the ePlex BCID Panels, compared ELECTROWETTING TECHNOLOGY Electrowetting uses electrical fields to directly manipulate discrete droplets on the surface of a hydrophobically coated printed circuit board (PCB). Droplets of sample and reagents are moved in a programmable fashion and can be efficiently transferred and transported within aHCVG DIRECT TEST
GenMark’s eSensor HCVg Direct Test is designed for typing and subtyping of HCV 1a, 1b, 2a/c, 2b, 3, 4, 5, and 6 from serum orplasma.
GENMARK RECEIVES FDA 510(K) MARKET CLEARANCE FOR ITS EPLEX The True Sample-to-Answer Solution™ Brings New Capabilities to Multiplex Molecular Diagnostics. CARLSBAD, Calif.--(BUSINESS WIRE)-- GenMark Diagnostics, Inc. (Nasdaq:GNMK), a leading provider of automated, multiplex molecular diagnostic testing systems, today announced that it has received 510(k) market clearance from the U.S. Food and Drug Administration for both its ePlex ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information. EPLEX BLOOD CULTURE IDENTIFICATION PANELS Clinical Impact 4 *ePlex processing time = 90 mins. ePlex BCID panels were only used during working hours so there was a delay in some cases between blood culture flagging positive and being loaded onto the ePlex machine (i.e. if flagged positive TECHNOLOGY | GENMARK DIAGNOSTICS Technology eSensor® Technology GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is not based on fluorescent or optical detection. As a result, diagnostic tests are RESPIRATORY PATHOGEN PANELS Respiratory Pathogen Panel (RP)1 and NEW Respiratory Pathogen Panel 2 (RP2)2 GenMark’s Respiratory Pathogen Panels identify the most common viral and bacterial organisms associated with upper respiratory infection, including SARS-CoV-2, the virus that causes COVID-19XT-8 SYSTEMS
XT-8® Systems XT-8 System Benefits The XT-8 system is the pioneer of true multiplex molecular testing, offering post-PCR detection for infectious disease, genetics, and pharmacogenetics targets. DOWNLOAD XT-8 SYSTEM BROCHURE Simple To Use An intuitive touch-screen user interface and easy-to-interpret reports enable seamless integration of DETECTION OF VARIANT SARS-COV-2 STRAINS ON THE EPLEX Detection of variant SARS-CoV-2 strains on the ePlex ® RP2 Panel * March 22, 2021. Genetic surveillance of SARS-CoV-2 strains circulating globally has identified multiple variants containing one or more mutations that can potentially impact detection by nucleic acid basedtesting methods.
INVESTORS OVERVIEW
Investors Overview. GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. Utilizing GenMark’s proprietary eSensor ® detection technology, GenMark’s eSensor XT-8 ® and ePlex ® systems are designed tosupport a
LOGIN — CUSTOMER RESOURCE CENTER Login to the Customer Resource Center to view SDS Sheets, Package Inserts, Worksheets, Training Presentations and Brochures. WEBINARS: MOLECULAR DIAGNOSTIC SOLUTIONS DESIGNED FOR THE Webinars Molecular Diagnostic Solutions Designed for the Patient and Optimized for the Laboratory in a Pandemic Presented by: Heba Mostafa, MD, Ph.D, D(ABMM) Dr. Heba Mostafa and her team have taken a non-conventional approach to guide and build a testing algorithm forthe
ESENSOR TECHNOLOGY
eSensor Technology. Use Up/Down Arrow keys to increase or decrease volume. GenMark’s eSensor technology is based on the principles of competitive DNA hybridization and electrochemical detection. eSensor technology is highly specific for the target biomarker and is ESENSOR XT-8 USER MANUAL UM0001 Rev A Page 7 of 62 I. Safety Information Important! Before using the eSensor® XT-8™, carefully read this entire manual, including the XT-8 overview and all precautionary information.OPERATOR MANUAL
PI1066 – Rev. G 1 Operator Manual GenMark Diagnostics Inc. EMERCO EUROP 5964 La Place Court Prinsessegracht 20* Panels
* ePlex Panels
* Respiratory
* Blood Culture
* XT-8 Panels
* Systems
* ePlex System
* Software Solutions* Patient Safety
* XT-8 System
* Company
* The GenMark Story
* Management Team
* Board of Directors* Careers
* Opportunities
* Investors
* Newsroom
* Education
* Disease State
* Sepsis
* Respiratory Infections * Genetic Conditions* Hepatitis C
* GI Infections
* Technology
* Resources
* Support
* Contact Us
* FAQs
* Safety Data Sheets (SDS) * Your Long-Term PartnerSELECT YOUR REGION:
* United States
* International
| CONTACT US | CUSTOMER RESOURCE CENTER* Panels
* ePlex Panels
* Respiratory
* Blood Culture
* XT-8 Panels
* Systems
* ePlex System
* Software Solutions* Patient Safety
* XT-8 System
* Company
* The GenMark Story
* Management Team
* Board of Directors* Careers
* Opportunities
* Investors
* Newsroom
* Education
* Disease State
* Sepsis
* Respiratory Infections * Genetic Conditions* Hepatitis C
* GI Infections
* Technology
* Resources
* Support
* Contact Us
* FAQs
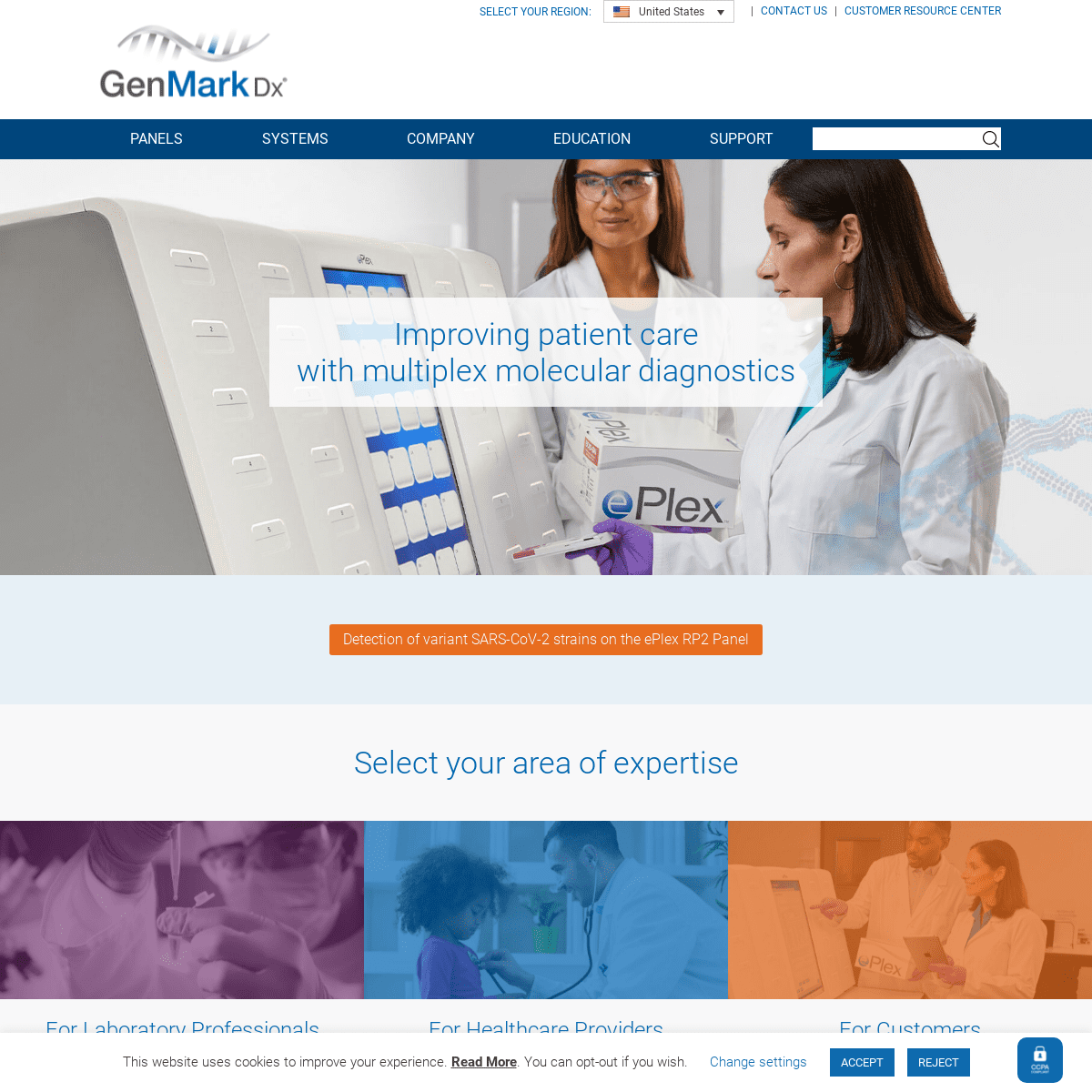
* Safety Data Sheets (SDS) * Your Long-Term Partner IMPROVING PATIENT CARE WITH MULTIPLEX MOLECULAR DIAGNOSTICS Detection of variant SARS-CoV-2 strains on the ePlex RP2 Panel SELECT YOUR AREA OF EXPERTISE FOR LABORATORY PROFESSIONALS FOR HEALTHCARE PROVIDERSFOR CUSTOMERS
EPLEX SYSTEM
THE TRUE SAMPLE-TO-ANSWER SOLUTION® Eliminate unnecessary ED wait times and experience the benefits of rapid comprehensive testing with the ePlex Respiratory Pathogen Panel. Contact GenMark today to learn how we can help you prepare for this flu season with a no commitment evaluation program and special pricing on the ePlex RP Panel. Learn More Request aDemo
EPLEX PANELS
GenMark Diagnostics is committed to addressing the need to improve patient care through the design and development of comprehensive multiplex panels. Our growing infectious disease menu includes: Blood Culture Identification (BCID) PanelsLearn More
Respiratory Pathogen PanelsLearn More
GenMark Welcomes Wade Stevenson, SVP, North America SalesLearn More
Detection of variant SARS-CoV-2 strains on the ePlex RP2 PanelLearn More
Major teaching hospital in Grenoble, France demonstrates that ePlex BCID results could have modified antimicrobial treatment in 45% ofpatients
Learn More
WEBINAR: Navigating the Choppy Waters of Implementing Rapid Molecular Blood Culture Identification TestsRegister Now
PASCV Molecular Virology WorkshopMay 5th – 7th
Virtual Meeting
Visit Site
World Microbe Forum
June 20th – 24th
Virtual
Visit Site
View All News & Events GenMark Diagnostics, Inc.5964 La Place Court
Carlsbad, CA 92008
� Copyright 2021 GenMark Diagnostics, Inc. All rights reserved.info@genmarkdx.com
P: +1 760-448-4300
F: +1 760-448-4301
Legal | Privacy Policy This website uses cookies to improve your experience. Read More . You can opt-out if you wish. Change settingsACCEPT REJECTManage consent
Close
PRIVACY OVERVIEW
This website uses cookies to improve your experience while you navigate through the website. Out of these, the cookies that are categorized as necessary are stored on your browser as they are essential for the working of basic functionalities of the ...Necessary
Necessary Always Enabled Necessary cookies are absolutely essential for the website to function properly. This category only includes cookies that ensures basic functionalities and security features of the website. These cookies do not store any personal information.Non-necessary
Non-necessary
Any cookies that may not be particularly necessary for the website to function and is used specifically to collect user personal data via analytics, ads, other embedded contents are termed as non-necessary cookies. It is mandatory to procure user consent prior to running these cookies on your website. Learn More About Your Privacy Rights Exercise your consumer rights by contacting us below Read our Privacy Policy privacy@genmarkdx.com800-373-6767
OPT OUT SUCCESSFUL
Thank you for exercising your right to opt out Personalized advertisements Personalized advertisements Turning this off will opt you out of personalized advertisements delivered from Google on this website.Confirm
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0