Are you over 18 and want to see adult content?
More Annotations
A complete backup of originalstyle.com
Are you over 18 and want to see adult content?
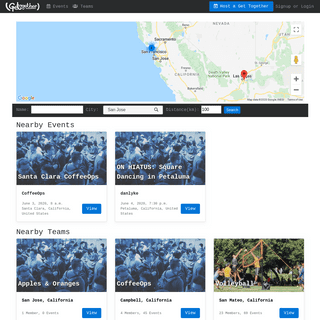
A complete backup of gettogether.community
Are you over 18 and want to see adult content?
A complete backup of talesofabackpacker.com
Are you over 18 and want to see adult content?

A complete backup of essays-writer.org
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of software179.blogspot.com
Are you over 18 and want to see adult content?

A complete backup of freshtrackstransportation.com
Are you over 18 and want to see adult content?

A complete backup of theaffiliatetitan.com
Are you over 18 and want to see adult content?
A complete backup of descargatest2.jimdo.com
Are you over 18 and want to see adult content?

A complete backup of deporvillage.com
Are you over 18 and want to see adult content?

A complete backup of ballettalentevolution.com
Are you over 18 and want to see adult content?
Text
commercialization.
PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | INVESTOR OVERVIEW Investors. As a late clinical-stage biopharmaceutical company focused on oncology, our business model is to in-license novel compounds, develop them through clinical stages, and then partner with pharmaceutical companies for further clinical development and/or commercialization. Our lead program, motixafortide (BL-8040), is acancer therapy
BIOLINERX | BIOLINERX ANNOUNCES FINAL RESULTS FROM PHASE TEL AVIV, Israel, Dec. 16, 2020 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced results from the triple combination arm of the Company's COMBAT/KEYNOTE-202 clinical study evaluating motixafortide (BL-8040) in combination with KEYTRUDA ® (pembrolizumab) and chemotherapy in MOTIXAFORTIDE (BL-8040) Development. Motixafortide - BL-8040 (formerly known as BKT140 and developed by Biokine Therapeutics Ltd.) is a novel selective inhibitor of the CXCR4 chemokine receptor that is currently being developed for several indications including treatment of solid tumors, mobilization of hematopoietic stem cells (HSCs) for autologous transplantations and hematological malignancies. MOTIXAFORTIDE (BL-8040) Motixafortide is a short synthetic peptide used as a platform for cancer immunotherapy with unique features allowing it to function as a best-in-class antagonist of CXCR4. It shows high-affinity, long receptor occupancy and acts as an inverse agonist. In a number of clinical and preclinical studies, motixafortide has been shown toaffect
AGI-134 | SOLID TUMORS | BIOLINERX AGI-134 is a synthetic alpha-Gal glycolipid in development for solid tumors that is highly differentiated from other cancer immunotherapies. AGI-134 is designed to label cancer cells with alpha-Gal via intra-tumoral administration, thereby targeting the body’s pre-existing, highly abundant anti-alpha-Gal (anti-Gal) antibodies and redirecting them to treated tumors. BIOLINERX ANNOUNCES PRESENTATION AT 2021 AMERICAN BioLineRx Announces Presentation at 2021 American Association for Cancer Research (AACR) Annual Meeting April 15, 2021 - Poster includes analysis of results by liver metastases, further strengthening results reported in December 2020 of the A MULTI-CENTER PHASE 2A TRIAL OF THE CXCR4 INHIBITOR A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL-8040) in Combination with Pembrolizumab and Chemotherapy, in Patients with Metastatic Pancreatic Adenocarcinoma THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of HOME | BIOLINERXABOUTPIPELINECLINICAL STUDIESPARTNERINGINVESTORSCAREERS BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | INVESTOR OVERVIEW Investors. As a late clinical-stage biopharmaceutical company focused on oncology, our business model is to in-license novel compounds, develop them through clinical stages, and then partner with pharmaceutical companies for further clinical development and/or commercialization. Our lead program, motixafortide (BL-8040), is acancer therapy
BIOLINERX | BIOLINERX ANNOUNCES FINAL RESULTS FROM PHASE TEL AVIV, Israel, Dec. 16, 2020 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced results from the triple combination arm of the Company's COMBAT/KEYNOTE-202 clinical study evaluating motixafortide (BL-8040) in combination with KEYTRUDA ® (pembrolizumab) and chemotherapy in MOTIXAFORTIDE (BL-8040) Development. Motixafortide - BL-8040 (formerly known as BKT140 and developed by Biokine Therapeutics Ltd.) is a novel selective inhibitor of the CXCR4 chemokine receptor that is currently being developed for several indications including treatment of solid tumors, mobilization of hematopoietic stem cells (HSCs) for autologous transplantations and hematological malignancies. MOTIXAFORTIDE (BL-8040) Motixafortide is a short synthetic peptide used as a platform for cancer immunotherapy with unique features allowing it to function as a best-in-class antagonist of CXCR4. It shows high-affinity, long receptor occupancy and acts as an inverse agonist. In a number of clinical and preclinical studies, motixafortide has been shown toaffect
AGI-134 | SOLID TUMORS | BIOLINERX AGI-134 is a synthetic alpha-Gal glycolipid in development for solid tumors that is highly differentiated from other cancer immunotherapies. AGI-134 is designed to label cancer cells with alpha-Gal via intra-tumoral administration, thereby targeting the body’s pre-existing, highly abundant anti-alpha-Gal (anti-Gal) antibodies and redirecting them to treated tumors. BIOLINERX ANNOUNCES PRESENTATION AT 2021 AMERICAN BioLineRx Announces Presentation at 2021 American Association for Cancer Research (AACR) Annual Meeting April 15, 2021 - Poster includes analysis of results by liver metastases, further strengthening results reported in December 2020 of the A MULTI-CENTER PHASE 2A TRIAL OF THE CXCR4 INHIBITOR A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL-8040) in Combination with Pembrolizumab and Chemotherapy, in Patients with Metastatic Pancreatic Adenocarcinoma THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
CLINICAL STUDIES
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | BIOLINERX ANNOUNCES PRESENTATION AT 2021 TEL AVIV, Israel, April 15, 2021 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced that the Company has presented a poster at the AACR Annual Meeting, which is being held April 10-15 on a virtual basis.". The poster, entitled: "A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL BIOLINERX TO REPORT FIRST QUARTER 2021 RESULTS ON MAY 26 TEL AVIV, Israel, May 20, 2021 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical company focused on oncology, announced today it will release its unaudited financial results for the quarter ended March 31, 2021 on Wednesday, May 26, 2021, before the US markets open.. The Company will host a conference call on Wednesday, May 26, 2021 at MOTIXAFORTIDE (BL-8040) Motixafortide is a short synthetic peptide used as a platform for cancer immunotherapy with unique features allowing it to function as a best-in-class antagonist of CXCR4. It shows high-affinity, long receptor occupancy and acts as an inverse agonist. In a number of clinical and preclinical studies, motixafortide has been shown toaffect
BIOLINERX | SEC FILING “Severe COVID-19 cases, where patients are hospitalized with ARDS and require ventilation, have generated renewed interest in the underlying pathology of acute respiratory stress disorder,” noted Dr. Maor. “Substantial data is emerging regarding the involvement of neutrophils, neutrophil extracellular traps (NETs), monocytes and macrophages in the development of ARDS secondary to COVID BIOLINERX | BIOLINERX ANNOUNCES BL-8040 IN COMBINATION TEL AVIV, Israel, Nov. 5, 2019 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) and (TASE: BLRX), a clinical-stage biopharmaceutical company focused on oncology, announced today results of a Phase 2b trial assessing the efficacy of BL-8040 in combination with KEYTRUDA ® (pembrolizumab) for the treatment of metastatic pancreatic cancer. The data demonstrated that the dual combination BIOLINERX | SEC FILING BioLineRx | SEC Filing. SECURITIES AND EXCHANGE COMMISSION. WASHINGTON, D.C. 20549. FORM 6-K. REPORT OF FOREIGN PRIVATE ISSUER. PURSUANT TO RULE 13a-16 OR 15d-16 OF. THE SECURITIES EXCHANGE ACT OF 1934. For the month of April 2021. Commission file number: 001-35223. THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of B I OL I N E R X L TD u n i t e d s tat e s s e c u r i t i e s a n d e x c h a n g e c o m m i s s i o n washington, d.c. 20549 f o r m 20-f (mark one) ☐ registration statement pursuant to section 12(b) or (g) of thesecurities exchange
HOME | BIOLINERXABOUTPIPELINECLINICAL STUDIESPARTNERINGINVESTORSCAREERS BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
CLINICAL STUDIES
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
MOTIXAFORTIDE (BL-8040) Motixafortide is a short synthetic peptide used as a platform for cancer immunotherapy with unique features allowing it to function as a best-in-class antagonist of CXCR4. It shows high-affinity, long receptor occupancy and acts as an inverse agonist. In a number of clinical and preclinical studies, motixafortide has been shown toaffect
BIOLINERX | INVESTOR OVERVIEW Investors. As a late clinical-stage biopharmaceutical company focused on oncology, our business model is to in-license novel compounds, develop them through clinical stages, and then partner with pharmaceutical companies for further clinical development and/or commercialization. Our lead program, motixafortide (BL-8040), is acancer therapy
BIOLINERX | BIOLINERX ANNOUNCES FINAL RESULTS FROM PHASEBIOLINERX INSTITUTIONAL OWNERSHIPBIOLINERX LTD TEL AVIV, Israel, Dec. 16, 2020 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced results from the triple combination arm of the Company's COMBAT/KEYNOTE-202 clinical study evaluating motixafortide (BL-8040) in combination with KEYTRUDA ® (pembrolizumab) and chemotherapy in MOTIXAFORTIDE (BL-8040) Development. Motixafortide - BL-8040 (formerly known as BKT140 and developed by Biokine Therapeutics Ltd.) is a novel selective inhibitor of the CXCR4 chemokine receptor that is currently being developed for several indications including treatment of solid tumors, mobilization of hematopoietic stem cells (HSCs) for autologous transplantations and hematological malignancies. AGI-134 | OVERVIEW | BIOLINERX AGI-134 is a synthetic alpha-Gal glycolipid in development for solid tumors that is highly differentiated from other cancer immunotherapies. AGI-134 is designed to label cancer cells with alpha-Gal via intra-tumoral administration, thereby targeting the body’s pre-existing, highly abundant anti-alpha-Gal (anti-Gal) antibodies and redirecting them to treated tumors. A MULTI-CENTER PHASE 2A TRIAL OF THE CXCR4 INHIBITOR A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL-8040) in Combination with Pembrolizumab and Chemotherapy, in Patients with Metastatic Pancreatic Adenocarcinoma THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of HOME | BIOLINERXABOUTPIPELINECLINICAL STUDIESPARTNERINGINVESTORSCAREERS BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
CLINICAL STUDIES
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
MOTIXAFORTIDE (BL-8040) Motixafortide is a short synthetic peptide used as a platform for cancer immunotherapy with unique features allowing it to function as a best-in-class antagonist of CXCR4. It shows high-affinity, long receptor occupancy and acts as an inverse agonist. In a number of clinical and preclinical studies, motixafortide has been shown toaffect
BIOLINERX | INVESTOR OVERVIEW Investors. As a late clinical-stage biopharmaceutical company focused on oncology, our business model is to in-license novel compounds, develop them through clinical stages, and then partner with pharmaceutical companies for further clinical development and/or commercialization. Our lead program, motixafortide (BL-8040), is acancer therapy
BIOLINERX | BIOLINERX ANNOUNCES FINAL RESULTS FROM PHASEBIOLINERX INSTITUTIONAL OWNERSHIPBIOLINERX LTD TEL AVIV, Israel, Dec. 16, 2020 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced results from the triple combination arm of the Company's COMBAT/KEYNOTE-202 clinical study evaluating motixafortide (BL-8040) in combination with KEYTRUDA ® (pembrolizumab) and chemotherapy in MOTIXAFORTIDE (BL-8040) Development. Motixafortide - BL-8040 (formerly known as BKT140 and developed by Biokine Therapeutics Ltd.) is a novel selective inhibitor of the CXCR4 chemokine receptor that is currently being developed for several indications including treatment of solid tumors, mobilization of hematopoietic stem cells (HSCs) for autologous transplantations and hematological malignancies. AGI-134 | OVERVIEW | BIOLINERX AGI-134 is a synthetic alpha-Gal glycolipid in development for solid tumors that is highly differentiated from other cancer immunotherapies. AGI-134 is designed to label cancer cells with alpha-Gal via intra-tumoral administration, thereby targeting the body’s pre-existing, highly abundant anti-alpha-Gal (anti-Gal) antibodies and redirecting them to treated tumors. A MULTI-CENTER PHASE 2A TRIAL OF THE CXCR4 INHIBITOR A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL-8040) in Combination with Pembrolizumab and Chemotherapy, in Patients with Metastatic Pancreatic Adenocarcinoma THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of BIOLINERX | SEC FILING Tel Aviv, Israel, October 29, 2020 – BioLineRx Ltd. (NASDAQ/TASE:BLRX), a clinical-stage biopharmaceutical Company focused on oncology, today announced that the Company’s lead drug candidate, the CXCR4-inhibitor Motixafortide, will be tested in combination with the anti-PD-1 cemiplimab (LIBTAYO ®) and standard-of-care chemotherapy (gemcitabine and nab-paclitaxel) in first-lineCAREERS | BIOLINERX
Careers. Successful drug development is comprised of multiple disciplines and activities that cut across compound types and indications, including ADME, toxicology, pharmacokinetics, CMC, and clinical trial management. We integrate a core function-specific development team with OUR TEAM | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BOARD OF DIRECTORS
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
MOTIXAFORTIDE (BL-8040) Motixafortide is a short synthetic peptide used as a platform for cancer immunotherapy with unique features allowing it to function as a best-in-class antagonist of CXCR4. It shows high-affinity, long receptor occupancy and acts as an inverse agonist. In a number of clinical and preclinical studies, motixafortide has been shown toaffect
SHARE CHART
Share Chart. Chart. Select share. BioLineRx ADS BioLineRx. Select market. NASDAQ. Ticker table to check the latest price updates. Date & Time: 23 April 2021 23:00 (GMT+03:00) Share. BIOLINERX ANNOUNCES PRESENTATION AT 2021 AMERICAN BioLineRx Announces Presentation at 2021 American Association for Cancer Research (AACR) Annual Meeting April 15, 2021 - Poster includes analysis of results by liver metastases, further strengthening results reported in December 2020 of the BIOLINERX | MALI ZEEVI, CPA | MANAGEMENT Before joining BioLineRx, Ms. Zeevi was employed by Tescom Software Systems Testing Ltd., her last position there being Vice President Finance. Ms. Zeevi also served as a CPA at Kesselman & Kesselman, a member firm of PricewaterhouseCoopers International Limited. She holds a B.A. in business and accountancy from the College of Management THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of BIOLINERX ANNOUNCES ACQUISITION OF AGALIMMUNE LTD. TO BioLineRx Announces Acquisition of Agalimmune Ltd. to Accelerate Expansion of Immuno-Oncology Pipeline March 23, 2017 Agalimmune's Lead Asset, AGI-134, Provides a Unique Approach for Eliciting Patient-specific, Anti-tumor Immune HOME | BIOLINERXABOUTPIPELINECLINICAL STUDIESPARTNERINGINVESTORSCAREERS BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
CLINICAL STUDIES
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | INVESTOR OVERVIEW May 26, 2021. BioLineRx Reports First Quarter 2021 Financial Results and Provides Corporate Update. May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021 MOTIXAFORTIDE (BL-8040) Stem-cell mobilization. In March 2015, we reported successful top-line safety and efficacy results from a Phase 1 safety and efficacy trial for the use of motixafortide as a novel hematopoietic stem-cell mobilization treatment for allogeneic bone marrow transplantation. MOTIXAFORTIDE (BL-8040) Development. Motixafortide - BL-8040 (formerly known as BKT140 and developed by Biokine Therapeutics Ltd.) is a novel selective inhibitor of the CXCR4 chemokine receptor that is currently being developed for several indications including treatment of solid tumors, mobilization of hematopoietic stem cells (HSCs) for autologous transplantations and hematological malignancies. BIOLINERX | BIOLINERX ANNOUNCES FINAL RESULTS FROM PHASEBIOLINERX INSTITUTIONAL OWNERSHIPBIOLINERX LTD TEL AVIV, Israel, Dec. 16, 2020 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced results from the triple combination arm of the Company's COMBAT/KEYNOTE-202 clinical study evaluating motixafortide (BL-8040) in combination with KEYTRUDA ® (pembrolizumab) and chemotherapy in AGI-134 | OVERVIEW | BIOLINERX AGI-134 is a synthetic alpha-Gal glycolipid in development for solid tumors that is highly differentiated from other cancer immunotherapies. AGI-134 is designed to label cancer cells with alpha-Gal via intra-tumoral administration, thereby targeting the body’s pre-existing, highly abundant anti-alpha-Gal (anti-Gal) antibodies and redirecting them to treated tumors. THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of A MULTI-CENTER PHASE 2A TRIAL OF THE CXCR4 INHIBITOR A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL-8040) in Combination with Pembrolizumab and Chemotherapy, in Patients with Metastatic Pancreatic Adenocarcinoma HOME | BIOLINERXABOUTPIPELINECLINICAL STUDIESPARTNERINGINVESTORSCAREERS BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
CLINICAL STUDIES
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | INVESTOR OVERVIEW May 26, 2021. BioLineRx Reports First Quarter 2021 Financial Results and Provides Corporate Update. May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021 MOTIXAFORTIDE (BL-8040) Stem-cell mobilization. In March 2015, we reported successful top-line safety and efficacy results from a Phase 1 safety and efficacy trial for the use of motixafortide as a novel hematopoietic stem-cell mobilization treatment for allogeneic bone marrow transplantation. MOTIXAFORTIDE (BL-8040) Development. Motixafortide - BL-8040 (formerly known as BKT140 and developed by Biokine Therapeutics Ltd.) is a novel selective inhibitor of the CXCR4 chemokine receptor that is currently being developed for several indications including treatment of solid tumors, mobilization of hematopoietic stem cells (HSCs) for autologous transplantations and hematological malignancies. BIOLINERX | BIOLINERX ANNOUNCES FINAL RESULTS FROM PHASEBIOLINERX INSTITUTIONAL OWNERSHIPBIOLINERX LTD TEL AVIV, Israel, Dec. 16, 2020 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced results from the triple combination arm of the Company's COMBAT/KEYNOTE-202 clinical study evaluating motixafortide (BL-8040) in combination with KEYTRUDA ® (pembrolizumab) and chemotherapy in AGI-134 | OVERVIEW | BIOLINERX AGI-134 is a synthetic alpha-Gal glycolipid in development for solid tumors that is highly differentiated from other cancer immunotherapies. AGI-134 is designed to label cancer cells with alpha-Gal via intra-tumoral administration, thereby targeting the body’s pre-existing, highly abundant anti-alpha-Gal (anti-Gal) antibodies and redirecting them to treated tumors. THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of A MULTI-CENTER PHASE 2A TRIAL OF THE CXCR4 INHIBITOR A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL-8040) in Combination with Pembrolizumab and Chemotherapy, in Patients with Metastatic Pancreatic Adenocarcinoma BIOLINERX | SEC FILING Tel Aviv, Israel, October 29, 2020 – BioLineRx Ltd. (NASDAQ/TASE:BLRX), a clinical-stage biopharmaceutical Company focused on oncology, today announced that the Company’s lead drug candidate, the CXCR4-inhibitor Motixafortide, will be tested in combination with the anti-PD-1 cemiplimab (LIBTAYO ®) and standard-of-care chemotherapy (gemcitabine and nab-paclitaxel) in first-lineCAREERS | BIOLINERX
Successful drug development is comprised of multiple disciplines and activities that cut across compound types and indications, including ADME, toxicology, phar PARTNERING | BIOLINERX In-Licensing. We are committed to the identification, in-licensing and systematic development of promising therapeutic candidates. Via in-licensing initiatives, we are eager to evaluate therapeutic frontiers in late-preclinical and clinical stages of development withan emphasis on
OUR TEAM | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | BIOLINERX ANNOUNCES PRESENTATION AT 2021 TEL AVIV, Israel, April 15, 2021 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced that the Company has presented a poster at the AACR Annual Meeting, which is being held April 10-15 on a virtual basis.". The poster, entitled: "A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BLSHARE CHART
May 26, 2021. BioLineRx Reports First Quarter 2021 Financial Results and Provides Corporate Update. May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021BOARD OF DIRECTORS
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
ANNUAL REPORTS
May 26, 2021. BioLineRx Reports First Quarter 2021 Financial Results and Provides Corporate Update. May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021 BIOLINERX | MALI ZEEVI, CPA | MANAGEMENT May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021. May 4, 2021. BioLineRx Announces Positive Top-Line Results from GENESIS Phase 3 Trial of Motixafortide in Stem-Cell Mobilization for Autologous Bone Marrow Transplantation in Multiple MyelomaPatients
BIOLINERX ANNOUNCES PRESENTATION AT 2021 AMERICAN BioLineRx Announces Presentation at 2021 American Association for Cancer Research (AACR) Annual Meeting April 15, 2021 - Poster includes analysis of results by liver metastases, further strengthening results reported in December 2020 of the HOME | BIOLINERXABOUTPIPELINECLINICAL STUDIESPARTNERINGINVESTORSCAREERS BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
CLINICAL STUDIES
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | INVESTOR OVERVIEW May 26, 2021. BioLineRx Reports First Quarter 2021 Financial Results and Provides Corporate Update. May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021 MOTIXAFORTIDE (BL-8040) Stem-cell mobilization. In March 2015, we reported successful top-line safety and efficacy results from a Phase 1 safety and efficacy trial for the use of motixafortide as a novel hematopoietic stem-cell mobilization treatment for allogeneic bone marrow transplantation. MOTIXAFORTIDE (BL-8040) Development. Motixafortide - BL-8040 (formerly known as BKT140 and developed by Biokine Therapeutics Ltd.) is a novel selective inhibitor of the CXCR4 chemokine receptor that is currently being developed for several indications including treatment of solid tumors, mobilization of hematopoietic stem cells (HSCs) for autologous transplantations and hematological malignancies. BIOLINERX | BIOLINERX ANNOUNCES FINAL RESULTS FROM PHASEBIOLINERX INSTITUTIONAL OWNERSHIPBIOLINERX LTD TEL AVIV, Israel, Dec. 16, 2020 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced results from the triple combination arm of the Company's COMBAT/KEYNOTE-202 clinical study evaluating motixafortide (BL-8040) in combination with KEYTRUDA ® (pembrolizumab) and chemotherapy in AGI-134 | OVERVIEW | BIOLINERX AGI-134 is a synthetic alpha-Gal glycolipid in development for solid tumors that is highly differentiated from other cancer immunotherapies. AGI-134 is designed to label cancer cells with alpha-Gal via intra-tumoral administration, thereby targeting the body’s pre-existing, highly abundant anti-alpha-Gal (anti-Gal) antibodies and redirecting them to treated tumors. THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of A MULTI-CENTER PHASE 2A TRIAL OF THE CXCR4 INHIBITOR A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL-8040) in Combination with Pembrolizumab and Chemotherapy, in Patients with Metastatic Pancreatic Adenocarcinoma HOME | BIOLINERXABOUTPIPELINECLINICAL STUDIESPARTNERINGINVESTORSCAREERS BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
PIPELINE | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
CLINICAL STUDIES
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | INVESTOR OVERVIEW May 26, 2021. BioLineRx Reports First Quarter 2021 Financial Results and Provides Corporate Update. May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021 MOTIXAFORTIDE (BL-8040) Stem-cell mobilization. In March 2015, we reported successful top-line safety and efficacy results from a Phase 1 safety and efficacy trial for the use of motixafortide as a novel hematopoietic stem-cell mobilization treatment for allogeneic bone marrow transplantation. MOTIXAFORTIDE (BL-8040) Development. Motixafortide - BL-8040 (formerly known as BKT140 and developed by Biokine Therapeutics Ltd.) is a novel selective inhibitor of the CXCR4 chemokine receptor that is currently being developed for several indications including treatment of solid tumors, mobilization of hematopoietic stem cells (HSCs) for autologous transplantations and hematological malignancies. BIOLINERX | BIOLINERX ANNOUNCES FINAL RESULTS FROM PHASEBIOLINERX INSTITUTIONAL OWNERSHIPBIOLINERX LTD TEL AVIV, Israel, Dec. 16, 2020 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced results from the triple combination arm of the Company's COMBAT/KEYNOTE-202 clinical study evaluating motixafortide (BL-8040) in combination with KEYTRUDA ® (pembrolizumab) and chemotherapy in AGI-134 | OVERVIEW | BIOLINERX AGI-134 is a synthetic alpha-Gal glycolipid in development for solid tumors that is highly differentiated from other cancer immunotherapies. AGI-134 is designed to label cancer cells with alpha-Gal via intra-tumoral administration, thereby targeting the body’s pre-existing, highly abundant anti-alpha-Gal (anti-Gal) antibodies and redirecting them to treated tumors. THE CXCR4 ANTAGONIST BL-8040 SYNERGIZES WITH THE FLT3 Conclusions Survival of AML cells in the blood following BL-8040 treatment or in combination with AC220 Background Acute Myeloid Leukemia (AML) is a heterogeneous group of A MULTI-CENTER PHASE 2A TRIAL OF THE CXCR4 INHIBITOR A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BL-8040) in Combination with Pembrolizumab and Chemotherapy, in Patients with Metastatic Pancreatic Adenocarcinoma BIOLINERX | SEC FILING Tel Aviv, Israel, October 29, 2020 – BioLineRx Ltd. (NASDAQ/TASE:BLRX), a clinical-stage biopharmaceutical Company focused on oncology, today announced that the Company’s lead drug candidate, the CXCR4-inhibitor Motixafortide, will be tested in combination with the anti-PD-1 cemiplimab (LIBTAYO ®) and standard-of-care chemotherapy (gemcitabine and nab-paclitaxel) in first-lineCAREERS | BIOLINERX
Successful drug development is comprised of multiple disciplines and activities that cut across compound types and indications, including ADME, toxicology, phar PARTNERING | BIOLINERX In-Licensing. We are committed to the identification, in-licensing and systematic development of promising therapeutic candidates. Via in-licensing initiatives, we are eager to evaluate therapeutic frontiers in late-preclinical and clinical stages of development withan emphasis on
OUR TEAM | BIOLINERX BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
BIOLINERX | BIOLINERX ANNOUNCES PRESENTATION AT 2021 TEL AVIV, Israel, April 15, 2021 /PRNewswire/ -- BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a late clinical-stage biopharmaceutical Company focused on oncology, today announced that the Company has presented a poster at the AACR Annual Meeting, which is being held April 10-15 on a virtual basis.". The poster, entitled: "A Multi-Center Phase 2a Trial of the CXCR4 inhibitor Motixafortide (BLSHARE CHART
May 26, 2021. BioLineRx Reports First Quarter 2021 Financial Results and Provides Corporate Update. May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021BOARD OF DIRECTORS
BioLineRx is a clinical-stage biopharmaceutical company focused on oncology. The Company in-licenses novel compounds, develops them through pre-clinical and/or clinical stages, and then partners with pharmaceutical companies for advanced clinical development and/orcommercialization.
ANNUAL REPORTS
May 26, 2021. BioLineRx Reports First Quarter 2021 Financial Results and Provides Corporate Update. May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021 BIOLINERX | MALI ZEEVI, CPA | MANAGEMENT May 20, 2021. BioLineRx to Report First Quarter 2021 Results on May 26, 2021. May 4, 2021. BioLineRx Announces Positive Top-Line Results from GENESIS Phase 3 Trial of Motixafortide in Stem-Cell Mobilization for Autologous Bone Marrow Transplantation in Multiple MyelomaPatients
BIOLINERX ANNOUNCES PRESENTATION AT 2021 AMERICAN BioLineRx Announces Presentation at 2021 American Association for Cancer Research (AACR) Annual Meeting April 15, 2021 - Poster includes analysis of results by liver metastases, further strengthening results reported in December 2020 of theBioLineRx
* About
* Our Team
* Board of Directors* Pipeline
* Motixafortide (BL-8040)* AGI-134
* Clinical Studies
* Partnering
* Investors
* Share Performance
* News & Events
* Financials
* Analyst Coverage
* SEC Filings
* Corporate Governance * Information Request* IR Contacts
* Careers
* Contact
* About
* Back
* About
* Our Team
* Board of Directors* Pipeline
* Back
* Pipeline
* Motixafortide (BL-8040)* AGI-134
* Clinical Studies
* Partnering
* Investors
* Back
* Share Performance
* News & Events
* Financials
* Analyst Coverage
* SEC Filings
* Corporate Governance * Information Request* IR Contacts
* Careers
* Contact
TRANSFORMING SCIENCE INTO MEDICINE ‘We are committed to developing novel compounds in oncology that will deliver life-changing innovations for patients.’Philip Serlin, CEO
PRESS RELEASE MAY 26, 2021 BIOLINERX REPORTS FIRST QUARTER 2021 FINANCIAL RESULTS AND PROVIDES…EVENTS
Corporate PresentationPIPELINE
AGI-134
SOLID TUMORS
AGI-134 is a universal drug that evokes a vaccine effect via a unique, hyper-acute, multi-arm mechanism that targets patient-specificneoantigens
PIPELINE
MOTIXAFORTIDE (BL-8040) STEM CELL MOBILIZATION Motixafortide (BL-8040) blocks the interaction between CXCL12 and CXCR4 leading to robust mobilization of stem cells to the peripheral blood for collection and transplant.PIPELINE
MOTIXAFORTIDE (BL-8040) ACUTE MYELOID LEUKEMIA (AML) Motixafortide (BL-8040) mobilizes leukemic cells from bone marrow’s protective niche resulting in sensitization to anti-cancer treatment; induction of apoptosis.PIPELINE
MOTIXAFORTIDE (BL-8040)SOLID TUMORS
Motixafortide (BL-8040) mobilizes immune cells to peripheral blood circulation, leading to Increase in immune cell infiltration into tumors and reduction in immunosuppression in tumor microenvironment.PIPELINE
AGI-134
SOLID TUMORS
AGI-134 is a universal drug that evokes a vaccine effect via a unique, hyper-acute, multi-arm mechanism that targets patient-specificneoantigens
PIPELINE
MOTIXAFORTIDE (BL-8040) STEM CELL MOBILIZATION Motixafortide (BL-8040) blocks the interaction between CXCL12 and CXCR4 leading to robust mobilization of stem cells to the peripheral blood for collection and transplant.PIPELINE
MOTIXAFORTIDE (BL-8040) ACUTE MYELOID LEUKEMIA (AML) Motixafortide (BL-8040) mobilizes leukemic cells from bone marrow’s protective niche resulting in sensitization to anti-cancer treatment; induction of apoptosis.PIPELINE
MOTIXAFORTIDE (BL-8040)SOLID TUMORS
Motixafortide (BL-8040) mobilizes immune cells to peripheral blood circulation, leading to Increase in immune cell infiltration into tumors and reduction in immunosuppression in tumor microenvironment.PIPELINE
AGI-134
SOLID TUMORS
AGI-134 is a universal drug that evokes a vaccine effect via a unique, hyper-acute, multi-arm mechanism that targets patient-specificneoantigens
View Pipeline Stem Cell MobilizationMotixafortide (BL-8040) AMLMotixafortide (BL-8040) Solid TumorsMotixafortide (BL-8040) Solid TumorsAGI-134PARTNERING
IN-LICENSING
BioLineRx is committed to the identification, in-licensing and systematic development of promising therapeutic candidates. Through this strategy, BioLineRx is building a pipeline of powerful technologies in oncology.OUT-LICENSING
BioLineRx seeks to collaborate with leading global pharmaceutical companies with a proven track record in clinical experience, for completion of the final phases of the clinical trials, and commercial capabilities, to successfully bring our innovative developments to market. We believe that this is the way to combine our drug development expertise with your commercial development capabilities.INVESTORS
CHANGE 0.11 (4.01 %)VOLUME 1,151,941
EXCHANGE NASDAQ
2.74
58.40
CHANGE 3.80 (6.51 %)VOLUME 1,982,921
EXCHANGE TEL-AVIV
Delayed up to 30 minutes - See terms Delayed up to 30 minutes - See terms Investors Share PerformanceAnalyst
Coverage Press ReleasesBioLineRx
�2021 BioLineRx Ltd. All rights reserved. Terms Privacy & CookiesDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0