Are you over 18 and want to see adult content?
More Annotations
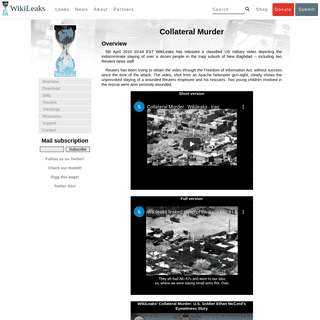
A complete backup of collateralmurder.com
Are you over 18 and want to see adult content?

A complete backup of bridalshapewear.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of khabar.ndtv.com/news/health/what-is-surrogacy-shilpa-shetty-raj-kundra-become-parents-to-a-baby-girl-born-t
Are you over 18 and want to see adult content?

A complete backup of www.shine.cn/biz/economy/2002202403/
Are you over 18 and want to see adult content?
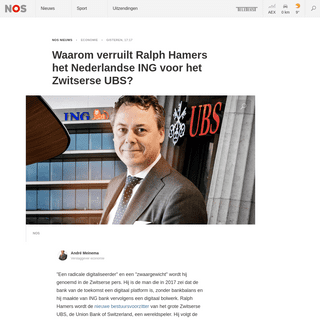
A complete backup of nos.nl/l/2323874
Are you over 18 and want to see adult content?

A complete backup of aajtak.intoday.in/gallery/gold-reserves-development-of-india-positive-report-sonbhadra-up-gold-in-the-hill-
Are you over 18 and want to see adult content?
Text
cosmetics
SAFETY ASSESSMENT OF ROSA DAMASCENA-DERIVED INGREDIENTS AS Safety Assessment of Rosa damascena-derived Ingredients . as Used in Cosmetics . Status: Scientific Literature Review for Public Comment . Release Date: November 19, 2020 . Panel Meeting Date: March 11 – 12,2021
SAFETY ASSESSMENT OF ETHERS AND ESTERS OF ASCORBIC ACID AS According to one source, l-ascorbic acid is an unstable molecule to formulate for topical use, and more stable derivatives of l-ascorbic acid have been utilized in topical formulations.29 Although esters of ascorbic acid are more stable and readily converted to L-ascorbic acid after oral ingestion, it is not clear that derivatives, after topicalapplication, are
SAFETY ASSESSMENT OF HYDROXYACETOPHENONE AS USED IN COSMETICS INTRODUCTION This assessment reviews the safety of Hydroxyacetophenone as used in cosmetic formulations. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), this ingredient is reported to function in cosmetics as an antioxidant and skin-conditioning agent.1 AMENDED SAFETY ASSESSMENT OF SARCOSINES AND SARCOSINATE The Panel voted unanimously in favor of issuing a Tentative Report with the following conclusion: On the basis of the animal and clinical data presented in this report, the Expert Panel concluded that Cocoyl Sarcosine, Lauroyl Sarcosine, Myristoyl SAFETY ASSESSMENT OF SQUALANE AND SQUALENE AS USED IN Minutes from the 1st Re-Review of Squalene and Squalane – September 10-11, 2001 Dr. Belsito noted that a CIR Final Report with the following conclusion on Squalene and Squalane was published in 1982: On the basis of the available information presented FINAL SAFETY ASSESSMENT ON THE SAFETY ASSESSMENT OF ALKYL Final Safety Assessment On the Safety Assessment of Alkyl Glyceryl Ethers As Used in Cosmetics December 19, 2011 The 2011 Cosmetic Ingredient Review Expert Panel members are: Chair, Wilma F. Bergfeld, M.D., F.A.C.P.; Donald V. SAFETY ASSESSMENT OF SILK PROTEINS AS USED IN COSMETICS rat for 24 h. Gauze moistened with physiological saline served as the control. The application of silk protein film did not result in any abnormal clinical signs during the 14-day observation period, and body weights, biochemical parameters and SAFETY ASSESSMENT OF GLYCERIN AS USED IN COSMETICS ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). Glycerin is reported to be used in 15 654 cosmetic products; 10 046 are leave-on products, 5441 are rinse-off products, and CHEMISTRY OF HAIR COLORING Chemistry of Hair Coloring CIR Expert Panel Meeting December 4, 2017 . Carsten Goebel, Ph.D. Cosmetics Europe . Hair Colorant Product Safety COSMETIC INGREDIENT REVIEWABOUTPANEL MEETINGSINGREDIENTSCIR FINDINGS & RESOURCE DOCUMENTSEVENTSHOW DOES CIR WORK The Cosmetic Ingredient Review (CIR) studies individual chemical compounds as they are used in cosmetic products. CIR relies heavily on the International Nomenclature of Cosmetic Ingredients (INCI) when identifying the ingredients to be assessed. 40 Years of CIR. For 40 years the Cosmetic Ingredient Review has worked with FDA, thecosmetics
SAFETY ASSESSMENT OF ROSA DAMASCENA-DERIVED INGREDIENTS AS Safety Assessment of Rosa damascena-derived Ingredients . as Used in Cosmetics . Status: Scientific Literature Review for Public Comment . Release Date: November 19, 2020 . Panel Meeting Date: March 11 – 12,2021
SAFETY ASSESSMENT OF ETHERS AND ESTERS OF ASCORBIC ACID AS According to one source, l-ascorbic acid is an unstable molecule to formulate for topical use, and more stable derivatives of l-ascorbic acid have been utilized in topical formulations.29 Although esters of ascorbic acid are more stable and readily converted to L-ascorbic acid after oral ingestion, it is not clear that derivatives, after topicalapplication, are
SAFETY ASSESSMENT OF HYDROXYACETOPHENONE AS USED IN COSMETICS INTRODUCTION This assessment reviews the safety of Hydroxyacetophenone as used in cosmetic formulations. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), this ingredient is reported to function in cosmetics as an antioxidant and skin-conditioning agent.1 AMENDED SAFETY ASSESSMENT OF SARCOSINES AND SARCOSINATE The Panel voted unanimously in favor of issuing a Tentative Report with the following conclusion: On the basis of the animal and clinical data presented in this report, the Expert Panel concluded that Cocoyl Sarcosine, Lauroyl Sarcosine, Myristoyl SAFETY ASSESSMENT OF SQUALANE AND SQUALENE AS USED IN Minutes from the 1st Re-Review of Squalene and Squalane – September 10-11, 2001 Dr. Belsito noted that a CIR Final Report with the following conclusion on Squalene and Squalane was published in 1982: On the basis of the available information presented FINAL SAFETY ASSESSMENT ON THE SAFETY ASSESSMENT OF ALKYL Final Safety Assessment On the Safety Assessment of Alkyl Glyceryl Ethers As Used in Cosmetics December 19, 2011 The 2011 Cosmetic Ingredient Review Expert Panel members are: Chair, Wilma F. Bergfeld, M.D., F.A.C.P.; Donald V. SAFETY ASSESSMENT OF SILK PROTEINS AS USED IN COSMETICS rat for 24 h. Gauze moistened with physiological saline served as the control. The application of silk protein film did not result in any abnormal clinical signs during the 14-day observation period, and body weights, biochemical parameters and SAFETY ASSESSMENT OF GLYCERIN AS USED IN COSMETICS ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). Glycerin is reported to be used in 15 654 cosmetic products; 10 046 are leave-on products, 5441 are rinse-off products, and CHEMISTRY OF HAIR COLORING Chemistry of Hair Coloring CIR Expert Panel Meeting December 4, 2017 . Carsten Goebel, Ph.D. Cosmetics Europe . Hair Colorant Product Safety SAFETY ASSESSMENT OF DIMETHICONE, METHICONE, AND Frequency and concentration of use has generally increased for these ingredients, with some of the increases being quite significant. Particularly, the frequency of use of Dimethicone has increasedfrom 1659 reported uses in 1998 to 12,934DIPROPYLENE GLYCOL
1620 L St NW Suite 1200 Washington DC 20036-4702 phone: 202-331-0651fax: 202-331-0088
FATTY ESTER END-CAPPED ALKOXYLATES NTP Fatty Ester End-Capped Alkoxylates NTP > Download PDF (209.67kB, 2PP) Download PDF (209.67kB, 2PP) 1620 L St NW Suite 1200 Washington DC20036-4702
SAFETY ASSESSMENT OF RED ALGAE-DERIVED INGREDIENTS AS USED ABSTRACT The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of red algae-derived ingredients; 60 red algae-derived ingredients were found in the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), however, several of these ingredients may be equivalent according to accepted scientific names. SAFETY ASSESSMENT OF MANNITOL, SORBITOL, AND XYLITOL AS Enclosed is the Draft Tentative Report on the Safety Assessment of Mannitol, Sorbitol, and Xylitol as Used in Cosmetics (identified as xylito092019rep in the pdf document). At the April 2019 meeting, the Panel issued an insufficient data FINAL SAFETY ASSESSMENT ON THE SAFETY ASSESSMENT OF ALKYL Final Safety Assessment On the Safety Assessment of Alkyl Glyceryl Ethers As Used in Cosmetics December 19, 2011 The 2011 Cosmetic Ingredient Review Expert Panel members are: Chair, Wilma F. Bergfeld, M.D., F.A.C.P.; Donald V. SAFETY ASSESSMENT OF EDTA & SALTS AS USED IN COSMETICS Safety Assessment of EDTA & Salts as Used in Cosmetics Status: Re-Review for Panel Review Release Date: May 10, 2019 Panel Meeting Date: June 6-7, 2019 SAFETY ASSESSMENT OF CHLORPHENESIN AS USED IN COSMETICS 1 ABSTRACT: Chlorphenesin functions as a preservative, and is used at concentrations up to 0.32% in rinse-off products and up to 0.3 % in leave-on products. The Cosmetic Ingredient Review (CIR) Expert Panel (The Panel) noted that chlorphenesin was well-absorbed when applied to the skin of rats; however, their concern was minimized because of thenegative toxicity
SAFETY ASSESSMENT OF ALKANOYL LACTYL LACTATE SALTS AS USED The Panel issued a tentative report for public comment with the conclusion that the 10 alkyl lactyl lactate salts listed below are safe in cosmetics in the present practices of use and concentration described in the safety assessment, SAFETY ASSESSMENT OF ALKYL BETAINES AS USED IN COSMETICS INTRODUCTION This safety assessment addresses the safety of 11 alkyl betaines as used in cosmetics. The parent compound, betaine, is a naturally occurring N-trimethylated amino acid, also called trimethylglycine, and can be isolated from sugar beets. This website uses cookies to ensure you get the best experience on ourwebsite. Learn more
Got it!
* Home
* About
* Panel Meetings
* Ingredients
* CIR Findings & Resource Documents* Events
About CIR
The Cosmetic Ingredient Review (CIR) studies individual chemical compounds as they are used in cosmetic products. CIR relies heavily on the International Nomenclature of Cosmetic Ingredients (INCI) when identifying the ingredients to be assessed. Find Ingredient Reviews The panel books presented to the Expert Panel for Cosmetic Ingredient Safety contain safety assessments and other materials related to the ingredient or ingredients being considered.40 Years of CIR
For 40 years the Cosmetic Ingredient Review has worked with FDA, the cosmetics industry, and consumers to help keep cosmetics safe.1
2
3
CURRENTLY UNDER REVIEWPreviousNext
1 of 10
DOWNLOAD FULL REPORT > Safety Assessment of Dimethicone, Methicone, and Substituted-Methicone Polymers, as Used in Cosmetics> Read More
DOWNLOAD FULL REPORT > Admin; Memo, Agenda, Minutes, RRSUMS, Strategy Memo, ResourceDocument
> Read More
DOWNLOAD FULL REPORT > Safety Assessment of Caprylhydroxamic Acid as Used in Cosmetics> Read More
DOWNLOAD FULL REPORT > Safety Assessment of Cocos nucifera (Coconut)-Derived Ingredients asUsed in Cosmetics
> Read More
DOWNLOAD FULL REPORT > Amended Safety Assessment of Methylchloroisothiazolinone and Methylisothiazolinone as Used in Cosmetics> Read More
DOWNLOAD FULL REPORT > Safety Assessment of Punica granatum (Pomegranate)-Derived Ingredients as Used in Cosmetics> Read More
DOWNLOAD FULL REPORT > Safety Assessment of Glycerin Ethoxylates as Used in Cosmetics> Read More
DOWNLOAD FULL REPORT > Safety Assessment of Alkyl Amide MIPA Ingredients as Used inCosmetics
> Read More
DOWNLOAD FULL REPORT > Safety Assessment of Beta-Alanine Diacetic Acid and Tetrasodium Glutamate Diacetate as Used in Cosmetics> Read More
DOWNLOAD FULL REPORT > Safety Assessment of Vanilla-Derived Ingredients as Used in Cosmetics> Read More
UPCOMING PANEL MEETINGS 154th Expert Panel Meeting Jun 08, 2020 - Jun 09, 2020Washington
MOST RECENT PANEL MEETING 153rd CIR Expert Panel Meeting Dec 09, 2019 - Dec 10, 2019Washington
1620 L St NW Suite 1200 Washington DC 20036-4702 phone: 202-331-0651 fax: 202-331-0088* Help
* Privacy
* Terms of Use
* Feedback
* About
� 2016 Cosmetic Ingredient ReviewDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0