Are you over 18 and want to see adult content?
More Annotations

A complete backup of in-depthoutdoors.com
Are you over 18 and want to see adult content?

A complete backup of cuidamemucho.com.ar
Are you over 18 and want to see adult content?
Favourite Annotations
A complete backup of hxshuma.tmall.com
Are you over 18 and want to see adult content?
A complete backup of areolasupreme.tumblr.com
Are you over 18 and want to see adult content?

A complete backup of pharmaxchange.info
Are you over 18 and want to see adult content?
A complete backup of marcellospizzeria.com
Are you over 18 and want to see adult content?
A complete backup of muhfathurrohman.wordpress.com
Are you over 18 and want to see adult content?
A complete backup of livetolovediabetes.com
Are you over 18 and want to see adult content?
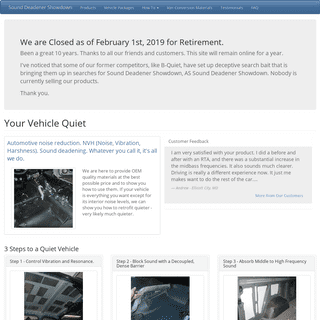
A complete backup of sounddeadenershowdown.com
Are you over 18 and want to see adult content?
Text
webinar on 21-APR
LIFE-SCIENCE PANORAMA Life-Science Panorama | A Journal for Industry Executives. 20 May/21. Reinventing the Value Network with the Factory of the Future. May 20, 2021 Latest, Research, Technology Axendia, Dassault Systemes, Delmia, Factory of the Future Administrator. The factory of CSA: A STREAMLINED APPROACH TO VALIDATION EBOOK CSA: A Streamlined Approach to Validation eBook. Computer System Validation (CSA) is a streamlined approach to validation that supports Manufacturing 4.0 in Life-Sciences. This new approach may result in a 30% to 50% reduction in validation time and cost accelerating innovation. In this Axendia eBook, Boston Scientific, FDA, Medtronic,SIEMENS
SAP RE-IMAGINES SUPPLY CHAIN COLLABORATION SAP Briefing Note. By Daniel R. Matlis. SAP’s Life Sciences leadership team recently briefed Axendia on its re-imagined Supply Chain Collaboration strategy.. Globalization and outsourcing have increased the volume of geographically dispersed partners, facilities, and suppliers in the Life-Science global supply chain. EBOOK: DIGITAL TRANSFORMATION AND MANUFACTURING In this eBook, we discuss: Digital transformation and modernizing manufacturing – why they are not the same. Overcoming regulatory inertia. The current state of digital transformation initiatives and the barriers. The need to modernize manufacturing and the barriers. Adopting emerging technologies to improve quality. INSIGHT BRIEF: IMPROVING PRODUCT QUALITY & PATIENT An effective Quality and Risk Management program enables Life Science manufacturers to proactively manage product quality and patient safety. Risk management is a top priority for regulators around the world with a special focus on combination products and medicaldevices.
PTC | LIFE-SCIENCE PANORAMA PTC 2019 LiveWorx Event Brief. By: Eric M. Luyer, Industry Research Analyst. The theme at PTC LiveWorx this year was: “The Augmented Workforce – Taking Human Capabilities to New Heights.” Approximately 13,000 people, attending in person as well as live streaming, took part in the annual industry conference held in Boston,MA, in early June.
MASTERCONTROL BRIEFING NOTE �2019 Axendia, Inc. 3 MASTERCONTROL BRIEFING NOTE IN BRIEF In interviews with our Industry clients, we have witnessed more and moreLife-Science
AXENDIA | TRUSTED ADVISORS FOR LIFE-SCIENCES EXECUTIVESRESOURCE LIBRARYBLOGCONTACTEBOOK-FACTORY OF THE FUTEBOOK-DIGITAL TRANSFORMA Axendia provides Executives with strategic advice on key initiatives. We understand the issues and priorities facing Industry Executives. OUR PEOPLE | AXENDIA Our People. Axendia Analysts offer a unique combination of hands-on exprience coupled with strategic vision. We deliver the insight necessary for our clients to informed decisions in these complex and highly regulated markets. Our Industry Analysts have Decades of WEBINAR | LIFE-SCIENCE PANORAMA COVID-19 forced Life-Science companies to reimagine the way they operate. Pfizer has adapted its Culture, Systems and Processes to drive increased visibility, control and collaboration that have achieved unprecedented results. Join Pfizer’s Hugo Felix and Axendia’s Daniel R. Matlis for a “Straight from the Source” livewebinar on 21-APR
LIFE-SCIENCE PANORAMA Life-Science Panorama | A Journal for Industry Executives. 20 May/21. Reinventing the Value Network with the Factory of the Future. May 20, 2021 Latest, Research, Technology Axendia, Dassault Systemes, Delmia, Factory of the Future Administrator. The factory of CSA: A STREAMLINED APPROACH TO VALIDATION EBOOK CSA: A Streamlined Approach to Validation eBook. Computer System Validation (CSA) is a streamlined approach to validation that supports Manufacturing 4.0 in Life-Sciences. This new approach may result in a 30% to 50% reduction in validation time and cost accelerating innovation. In this Axendia eBook, Boston Scientific, FDA, Medtronic,SIEMENS
SAP RE-IMAGINES SUPPLY CHAIN COLLABORATION SAP Briefing Note. By Daniel R. Matlis. SAP’s Life Sciences leadership team recently briefed Axendia on its re-imagined Supply Chain Collaboration strategy.. Globalization and outsourcing have increased the volume of geographically dispersed partners, facilities, and suppliers in the Life-Science global supply chain. EBOOK: DIGITAL TRANSFORMATION AND MANUFACTURING In this eBook, we discuss: Digital transformation and modernizing manufacturing – why they are not the same. Overcoming regulatory inertia. The current state of digital transformation initiatives and the barriers. The need to modernize manufacturing and the barriers. Adopting emerging technologies to improve quality. INSIGHT BRIEF: IMPROVING PRODUCT QUALITY & PATIENT An effective Quality and Risk Management program enables Life Science manufacturers to proactively manage product quality and patient safety. Risk management is a top priority for regulators around the world with a special focus on combination products and medicaldevices.
PTC | LIFE-SCIENCE PANORAMA PTC 2019 LiveWorx Event Brief. By: Eric M. Luyer, Industry Research Analyst. The theme at PTC LiveWorx this year was: “The Augmented Workforce – Taking Human Capabilities to New Heights.” Approximately 13,000 people, attending in person as well as live streaming, took part in the annual industry conference held in Boston,MA, in early June.
MASTERCONTROL BRIEFING NOTE �2019 Axendia, Inc. 3 MASTERCONTROL BRIEFING NOTE IN BRIEF In interviews with our Industry clients, we have witnessed more and moreLife-Science
WHAT WE DO | AXENDIA Axendia is the leading analyst and strategic advisory firm enabling Positive Disruption across the Life-Sciences Value Network. Since 2005, Industry Executive, Technology Innovators and regulators have trusted Axendia to provide analysis, advice and insights at the intersection of Business, Regulatory and Technology domains based ontrusted
OFFERINGS | AXENDIA
Axendia provides analysis and insights through a wide range of offerings to our clients. A changing regulatory landscape and technological innovation has made it impossible for companies to operate on silo’d point solutions. Trends we cover include: Let Axendia help with your organizations needs. Contact us at:info@axendia.com.
LOGIN | AXENDIA
Login. Axendia’s Research is complimentary for Life-Science & Healthcare Industry professionals and regulators. To request your complimentary access code, email research@axendia.com. Technology and service providers interested in accessing Axendia’s research should contact info@axendia.com for additional information. RESEARCH | AXENDIA | TRUSTED ADVISORS FOR LIFE-SCIENCES Actively managing products across the total product life cycle (including design, manufacturing and service management) drives improvements in product quality and accelerates new product introductions in a timely and cost-effective manner while supporting regulatory compliance. research@axendia.com for a complementary accesscode to view this
CONTACT | AXENDIA
Contact You are here: Home » Contact Global and Diverse Headquartered in Greater Philadelphia, Axendia has long term relationships with Life-Science companies and organizations in the region and around the world, including in North America, Europe, Asia, Latin America, andthe Middle East.
OUR CLIENTS
Axendia is committed to building, nurturing and growing long term relationships based on our core values of Respect, Integrity, and Passion. This commitment to our clients, partners and team members has fostered a spirit of cooperation, coordination and trust that is essential to ensure success. Axendia has advised organizationsincluding:
LIFE-SCIENCE PANORAMA Life-Science Panorama | A Journal for Industry Executives. 20 May/21. Reinventing the Value Network with the Factory of the Future. May 20, 2021 Latest, Research, Technology Axendia, Dassault Systemes, Delmia, Factory of the Future Administrator. The factory of ERGONOMICS | AXENDIA Axendia’s Ergonomics Practice is led by Mónica V. Matlis, CPE. Ms. Matlis has over 25 years of experience providing ergonomics leadership and sup port worldw ide and is a recognized leader within the field of ergonomics. Ms. Matlis provides strategic advice to companies with the development, implementation and/or improvement of Safety & Ergonomicsprograms worldwide.
SPEAKERS BUREAU
Speakers Bureau You are here: Home » Speakers Bureau Your Source of Trusted Life-Science Advice Analyst participation in your event can add considerable credibility by providing experienced thought-leaders who deliver presentations to customers, prospects, sellers, partners,and the press.
FACTORY OF THE FUTURE EBOOK Factory of the Future eBook. Life-Science Executives are focused on ensuring that their facilities can support next generation medical products. Innovative companies are addressing the need to solve tomorrow’s challenges of designing, sourcing, and manufacturing high quality, complex medical products in a stringent regulatoryenvironment.
AXENDIA | TRUSTED ADVISORS FOR LIFE-SCIENCES EXECUTIVESRESOURCE LIBRARYBLOGCONTACTEBOOK-FACTORY OF THE FUTEBOOK-DIGITAL TRANSFORMA Axendia provides Executives with strategic advice on key initiatives. We understand the issues and priorities facing Industry Executives. OUR PEOPLE | AXENDIA Our People. Axendia Analysts offer a unique combination of hands-on exprience coupled with strategic vision. We deliver the insight necessary for our clients to informed decisions in these complex and highly regulated markets. Our Industry Analysts have Decades of WEBINAR | LIFE-SCIENCE PANORAMA COVID-19 forced Life-Science companies to reimagine the way they operate. Pfizer has adapted its Culture, Systems and Processes to drive increased visibility, control and collaboration that have achieved unprecedented results. Join Pfizer’s Hugo Felix and Axendia’s Daniel R. Matlis for a “Straight from the Source” livewebinar on 21-APR
LIFE-SCIENCE PANORAMA Life-Science Panorama | A Journal for Industry Executives. 20 May/21. Reinventing the Value Network with the Factory of the Future. May 20, 2021 Latest, Research, Technology Axendia, Dassault Systemes, Delmia, Factory of the Future Administrator. The factory of CSA: A STREAMLINED APPROACH TO VALIDATION EBOOK CSA: A Streamlined Approach to Validation eBook. Computer System Validation (CSA) is a streamlined approach to validation that supports Manufacturing 4.0 in Life-Sciences. This new approach may result in a 30% to 50% reduction in validation time and cost accelerating innovation. In this Axendia eBook, Boston Scientific, FDA, Medtronic,SIEMENS
SAP RE-IMAGINES SUPPLY CHAIN COLLABORATION SAP Briefing Note. By Daniel R. Matlis. SAP’s Life Sciences leadership team recently briefed Axendia on its re-imagined Supply Chain Collaboration strategy.. Globalization and outsourcing have increased the volume of geographically dispersed partners, facilities, and suppliers in the Life-Science global supply chain. EBOOK: DIGITAL TRANSFORMATION AND MANUFACTURING In this eBook, we discuss: Digital transformation and modernizing manufacturing – why they are not the same. Overcoming regulatory inertia. The current state of digital transformation initiatives and the barriers. The need to modernize manufacturing and the barriers. Adopting emerging technologies to improve quality. INSIGHT BRIEF: IMPROVING PRODUCT QUALITY & PATIENT An effective Quality and Risk Management program enables Life Science manufacturers to proactively manage product quality and patient safety. Risk management is a top priority for regulators around the world with a special focus on combination products and medicaldevices.
PTC | LIFE-SCIENCE PANORAMA PTC 2019 LiveWorx Event Brief. By: Eric M. Luyer, Industry Research Analyst. The theme at PTC LiveWorx this year was: “The Augmented Workforce – Taking Human Capabilities to New Heights.” Approximately 13,000 people, attending in person as well as live streaming, took part in the annual industry conference held in Boston,MA, in early June.
MASTERCONTROL BRIEFING NOTE �2019 Axendia, Inc. 3 MASTERCONTROL BRIEFING NOTE IN BRIEF In interviews with our Industry clients, we have witnessed more and moreLife-Science
AXENDIA | TRUSTED ADVISORS FOR LIFE-SCIENCES EXECUTIVESRESOURCE LIBRARYBLOGCONTACTEBOOK-FACTORY OF THE FUTEBOOK-DIGITAL TRANSFORMA Axendia provides Executives with strategic advice on key initiatives. We understand the issues and priorities facing Industry Executives. OUR PEOPLE | AXENDIA Our People. Axendia Analysts offer a unique combination of hands-on exprience coupled with strategic vision. We deliver the insight necessary for our clients to informed decisions in these complex and highly regulated markets. Our Industry Analysts have Decades of WEBINAR | LIFE-SCIENCE PANORAMA COVID-19 forced Life-Science companies to reimagine the way they operate. Pfizer has adapted its Culture, Systems and Processes to drive increased visibility, control and collaboration that have achieved unprecedented results. Join Pfizer’s Hugo Felix and Axendia’s Daniel R. Matlis for a “Straight from the Source” livewebinar on 21-APR
LIFE-SCIENCE PANORAMA Life-Science Panorama | A Journal for Industry Executives. 20 May/21. Reinventing the Value Network with the Factory of the Future. May 20, 2021 Latest, Research, Technology Axendia, Dassault Systemes, Delmia, Factory of the Future Administrator. The factory of CSA: A STREAMLINED APPROACH TO VALIDATION EBOOK CSA: A Streamlined Approach to Validation eBook. Computer System Validation (CSA) is a streamlined approach to validation that supports Manufacturing 4.0 in Life-Sciences. This new approach may result in a 30% to 50% reduction in validation time and cost accelerating innovation. In this Axendia eBook, Boston Scientific, FDA, Medtronic,SIEMENS
SAP RE-IMAGINES SUPPLY CHAIN COLLABORATION SAP Briefing Note. By Daniel R. Matlis. SAP’s Life Sciences leadership team recently briefed Axendia on its re-imagined Supply Chain Collaboration strategy.. Globalization and outsourcing have increased the volume of geographically dispersed partners, facilities, and suppliers in the Life-Science global supply chain. EBOOK: DIGITAL TRANSFORMATION AND MANUFACTURING In this eBook, we discuss: Digital transformation and modernizing manufacturing – why they are not the same. Overcoming regulatory inertia. The current state of digital transformation initiatives and the barriers. The need to modernize manufacturing and the barriers. Adopting emerging technologies to improve quality. INSIGHT BRIEF: IMPROVING PRODUCT QUALITY & PATIENT An effective Quality and Risk Management program enables Life Science manufacturers to proactively manage product quality and patient safety. Risk management is a top priority for regulators around the world with a special focus on combination products and medicaldevices.
PTC | LIFE-SCIENCE PANORAMA PTC 2019 LiveWorx Event Brief. By: Eric M. Luyer, Industry Research Analyst. The theme at PTC LiveWorx this year was: “The Augmented Workforce – Taking Human Capabilities to New Heights.” Approximately 13,000 people, attending in person as well as live streaming, took part in the annual industry conference held in Boston,MA, in early June.
MASTERCONTROL BRIEFING NOTE �2019 Axendia, Inc. 3 MASTERCONTROL BRIEFING NOTE IN BRIEF In interviews with our Industry clients, we have witnessed more and moreLife-Science
WHAT WE DO | AXENDIA Axendia is the leading analyst and strategic advisory firm enabling Positive Disruption across the Life-Sciences Value Network. Since 2005, Industry Executive, Technology Innovators and regulators have trusted Axendia to provide analysis, advice and insights at the intersection of Business, Regulatory and Technology domains based ontrusted
OFFERINGS | AXENDIA
Axendia provides analysis and insights through a wide range of offerings to our clients. A changing regulatory landscape and technological innovation has made it impossible for companies to operate on silo’d point solutions. Trends we cover include: Let Axendia help with your organizations needs. Contact us at:info@axendia.com.
LOGIN | AXENDIA
Login. Axendia’s Research is complimentary for Life-Science & Healthcare Industry professionals and regulators. To request your complimentary access code, email research@axendia.com. Technology and service providers interested in accessing Axendia’s research should contact info@axendia.com for additional information. RESEARCH | AXENDIA | TRUSTED ADVISORS FOR LIFE-SCIENCES Actively managing products across the total product life cycle (including design, manufacturing and service management) drives improvements in product quality and accelerates new product introductions in a timely and cost-effective manner while supporting regulatory compliance. research@axendia.com for a complementary accesscode to view this
CONTACT | AXENDIA
Contact You are here: Home » Contact Global and Diverse Headquartered in Greater Philadelphia, Axendia has long term relationships with Life-Science companies and organizations in the region and around the world, including in North America, Europe, Asia, Latin America, andthe Middle East.
OUR CLIENTS
Axendia is committed to building, nurturing and growing long term relationships based on our core values of Respect, Integrity, and Passion. This commitment to our clients, partners and team members has fostered a spirit of cooperation, coordination and trust that is essential to ensure success. Axendia has advised organizationsincluding:
LIFE-SCIENCE PANORAMA Life-Science Panorama | A Journal for Industry Executives. 20 May/21. Reinventing the Value Network with the Factory of the Future. May 20, 2021 Latest, Research, Technology Axendia, Dassault Systemes, Delmia, Factory of the Future Administrator. The factory of ERGONOMICS | AXENDIA Axendia’s Ergonomics Practice is led by Mónica V. Matlis, CPE. Ms. Matlis has over 25 years of experience providing ergonomics leadership and sup port worldw ide and is a recognized leader within the field of ergonomics. Ms. Matlis provides strategic advice to companies with the development, implementation and/or improvement of Safety & Ergonomicsprograms worldwide.
SPEAKERS BUREAU
Speakers Bureau You are here: Home » Speakers Bureau Your Source of Trusted Life-Science Advice Analyst participation in your event can add considerable credibility by providing experienced thought-leaders who deliver presentations to customers, prospects, sellers, partners,and the press.
FACTORY OF THE FUTURE EBOOK Factory of the Future eBook. Life-Science Executives are focused on ensuring that their facilities can support next generation medical products. Innovative companies are addressing the need to solve tomorrow’s challenges of designing, sourcing, and manufacturing high quality, complex medical products in a stringent regulatoryenvironment.
Log in | Create an account*
*
*
*
Search for:
* Resource Library
* eBook-AI Drives Predictive * eBook-Building a Holistic… * eBook-CSA: A Stream… * eBook-Goodbye CSV…* Services
* Offerings
* Speakers Bureau
* Ergonomics
* About
* What We Do
* Our People
* Our Clients
* Our Privacy Policy* Testimonials
* Blog
* Contact
Main Menu...Resource Library--- eBook-AI Drives Predictive--- eBook-Building a Holistic…--- eBook-CSA: A Stream…--- eBook-Goodbye CSV…Services--- Offerings--- Speakers Bureau--- ErgonomicsAbout--- What We Do--- Our People--- Our Clients--- Our Privacy Policy--- TestimonialsBlogContactLearn More
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0