Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://3dprintingindustry.com
Are you over 18 and want to see adult content?
A complete backup of https://compstak.com
Are you over 18 and want to see adult content?
A complete backup of https://catadoptionteam.org
Are you over 18 and want to see adult content?

A complete backup of https://crudi-solidarite.com
Are you over 18 and want to see adult content?

A complete backup of https://clip-share.com
Are you over 18 and want to see adult content?

A complete backup of https://mmogo.com
Are you over 18 and want to see adult content?

A complete backup of https://reklamstore.com
Are you over 18 and want to see adult content?
A complete backup of https://archaeologiemuseum.it
Are you over 18 and want to see adult content?
A complete backup of https://blackmountaininstitute.org
Are you over 18 and want to see adult content?
A complete backup of https://mojtrg.rs
Are you over 18 and want to see adult content?

A complete backup of https://cpl.co.uk
Are you over 18 and want to see adult content?
A complete backup of https://thebeachwaterpark.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of hairymusclebearloverfan.tumblr.com
Are you over 18 and want to see adult content?
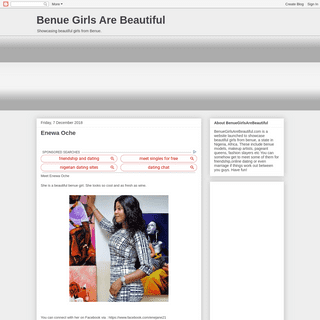
A complete backup of benuegirlsarebeautiful.blogspot.com
Are you over 18 and want to see adult content?

A complete backup of ganzjahresreifen-testsieger.de
Are you over 18 and want to see adult content?

A complete backup of hotcelebforum.com
Are you over 18 and want to see adult content?

A complete backup of ot-montsaintmichel.com
Are you over 18 and want to see adult content?
Text
ABOUT US - AGENUS
Agenus Inc. 3 Forbes Road. Lexington, MA 02421-7305. T: 781.674.4400.F: 781.674.4200
AGEN1777ANTI-TIGIT BISPECIFIC AGEN1777 is a first-in-class TIGIT bispecific which targets a major inhibitory receptor expressed on T and NK cells to improve anti-tumoractivity.
AGEN1181MULTIPURPOSE, 2ND GEN CTLA-4 AGEN1181 is a multipurpose, second-generation “Fc-engineered” anti-CTLA-4 antibody with enhanced tumor fighting abilities, designed to generate responses in a larger number of patients and improveimmunogenicity.
OPTIMALLY TARGETING TIGIT ptimally Targeting TIGIT Forward-ooking Statements: This newsletter contains forward-looking statements that are made pursuant to the safe harbor provisions of the federal securities laws, including statements regarding the anticipated therapeutic potential of anti-TIGIT antibodies, as well as clinical development and regulatory plans and timelines for AGEN1777 and its potential as a best-in BALSTILIMABANTI-PD-1 Enrolling in second-line cervical cancer NCT03894215. Randomized, blinded, non-comparative, two-arm Phase 2 clinical trial to assess the efficacy and safety of AGEN2034 administered with placebo (Treatment Arm 1 – monotherapy) or with AGEN1884 (Treatment Arm 2 – combination therapy) for treatment of patients with advanced cervical cancer who relapsed or progressed after receiving firstAGEN2373ANTI-CD137
American Society of Clinical Oncology (ASCO) June 3-7, 2021. Initial Findings of the First-in-human Phase I Study of AGEN2373, a Conditionally Active CD137 Agonist Antibody, in AGENUS TO PRESENT NEW CLINICAL DATA ON AGEN1181 AT AACR Clinical benefit in previously unresponsive tumors, including CRC, ovarian, and endometrial Confirmed responses in cold tumors as characterized by MSS No neuroendocrine toxicities or hypophysitis Registrational trials planned to commence in 2021 LEXINGTON, Mass., March 10, 2021 (GLOBE NEWSWIRE) -- AGENUS FC-ENHANCED TIGITS TO MAXIMIZE BENEFIT Vo 3 ss 2 o 2 22 After several years of slow progress, the field of immuno-oncology (I-O) is starting to register another wave ofadvances.
AGENUS SUBMITS BALSTILIMAB BIOLOGICS LICENSE APPLICATION Submission has been made for review under the accelerated approval pathway LEXINGTON, Mass., April 19, 2021 (GLOBE NEWSWIRE) -- Agenus Inc. (NASDAQ: AGEN), an immuno-oncology company with an extensive pipeline of agents which includes checkpoint antibodies, cell therapies, adjuvants, and vaccines AGENUS - THE RIGHT COMBINATIONABOUT USPIPELINECAPABILITIESPUBLICATIONSINVESTORSCAREERS Agenus discovers, manufactures and develops immuno-oncology products. The company’s I-O portfolio is comprised of checkpoint antibodies, cell therapies, vaccines, and adjuvants, representing a combination of synergistic agents to deliver curative patient outcomes.ABOUT US - AGENUS
Agenus Inc. 3 Forbes Road. Lexington, MA 02421-7305. T: 781.674.4400.F: 781.674.4200
AGEN1777ANTI-TIGIT BISPECIFIC AGEN1777 is a first-in-class TIGIT bispecific which targets a major inhibitory receptor expressed on T and NK cells to improve anti-tumoractivity.
AGEN1181MULTIPURPOSE, 2ND GEN CTLA-4 AGEN1181 is a multipurpose, second-generation “Fc-engineered” anti-CTLA-4 antibody with enhanced tumor fighting abilities, designed to generate responses in a larger number of patients and improveimmunogenicity.
OPTIMALLY TARGETING TIGIT ptimally Targeting TIGIT Forward-ooking Statements: This newsletter contains forward-looking statements that are made pursuant to the safe harbor provisions of the federal securities laws, including statements regarding the anticipated therapeutic potential of anti-TIGIT antibodies, as well as clinical development and regulatory plans and timelines for AGEN1777 and its potential as a best-in BALSTILIMABANTI-PD-1 Enrolling in second-line cervical cancer NCT03894215. Randomized, blinded, non-comparative, two-arm Phase 2 clinical trial to assess the efficacy and safety of AGEN2034 administered with placebo (Treatment Arm 1 – monotherapy) or with AGEN1884 (Treatment Arm 2 – combination therapy) for treatment of patients with advanced cervical cancer who relapsed or progressed after receiving firstAGEN2373ANTI-CD137
American Society of Clinical Oncology (ASCO) June 3-7, 2021. Initial Findings of the First-in-human Phase I Study of AGEN2373, a Conditionally Active CD137 Agonist Antibody, in AGENUS TO PRESENT NEW CLINICAL DATA ON AGEN1181 AT AACR Clinical benefit in previously unresponsive tumors, including CRC, ovarian, and endometrial Confirmed responses in cold tumors as characterized by MSS No neuroendocrine toxicities or hypophysitis Registrational trials planned to commence in 2021 LEXINGTON, Mass., March 10, 2021 (GLOBE NEWSWIRE) -- AGENUS FC-ENHANCED TIGITS TO MAXIMIZE BENEFIT Vo 3 ss 2 o 2 22 After several years of slow progress, the field of immuno-oncology (I-O) is starting to register another wave ofadvances.
AGENUS SUBMITS BALSTILIMAB BIOLOGICS LICENSE APPLICATION Submission has been made for review under the accelerated approval pathway LEXINGTON, Mass., April 19, 2021 (GLOBE NEWSWIRE) -- Agenus Inc. (NASDAQ: AGEN), an immuno-oncology company with an extensive pipeline of agents which includes checkpoint antibodies, cell therapies, adjuvants, and vaccinesLEADERSHIP - AGENUS
Dr. Marc van Dijk serves as Chief Technology Officer and leads our discovery and innovation center in Cambridge, UK. Marc is a molecular biologist and technologist whose work helping to design drug discovery platforms at Agenus has led to the advancement of more than 15SITC 2020 - AGENUS
Agenus Inc. 3 Forbes Road. Lexington, MA 02421-7305. T: 781.674.4400.F: 781.674.4200
AGENUSNEWS VOL.4 • ISS.11 • JUNE 2, 2021 Anti-PD-1 was one of the first validated immune checkpointinhibitors that paved the way for immunotherapy. PD-1 hasbeen shown to be a critical target in combating cancer, notonly alone, but also in combination with various other agents.The importance of this foundational pathway, and its positionas an essential combination partner, underscore the criticalityof Agenus having our own PD-1.CAREERS - AGENUS
We are RELENTLESS in our pursuit to deliver critical medicines to patients who are waiting for a solution. We are TENACIOUS in disrupting the paradigm of big pharma by delivering 15 INDs at recordspeed.
INVESTORS - AGENUS INC. Company Information. Agenus is an immuno-oncology company focused on the discovery and development of revolutionary new treatments that engage the body’s immune system to benefit patients suffering fromcancer.
VIRTUAL ANNUAL SHAREHOLDERS MEETING 2021 The Investor Relations website contains information about Agenus Inc.'s business for stockholders, potential investors, and financialanalysts.
AGENT-797ALLOGENEIC INKT CELL THERAPY At AgenTus, Agenus’s cell therapy subsidiary, we are advancing a pipeline of unique allogeneic cell therapies not only designed to address the limitations of autologous cell therapies but also many of the potential challenges of allogeneic cell therapy approaches currently being pursued. AGENUS PRESENTS ADDITIONAL CLINICAL RESPONSES AND NOVEL AGEN1181 : New clinical responses and intratumoral Treg depletion AGEN 1777 ( TIGIT ) is Fc enhanced for optimized immune performance & broader benefit AGEN2373 (anti-CD137) : Clinical benefit with no liver toxicity Allogeneic iNKTs : Potent tumor AGENUS’ FIRST BIOTECH ELECTRONIC SECURITY TOKEN (BEST) Last week we announced our plans to issue the first ever Digital Security backed by future sales of a biopharma product. The Biotech Electronic Security Token INCYTE AND AGENUS AMEND COLLABORATION AGREEMENT WILMINGTON, DE and LEXINGTON, MA, February 14, 2017 (BUSINESS WIRE) – Incyte Corporation (Nasdaq:INCY) and Agenus Inc. (Nasdaq:AGEN) announced today that the companies have amended the License, Development and Commercialization Agreement that was originally entered into January 9, 2015. The amended agreement converts the ongoing GITR and OX40 antibody programs AGENUS - THE RIGHT COMBINATIONABOUT USPIPELINECAPABILITIESPUBLICATIONSINVESTORSCAREERS Agenus discovers, manufactures and develops immuno-oncology products. The company’s I-O portfolio is comprised of checkpoint antibodies, cell therapies, vaccines, and adjuvants, representing a combination of synergistic agents to deliver curative patient outcomes.ABOUT US - AGENUS
About Us. Agenus discovers, manufactures and develops immuno-oncology products. The company’s I-O portfolio is comprised of checkpoint antibodies, cell therapies, vaccines, and adjuvants, representing a combination of synergistic agents to deliver curative patientoutcomes.
LEADERSHIP - AGENUS
Chairman and Chief Executive Officer. Dr. Garo Armen serves as Chairman and Chief Executive Officer of Agenus Inc., which he co-founded in 1994. Under his leadership, Agenus has been advancing breakthrough scientific technologies and immunotherapeutic products. In addition, Garo oversaw the successful restructuring of thebiopharmaceutical
CAREERS - AGENUS
Careers - Agenus. We are RELENTLESS in our pursuit to deliver critical medicines to patients who are waiting for a solution. We are TENACIOUS in disrupting the paradigm of big pharma by delivering 15 INDs at record speed. We are giving new meaning to the words SPEED and INNOVATION by challenging the norm. We build quickly on our successand WE
INVESTORS | AGENUS INC. Agenus is an immuno-oncology company focused on the discovery and development of revolutionary new treatments that engage the body’s immune system to benefit patients suffering from cancer. By combining multiple powerful platforms, Agenus has established a highly integrated approach for the discovery, development and manufacture ofmonoclonal
OPTIMALLY TARGETING TIGIT ptimally Targeting TIGIT Forward-ooking Statements: This newsletter contains forward-looking statements that are made pursuant to the safe harbor provisions of the federal securities laws, including statements regarding the anticipated therapeutic potential of anti-TIGIT antibodies, as well as clinical development and regulatory plans and timelines for AGEN1777 and its potential as a best-in AGENUS - THE RIGHT COMBINATIONABOUT USPIPELINECAPABILITIESPUBLICATIONSINVESTORSCAREERS Agenus discovers, manufactures and develops immuno-oncology products. The company’s I-O portfolio is comprised of checkpoint antibodies, cell therapies, vaccines, and adjuvants, representing a combination of synergistic agents to deliver curative patient outcomes.ABOUT US - AGENUS
About Us. Agenus discovers, manufactures and develops immuno-oncology products. The company’s I-O portfolio is comprised of checkpoint antibodies, cell therapies, vaccines, and adjuvants, representing a combination of synergistic agents to deliver curative patientoutcomes.
LEADERSHIP - AGENUS
Chairman and Chief Executive Officer. Dr. Garo Armen serves as Chairman and Chief Executive Officer of Agenus Inc., which he co-founded in 1994. Under his leadership, Agenus has been advancing breakthrough scientific technologies and immunotherapeutic products. In addition, Garo oversaw the successful restructuring of thebiopharmaceutical
CAREERS - AGENUS
Careers - Agenus. We are RELENTLESS in our pursuit to deliver critical medicines to patients who are waiting for a solution. We are TENACIOUS in disrupting the paradigm of big pharma by delivering 15 INDs at record speed. We are giving new meaning to the words SPEED and INNOVATION by challenging the norm. We build quickly on our successand WE
INVESTORS | AGENUS INC. Agenus is an immuno-oncology company focused on the discovery and development of revolutionary new treatments that engage the body’s immune system to benefit patients suffering from cancer. By combining multiple powerful platforms, Agenus has established a highly integrated approach for the discovery, development and manufacture ofmonoclonal
OPTIMALLY TARGETING TIGIT ptimally Targeting TIGIT Forward-ooking Statements: This newsletter contains forward-looking statements that are made pursuant to the safe harbor provisions of the federal securities laws, including statements regarding the anticipated therapeutic potential of anti-TIGIT antibodies, as well as clinical development and regulatory plans and timelines for AGEN1777 and its potential as a best-inCONTACT US - AGENUS
Agenus Inc. 3 Forbes Road. Lexington, MA 02421-7305. T: 781.674.4400.F: 781.674.4200
AGENT-797ALLOGENEIC INKT CELL THERAPY Further, if approved they can be offered at a fraction of the cost of existing cell therapies. The FDA has cleared the IND for AGENT-797 for use in patients with COVID-19 and cancer. AgenTus is advancing iNKT cell therapy to the clinic for these diseases. Together, Agenus and AgenTus are uniquely positioned to develop optimal combination AGENUS’ PRESENTATIONS AT ASCO 2021 DEMONSTRATE LEXINGTON, Mass., June 04, 2021 (GLOBE NEWSWIRE) -- Agenus (NASDAQ: AGEN), an immuno-oncology company with an extensive pipeline of checkpoint antibodies, cell therapies, adjuvants, and vaccines designed to activate immune response to cancers and infections, today presented data demonstrating the differentiation of balstilimab as an anti-PD-1 antibody as well as data from a Phase AGENUS ANNOUNCES POSITIVE PRELIMINARY RESULTS OF INKT CELL Agenus Announces Positive Preliminary Results of iNKT Cell Therapy Trial in COVID-19. LEXINGTON, Mass., Feb. 04, 2021 (GLOBE NEWSWIRE) -- Agenus Inc. (NASDAQ: AGEN), an immuno-oncology company with an extensive pipeline of checkpoint antibodies, cell therapies, adjuvants, and vaccines designed to activate immune response tocancers and
INITIAL FINDINGS OF THE FIRST-IN-HUMAN PHASE I STUDY OF Initial Findings of the First-in-human Phase I Study of AGEN2373, a Conditionally Active CD137 Agonist Antibody, in Patients (pts) With Advanced Solid TumorsAGEN2373ANTI-CD137
anti-CD137. CD137 (4-1BB) is a positive regulator of the immune system that is highly upregulated on activated T cells (adaptive immune cells) and NK cells (innate immune cells). The potential to dually target innate and adaptive immunity makes CD137 a highly attractive target for cancer immunotherapy. AGEN2373 is a fully human monoclonalBOARD OF DIRECTORS
Garo H. Armen, PhD. Chairman and CEO. Dr. Garo Armen serves as Chairman and Chief Executive Officer of Agenus Inc., which he co-founded in 1994. Under his leadership, Agenus has been advancing breakthrough scientific technologies and immunotherapeutic products. In addition, Garo oversaw the successful restructuring of thebiopharmaceutical
AGENUS ANNOUNCES THE APPOINTMENT OF DR. JENNIFER BUELL TO LEXINGTON, Mass., Jan. 9, 2020 /PRNewswire/ -- Agenus Inc. (NASDAQ: AGEN), an immuno-oncology company with an extensive pipeline of agents that activate immune response to cancers, is pleased to announce the appointment of Dr. Jennifer Buell to President and COO. "Jen's tenure with Agenus, her exceptional leadership and cultural attributes, and her deep understanding of our DIFFERENTIATED ACTIVITY PROFILE FOR THE PD-1 INHIBITOR Variable response rates have been observed across clinical trials for PD-1 antibodies in R/M CC. PD-L1 positivity is an incomplete biomarker in this setting, with both balstilimab and other PD-1 antibodies demonstrating the potential to elicit ASCO PRESENTATION OF AGENUS' AGEN1181 BY DR. STEVEN O'DAY Conference call scheduled on June 2, 2020. B.Riley FBR Senior Analyst Mayank Mamtani, will host a conference call for investors on Tuesday, June 2, 2020 with Dr. Steven O'Day and Dr. Charles Drake, Co-Director of the Cancer Immunotherapy Program and Co-Leader of the Tumor Biology & Microenvironment Program at Columbia University, and Jennifer Buell, PhD and President and COO of* About Us
* Overview
* Leadership
* Board of Directors * Scientific Advisory Board* Pipeline
* Cancer
* COVID-19
* Clinical Trials
* Zalifrelimab
anti-CTLA-4
* Balstilimab
anti-PD-1
* AGEN1181
Multipurpose, 2nd Gen CTLA-4* AGEN1327
anti-TIGIT
* AGEN1777
anti-TIGIT bispecific* AGEN2373
anti-CD137
* AGEN1223
* AGENT-797
Allogeneic iNKT cell Therapy* Capabilities
* Discovery and Development * GMP Manufacturing Facility * Clinical Development* Publications
* Publications and Presentations* Newsletters
* Investors
* Company Information* Stock Information
* Press releases
* Other Media Coverage* SEC Filings
* Analyst Coverage
* Events and Presentations * Corporate Governance* Investor Contacts
* Careers
* Contact Us
*
* About Us
* Overview
* Leadership
* Board of Directors * Scientific Advisory Board* Pipeline
* Cancer
* COVID-19
* Clinical Trials
* Zalifrelimab
anti-CTLA-4
* Balstilimab
anti-PD-1
* AGEN1181
Multipurpose, 2nd Gen CTLA-4* AGEN1327
anti-TIGIT
* AGEN1777
anti-TIGIT bispecific* AGEN2373
anti-CD137
* AGEN1223
* AGENT-797
Allogeneic iNKT cell Therapy* Capabilities
* Discovery and Development * GMP Manufacturing Facility * Clinical Development* Publications
* Publications and Presentations* Newsletters
* Investors
* Company Information* Stock Information
* Press releases
* Other Media Coverage* SEC Filings
* Analyst Coverage
* Events and Presentations * Corporate Governance* Investor Contacts
* Careers
* Contact Us
*
INNOVATION AND SPEED FOR CANCER CURESVIEW PIPELINE __
Find clinical trials __Join our team __
Read press releases __PIPELINE HIGHLIGHTS
Click an individual product to view a detailed description. Agenus fully-owned programs Partnered option programsMECHANISM/TARGET
PRODUCT
PARTNER
PRECLINICAL
PH1
PH2
PH3
FILED
APPROVED
PHASE
Immune modulating AntibodiesCTLA-4
Zalifrelimab (AGEN1884)__
Ph2
PD-1
Balstilimab (AGEN2034)__
Ph2
Multipurpose, second-generationCTLA-4
AGEN1181
__
Ph1
TIGIT
AGEN1327
__
Pre
TIGIT (bispecific)
AGEN1777
__
Pre
CD137
AGEN2373
Partner:
__
Ph1
Regulatory T cell depletion (bispecific)AGEN1223
Partner:
__
Ph1
Zalifrelimab (AGEN1884)CTLA-4
Agenus fully-owned
ph2
Ph2
Product Description
CTLA-4 (cytotoxic T-lymphocyte antigen-4), is a negative regulator of immune activation that is considered a foundational target within the immuno-oncology market. In 2018, the Nobel Prize in Medicine was awarded for the establishment of CTLA-4 as a cancer immunotherapy target. Agenus has applied these scientific findings to create a therapy designed to treat cancer, AGEN1884. AGEN1884 is a fully humanized monoclonal antibody that activates the immune system to destroy cancer cells by blocking the interaction between CTLA-4 and its binding partners CD80 and CD86. AGEN1884 is clinically active in patients with advanced or refractory cancer. In fact, we have seen a complete and durable response in a patient with refractory angiosarcoma. This patient was profiled in thenews.
Agenus’ CTLA-4 is in clinical trials as a monotherapy and a combination with our PD-1, AGEN2034 in an ongoing Phase 1/2, open-label, multi-arm trial to investigate the safety, tolerability, pharmacokinetics, biological and clinical activity of this regimen in patients with metastatic or locally advanced solid tumors including refractory cervical cancer. As of April 2020, our anti-CTLA-4 antibody in combination with our anti-PD-1 antibody has achieved 14 positive clinical responses in 55 patients with second-line cervical cancer (~26% ORR with ~12-month follow-up). _Agenus controls worldwide rights to AGEN1884, except for certain South American rights, which are controlled by Recepta Biopharma._ Balstilimab (AGEN2034)PD-1
Agenus fully-owned
ph2
Ph2
Product Description
PD-1 (programmed cell death protein 1) is a negative regulator of immune activation that is considered a foundational target within the immuno-oncology market. In 2018, the Nobel Prize in Medicine was awarded for the establishment of PD-1 as a cancer immunotherapy target. Agenus has applied these scientific principles to create a therapy designed to treat cancer, AGEN2034. AGEN2034 is a novel, fully human monoclonal immunoglobulin G4 (IgG4) designed to block PD-1 from interacting with its ligands PD-L1 andPD-L2.
AGEN2034 is clinically active in patients with advanced or refractorycancer.
Agenus’ PD-1 is in clinical trials as a monotherapy and a combination with our CTLA-4, AGEN1884 in an ongoing Phase 1/2, open-label, multi-arm trial to investigate the safety, tolerability, pharmacokinetics, biological and clinical activity of this regimen in patients with metastatic or locally advanced solid tumors including refractory cervical cancer. As of April 2020, our anti-CTLA-4 antibody in combination with our anti-PD-1 antibody has achieved 14 positive clinical responses in 55 patients with second-line cervical cancer (~26% ORR with ~12-month follow-up). _Agenus controls worldwide rights to AGEN1884, except for certain South American rights, which are controlled by Recepta Biopharma._AGEN1181
Multipurpose, second-generationCTLA-4
Agenus fully-owned
ph1-2
Ph1
Product Description
AGEN1181 is a multipurpose, second-generation “Fc-engineered” anti-CTLA-4 antibody with enhanced tumor fighting abilities, designed to generate responses in a larger number of patients and improveimmunogenicity.
AGEN1181, has achieved complete and partial responses, alone and in combination with AGEN’s anti-PD-1 (balstilimab / AGEN2034) in early clinical trials (as of April 2020). _DISCOVERY OF AGEN1181 AND ITS MECHANISM OF ACTION_Agenus
scientists discovered a new mechanism of action in which a specialized domain of the antibody engages with receptors on other immune cells to significantly bolster antitumor immunity. This discovery was publishedin Cancer cell
To develop AGEN1181, Agenus leveraged the above discovery to modify a part of the antibody known as the Fc region, which interacts with other immune cells to enhance antitumor immunity. In pre-clinical studies, this “Fc-engineered” antibody significantly improved the cross-talk between antigen presenting cells (APCs) and T cells. In cancer immunotherapy, such an interaction is necessary to mount a potent immune response against cancer.AGEN1327
TIGIT
Agenus fully-owned
preclinical
Pre
Product Description
TIGIT is a negative regulator of innate and adaptive immune responses. AGEN1327 is a fully human monoclonal antibody that boosts the immune response to cancer cells by blocking the interaction between TIGIT and its binding partner CD155. Moreover, AGEN1327 is one of our next-generation antibodies which has been “Fc engineered” to maximize antitumor activity (see AGEN1181). Therefore, we expect that AGEN1327 will bring clinical benefit above and beyond that of conventional anti-TIGIT antibodies. TIGIT signaling has also been identified as a key resistance mechanism to anti-PD-1 therapy, suggesting that AGEN1327 can address therapeutic resistance toanti-PD-1 therapy.
IND filing is expected in 1H 2021.AGEN1777
TIGIT (bispecific)
Agenus fully-owned
preclinical
Pre
Product Description
AGEN1777 with expected IND filing by year end – a first-in-class TIGIT bispecific which targets a major inhibitory receptor expressed on T and NK cells to improve anti-tumor activity. TIGIT is a negative regulator of innate and adaptive immune responses. AGEN1777 is a first-in-class TIGIT bispecific which targets a major inhibitory receptor expressed on T and NK cells to improve anti-tumoractivity.
IND filing is expected by end of 2020.AGEN1531
Tumor-associated macrophageAgenus fully-owned
preclinical
Pre
Cytokine (bispecific)Agenus fully-owned
preclinical
Pre
Stromal Conditioning (bispecific)Agenus fully-owned
preclinical
Pre
TME conditioning (bispecific)Agenus fully-owned
preclinical
Pre
AGEN2373
CD137
Partnered option programsph1
Ph1
Product Description
CD137 (4-1BB) is a positive regulator of the immune system that is highly upregulated on activated T cells (adaptive immune cells) and NK cells (innate immune cells). The potential to dually target innate and adaptive immunity makes CD137 a highly attractive target for cancer immunotherapy. AGEN2373 is a fully human monoclonal antibody that boosts the immune response to cancer cells by enhancing CD137 co-stimulatory signaling in activated immune cells. Importantly, the unique binding properties of AGEN2373 are expected to limit its activity outside of the tumor site and mitigate toxicities that may be associated with systemic activation of CD137 in humans. AGEN2373 is advancing in a clinical trial against solid tumors.AGEN1223
Regulatory T cell depletion (bispecific) Partnered option programsph1
Ph1
Product Description
Bispecific antibodies are engineered to simultaneously bind two different antigens. They can be used to tailor the binding dynamics and effector functions of antibodies to specific cell types. We are developing two bispecific antibodies based on highly innovative strategies to combat tumor escape mechanisms. AGEN1223 is designed to selectively deplete immunosuppressive T regulatory cells from the tumor microenvironment. This agent takes advantage of co-expression of the target antigens specifically on tumor-infiltrating T regulatory cells. In contrast to other strategies targeting T regulatory cells, AGEN1223 is expected to spare important immune regulatory peripheral Tregs and cancer-fighting effector Tcells.
AGEN1223 is advancing in a clinical trial against solid tumors.GS-1423
TME conditioning anti-CD73/TGFβ TRAP bifunctional fusion proteinPartnered
ph1
Ph1
Product Description
Bispecific antibodies are engineered to simultaneously bind two different antigens. They can be used to tailor the binding dynamics and effector functions of antibodies to specific cell types. We are developing two bispecific antibodies based on highly innovative strategies to combat tumor escape mechanisms. AGEN1423 combats two prominent resistance pathways for cancer immunotherapy. These pathways are present across many tumor types and are associated with poor responses to checkpoint blockade and other treatments. Notably, this agent has the potential to enhance the antitumor activity of myeloid cells, NK cells, T cells, and cancer associated fibroblasts. We expect our bi-specific program to move intothe clinic in 2019.
INCAGN1876
GITR
Partnered
ph1
Ph1
Product Description
GITR is an immune checkpoint agonist, one of a class of receptors that amplify the immune system’s response to cancer. GITR (glucocorticoid-induced TNFR-related protein) is a receptor expressed on select populations of T cells. Activation of GITR leads to a more powerful anti-tumor inflammatory response, increased production of inflammatory signaling molecules and increased resistance toimmunosuppression.
Our GITR agonist INCAGN1876 is part of our global alliance with Incyte. The program is funded by Incyte with Agenus eligible for potential milestones and 15% royalties, subject to reduction for certain third party obligations. GITR agonists selectively amplify an antigen-specific immune response in the context of cancer. We believe that antibodies targeting GITR will act synergistically with approved cancer therapies as well as with other checkpoint antibodies and cancer vaccines being developed. INCAGN1876 is currently in Phase 1 /2 clinical trials. (www.clinicaltrials.gov)
INCAGN1949
OX40
Partnered
ph1
Ph1
Product Description
OX40 (also known as CD134 and TNFRSF4), a member of the TNFR super-family, is an immune-response-enhancing receptor found on activated T cells. OX40 promotes proliferation of these activated T cells and prevents the immunosuppressive activity of inhibitory T cells. We believe that antibodies that activate OX40 may help increase immune system activity through both of these mechanisms. Furthermore, OX40 antibodies have the potential to work alone or in combination with other therapeutics. Our INCAGN1949 antibody targeting OX40 is part of our global alliance with Incyte. The program is funded by Incyte with Agenus eligible for potential milestones and 15% royalties, subject to reduction for certain third party obligations. INCAGN1949 is currently in Phase 1 / 2 clinical trials (www.clinicaltrials.gov).
INCAGN2390
TIM-3
Partnered
ph1
Ph1
Product Description
TIM-3 is a checkpoint receptor found on certain immune cells. TIM-3 stands for T-cell immunoglobulin and mucin domain-3. To prevent hyperactivation, natural ligands binding to TIM-3 reduce the activity of these immune cells. In some types of cancer, T cells express elevated levels of TIM-3, which results in excessive immune suppression. Blocking TIM-3 could stimulate immune responses and promote immune-mediated clearing of cancer cells. TIM-3 antibodies may have a role as a monotherapy and hold great potential in combination therapy, since they may help overcome resistance that patients may develop against other therapeutics. Our TIM-3 checkpoint inhibitor is part of our global alliance with Incyte. The program is funded by Incyte with Agenus eligible for potential milestones and royalties ranging from 6-12%, subject to reduction for certain third partyobligations.
INCAGN2385
LAG-3
Partnered
ph1
Ph1
Product Description
LAG-3 is a checkpoint protein expressed on the surface of certain cells of the immune system. LAG-3 (lymphocyte-activation gene 3) modulates signaling between immune cells and their targets. When LAG-3 is activated, the immune response is suppressed. Antibodies that block LAG-3 can block this inhibitory signal, thereby boosting the immune system’s response against cancer cells. LAG-3 acts synergistically with other checkpoint modulators. This suggests antibodies against LAG-3 may be valuable as combination therapies . This is why we believe our LAG-3 checkpoint inhibitor may have potential in both combination and monotherapy settings. Our LAG-3 checkpoint inhibitor is part of our global alliance with Incyte. The program is funded by Incyte with Agenus eligible for potential milestones and royalties ranging from 6-12%, subject to reduction for certain third partyobligations.
Undisclosed
Partnered
preclinical
Pre
MK-4830
ILT4
Partnered
ph1
Ph1
AGENT-797
(Unmodified iNKT Cell Therapy)Cancers
Agenus fully-owned
preclinical
Pre
Product Description
At AgenTus, we are advancing a pipeline of unique allogeneic cell therapies not only designed to address the limitations of autologous cell therapies but also many of the potential challenges of allogeneic cell therapy approaches currently being pursued. Our allogeneic iNKTs do not require any genetic manipulation and have been manufactured under GMP conditions and in significantly expanded quantities. Further, if approved they can be offered at a fraction of the cost of existing cell therapies. Our iNKTs can also be used in combination with our own immune checkpoint antibodies as well as an iNKT cell activating lipid ligand; both designed to improve patientresponse.
Our world-renowned scientific leadership team includes Dr. Mark Exley with over 70 peer reviewed publications specifically in this field. The advantages of our iNKT platform are summarized in the table below. The FDA has cleared the IND for AGENT-797.Learn more >
AGENT-797
(Unmodified iNKT Cell Therapy)COVID-19
Agenus fully-owned
preclinical
Pre
AutoSynVaxTM
Cancers
Agenus fully-owned
ph1
Ph1
Product Description
The immune system has the capacity to “see” cancer antigens or mutated molecules on cancer cells as foreign or abnormal and unleash an attack that destroys these cancerous cells. However, the number of mutations varies from one patient’s tumor to another. Current research suggests that our natural immune defense mechanisms may be insufficient to eliminate cancers in cases where mutations are too few in number. Even when cancer cells display many mutations, they can evade effective recognition by the immune system through a variety of immunosuppressive mechanisms. Agenus’ AutoSynVax cancer vaccine is being designed to boost immune recognition of cancer antigens to a higher level than that which is achieved naturally. The vaccine therefore may help the immune system better recognize cancer antigens even if there are only a small number of them on a given patient’s tumor. AutoSynVax is expected to work synergistically with other forms of immunotherapy including checkpoint inhibitors and other checkpoint modulators that counteract immune evasion mechanisms. AutoSynVax is an individualized vaccine made by combining synthetic versions of each patient’s own cancer antigens with heat shock protein 70 (Hsc70). Hsc70 is known to transport protein fragments called epitopes and to play a part in displaying them to T cells, immune cells that, when sufficiently activated, can mount a potent anti-tumor response. We identify the unique genetic sequence of an individual patient’s cancer and apply proprietary computational tools to predict which mutations, unique to that patient, are likely to be displayed by the tumor in the form of “neo-epitopes.” Accurate prediction of these neo-epitopes increases the likelihood that our AutoSynVax vaccine will stimulate a robust T cell responseagainst tumors.
We then synthesize these neo-epitopes in the form of short peptide sequences. After linking these peptides to Hsc70 in our GMP-certified manufacturing facility, AutoSynVax vaccine is administered along with our QS-21 Stimulon® adjuvant to provide maximum efficacy. By including numerous synthetic cancer antigens in each patient’s AutoSynVax vaccine, we increase the likelihood that tumors will be recognized by the immune system.PhosphoSynVaxTM
Cancers
Agenus fully-owned
preclinical
Pre
Product Description
Our PhosphoSynVax vaccine platform is based on technology we obtained when we acquired PhosImmune. PhosphoSynVax is being designed to induce immunity against a novel class of tumor specific neo-epitopes: those arising from inappropriate phosphorylation of various proteins in malignant cells. In cancer cells, the disruption of normal biochemical signaling can result in aberrant protein phosphorylation (the addition of phosphate groups on sites that would not be altered in normal cells). Some of these misphosphorylated proteins can be processed into antigens by the cellular machinery, resulting in presentation of this new type of antigen on the cell surface. We have a library of more than one thousand proprietary phosphopeptide tumor target antigens characteristic of many different cancers, including lung cancer, specific leukemias, ovarian cancer, colon cancer and others. We believe that using our PhosphoSynVax vaccines to teach the immune system to recognize these neo-epitopes can lead to the destruction of cancer cells. This technology is thus unique to Agenus and potentially very powerful in various patients across cancertypes.
ProphageTM
Glioblastoma (newly diagnosed)Agenus fully-owned
ph2
Ph2
Product Description
The Prophage vaccine platform involves extracting peptides (protein fragments) from a patient’s tumor that are naturally bound to heat shock protein gp96 (HSPPC-96). Subsequently, Prophage is designed to mobilize multiple immune processes to attack the tumor. The National Cancer Institute is currently conducting a clinical trial to evaluate Prophage vaccine in conjunction with Merck’s pembrolizumab on the overall survival rate of patients with newly diagnosed glioblastoma (ndGBM), a form of brain cancer. The standard of care treatment for glioblastoma is surgical resection, when possible, followed by radiation and chemotherapy. With today’s treatments, the vast majority of patients survive only 16 months orless.
At ASCO in June of 2015, we reported results from a single-arm, open-label, multi-institutional Phase 2 trialof
Prophage in ndGBM in combination with the standard of care treatment of surgical resection, radiation therapy and temozolomide chemotherapy. We reported that patients with ndGBM had a substantial improvement both in progression-free survival and overall survival with Prophage combined with standard of care compared to historicalcontrol data.
In addition, it was found that the most significant improvements (median progression-free survival approximating 27 months, median overall survival approximating 44.7 months) were seen in the seventeen patients with less elevated expression of the checkpoint protein PD-L1 on the circulating cells known as peripheral blood monocytes. This suggests that patients who are thought to be less immuno-suppressed when first diagnosed with ndGBM could benefit from adding Prophage vaccination to standard of care. It also raises the possibility that Prophage may be beneficial in the other half of patients who are more immuno-suppressed (those who have greater PD-L1 expression on peripheral blood monocytes at baseline) by adding checkpoint modulators that block PD-1 or PD-L1 to Prophage plus the standard ofcare.
QS-21 Stimulon®
Shingles
Partnered
approved
Generated with Avocode.Path 7Product Description
QS-21 STIMULON® ADJUVANT Agenus’ proprietary QS-21 Stimulon® is believed to be one of the most potent adjuvants known. QS-21 Stimulon is a saponin extracted from the bark of the _Quillaja saponaria_ (soap bark) evergreen tree native to warm temperate central Chile and purified using Agenus’ proprietary process. The adjuvant has been widely studied in over 120 clinical trials involving approximately 50,000 healthy volunteers and patients, and has consistently demonstrated powerful immune responses and a favorable safety profile QS-21 Stimulon® is a key component in several GSK vaccines, including the most efficacious shingles vaccine, Shingrix®, which has demonstrated >90% efficacy, as well as the first ever malaria vaccine, Mosquirix®. QS-21 Stimulon® improves a vaccine’s effectiveness by inducing strong antibody and cell-mediated immune responses. It also plays a key role by boosting immune response in older adults who often experience age related decline in immunity. In October 2017, the U.S. Food and Drug Administration (FDA) granted marketing authorization to GSK for Shingrix® for the prevention of shingles in adults aged 50 years and older. The Center for Disease Control and Prevention recently declared Shingrix® as the preferred shingles vaccine for approximately 62 million eligible adults in the United States. As a result, Shingrix® revenues have exceeded expectations, tripling analyst estimates for 2018, its first year of commercial sales. GSK has forecast that annual revenues will exceed $600M in 2018 – comparable to the $668M in revenues generated by Zostavax in 2017 after >10 years on the market. Additionally, in 2017, the World Health Organization announced that Mosquirix® will be made available to select African countries as a prophylactic measure targeted for youngchildren.
Adding QS-21 Stimulon® with our vaccines give us critical advantages: * Mutation-targeting vaccines often impart CD4 “helper” T cell responses. QS-21 Stimulon® generates both CD4 helper and CD8 killer Tcell responses;
* QS-21 Stimulon® activates the innate immune system which is a critical partner to the adaptive (T cell-based) immune response tocancer;
* In pre-clinical studies, the combination of HSP, peptides and QS-21 stimulates a potent memory response which is necessary forvaccine efficacy.
In September 2015, Agenus partially monetized its potential royalty stream from GSK through a $100M non-dilutive royalty transactionwith
an investor group led by Oberland Capital. In January 2018, Agenus repurchased the obligation from Oberland and sold the royalty stream outright to HealthCare Royalty Partners.QS-21 Stimulon®
Malaria
Partnered
filed
Filed
Product Description
QS-21 STIMULON® ADJUVANT Agenus’ proprietary QS-21 Stimulon® is believed to be one of the most potent adjuvants known. QS-21 Stimulon is a saponin extracted from the bark of the _Quillaja saponaria_ (soap bark) evergreen tree native to warm temperate central Chile and purified using Agenus’ proprietary process. The adjuvant has been widely studied in over 120 clinical trials involving approximately 50,000 healthy volunteers and patients, and has consistently demonstrated powerful immune responses and a favorable safety profile QS-21 Stimulon® is a key component in several GSK vaccines, including the most efficacious shingles vaccine, Shingrix®, which has demonstrated >90% efficacy, as well as the first ever malaria vaccine, Mosquirix®. QS-21 Stimulon® improves a vaccine’s effectiveness by inducing strong antibody and cell-mediated immune responses. It also plays a key role by boosting immune response in older adults who often experience age related decline in immunity. In October 2017, the U.S. Food and Drug Administration (FDA) granted marketing authorization to GSK for Shingrix® for the prevention of shingles in adults aged 50 years and older. The Center for Disease Control and Prevention recently declared Shingrix® as the preferred shingles vaccine for approximately 62 million eligible adults in the United States. As a result, Shingrix® revenues have exceeded expectations, tripling analyst estimates for 2018, its first year of commercial sales. GSK has forecast that annual revenues will exceed $600M in 2018 – comparable to the $668M in revenues generated by Zostavax in 2017 after >10 years on the market. Additionally, in 2017, the World Health Organization announced that Mosquirix® will be made available to select African countries as a prophylactic measure targeted for youngchildren.
Adding QS-21 Stimulon® with our vaccines give us critical advantages: * Mutation-targeting vaccines often impart CD4 “helper” T cell responses. QS-21 Stimulon® generates both CD4 helper and CD8 killer Tcell responses;
* QS-21 Stimulon® activates the innate immune system which is a critical partner to the adaptive (T cell-based) immune response tocancer;
* In pre-clinical studies, the combination of HSP, peptides and QS-21 stimulates a potent memory response which is necessary forvaccine efficacy.
In September 2015, Agenus partially monetized its potential royalty stream from GSK through a $100M non-dilutive royalty transactionwith
an investor group led by Oberland Capital. In January 2018, Agenus repurchased the obligation from Oberland and sold the royalty stream outright to HealthCare Royalty Partners. OUR INNOVATION ENGINE We have state-of-the-art technology and manufacturing platforms to repeatedly innovate with speedNOVEL TARGETS
For the development of optimal therapiesVISION
Integrated R&D platform for target discovery, drug development and intelligent clinical trial designINTELLIGENT DESIGN
VACCINE DESIGN
CELL LINE DEVELOPMENT ANTIBODY DISCOVERY AND DEVELOPMENT GMP MANUFACTURING FACILITY (Berkeley, CA and Lexington, MA) INTELLIGENT CLINICAL TRIALS DESIGNNOVEL TARGETS
For the development of optimal therapiesINTELLIGENT DESIGN
VACCINE DESIGN
CELL LINE DEVELOPMENT ANTIBODY DISCOVERY AND DEVELOPMENT GMP MANUFACTURING FACILITY (Berkeley, CA and Lexington, MA) INTELLIGENT CLINICAL TRIALS DESIGNVISION
Integrated R&D platform for target discovery, drug development and intelligent clinical trial designNOVEL TARGETS
For the development of optimal therapies AGENUS HAS SIGNIFICANT EXPERTISE IN ONCOLOGY, IMMUNOLOGY, FUNCTIONAL GENOMICS, COMPUTATIONAL BIOLOGY, STRUCTURAL BIOLOGY AND MANUFACTURING TO DRIVE AN INTEGRATED PROCESS FROM TARGET DISCOVERY TO ANTIBODY DEVELOPMENT AND MANUFACTURING.Meet our Leadership
LINKS
* About Us
* Pipeline
* Capabilities
* Investors
* Publications
AGENUS INC.
3 Forbes Road
Lexington, MA 02421-7305T: 781.674.4400
F: 781.674.4200
� 2020 Agenus Inc. All rights reserved.* Privacy Policy
* Terms and Conditions of UseDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0