Are you over 18 and want to see adult content?
Other Annotations
More Annotations

A complete backup of coworking-rive-sud.org
Are you over 18 and want to see adult content?

A complete backup of mastercard.com.br
Are you over 18 and want to see adult content?
A complete backup of commoditypandit.com
Are you over 18 and want to see adult content?

A complete backup of danger-theatre.blogspot.com
Are you over 18 and want to see adult content?

A complete backup of meupedecaju.com.br
Are you over 18 and want to see adult content?
Favourite Annotations
КомандаОПТ - канцтовари та дитяча іграшка оптом онлайн по Україні!
Are you over 18 and want to see adult content?

The Travel Intern - The Travel Intern
Are you over 18 and want to see adult content?
Shorpy Old Photos | Poster Art | Framed Prints
Are you over 18 and want to see adult content?

Flight Circle | Aircraft Scheduling & Management Software for Flight Schools and Flying Clubs
Are you over 18 and want to see adult content?

UM Janów Lubelski | Oficjalna strona miasta
Are you over 18 and want to see adult content?

SadeemRDP - Buy Cheap RDP - Buy RDP Online
Are you over 18 and want to see adult content?
Text
PRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders.INVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain Using NEUROMETRIX REPORTS Q4 AND FULL YEAR 2020 FINANCIAL The net loss of $0.3 million in Q4 2020 was an improvement of $700k from a loss of $1.1 million in the Q4 2019. For the year ended December 31, 2020, total revenues of $7.4 million declined from $9.3 million in 2019. However, gross margin improved to $5.2 million at a margin rate of 71.2% versus $2.2 million and a rate of 24.2% in theprior year.
OWNERSHIP PROFILE
The Investor Relations website contains information about NeuroMetrix 's business for stockholders, potential investors, and financialanalysts.
SHAI GOZANI
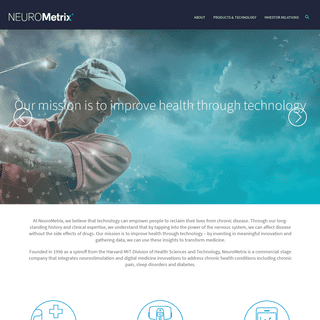
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering on NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeuticPRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders.INVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain Using NEUROMETRIX REPORTS Q4 AND FULL YEAR 2020 FINANCIAL The net loss of $0.3 million in Q4 2020 was an improvement of $700k from a loss of $1.1 million in the Q4 2019. For the year ended December 31, 2020, total revenues of $7.4 million declined from $9.3 million in 2019. However, gross margin improved to $5.2 million at a margin rate of 71.2% versus $2.2 million and a rate of 24.2% in theprior year.
OWNERSHIP PROFILE
The Investor Relations website contains information about NeuroMetrix 's business for stockholders, potential investors, and financialanalysts.
SHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering onPRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction OVERVIEW – NEUROMETRIX NeuroMetrix. Founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology, NeuroMetrix is a commercial stage, bioelectrical and digital medicine company driving innovation to address chronic health conditions including chronic pain and diabetes. Building on its roots in diagnostics technology,NeuroMetrix applied
CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capitalCOMPANY OVERVIEW
Company Overview. Company Overview. NeuroMetrix develops and commercializes health care products that utilize non-invasive neurostimulation and digital medicine. Our core expertise in biomedical engineering has been refined over two decades of designing, building and marketing medical devices that stimulate nerves and analyze nerve response for EVENTS & PRESENTATIONS Event Details. Summary Toggle. Apr 22, 2021 at 8:00 AM EDT. Q1 2021 NeuroMetrix Inc. Earnings Conference Call. Listen to webcast. Summary Toggle. Jan 28, 2021 at 8:00 AM EST. Q4 and Year-End 2020 NeuroMetrix Inc. Earnings Conference Call. Listen to webcast.PRESS RELEASES
Press Releases. WOBURN, Mass. , April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021 . The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological.INVESTOR FAQS
The phone number for Corporate and Investor Relations is. 1-781-890-9989. . The NeuroMetrix mailing address is. Investor Relations. NeuroMetrix, Inc. 1000 Winter Street. Waltham, MA 02451. You can also contact us through our online investor. NEUROMETRIX AND GLAXOSMITHKLINE CONSUMER HEALTHCARE WALTHAM, Mass., Jan. 17, 2018 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq:NURO) announced today a collaboration with GlaxoSmithKline (NYSE:GSK), one of the world's largest science-led global healthcare companies, involving Quell ® Wearable Pain Relief Technology™. The key elements of the collaboration are as follows: GSK Consumer Healthcare acquires exclusive ownership of QuellNEUROMETRIX, INC.
n e u r o m e t r i x , i n c . a n n u a l r e p o rt o n f o r m 10-k f o r t h e y e a r e n d e d d e c e m b e r 31, 2020 ta b l e of c o n t e n t s
NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeuticPRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders.INVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain Using NEUROMETRIX REPORTS Q4 AND FULL YEAR 2020 FINANCIAL The net loss of $0.3 million in Q4 2020 was an improvement of $700k from a loss of $1.1 million in the Q4 2019. For the year ended December 31, 2020, total revenues of $7.4 million declined from $9.3 million in 2019. However, gross margin improved to $5.2 million at a margin rate of 71.2% versus $2.2 million and a rate of 24.2% in theprior year.
OWNERSHIP PROFILE
The Investor Relations website contains information about NeuroMetrix 's business for stockholders, potential investors, and financialanalysts.
SHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering on NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeuticPRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders.INVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain Using NEUROMETRIX REPORTS Q4 AND FULL YEAR 2020 FINANCIAL The net loss of $0.3 million in Q4 2020 was an improvement of $700k from a loss of $1.1 million in the Q4 2019. For the year ended December 31, 2020, total revenues of $7.4 million declined from $9.3 million in 2019. However, gross margin improved to $5.2 million at a margin rate of 71.2% versus $2.2 million and a rate of 24.2% in theprior year.
OWNERSHIP PROFILE
The Investor Relations website contains information about NeuroMetrix 's business for stockholders, potential investors, and financialanalysts.
SHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering onPRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction OVERVIEW – NEUROMETRIX NeuroMetrix. Founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology, NeuroMetrix is a commercial stage, bioelectrical and digital medicine company driving innovation to address chronic health conditions including chronic pain and diabetes. Building on its roots in diagnostics technology,NeuroMetrix applied
CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capitalCOMPANY OVERVIEW
Company Overview. Company Overview. NeuroMetrix develops and commercializes health care products that utilize non-invasive neurostimulation and digital medicine. Our core expertise in biomedical engineering has been refined over two decades of designing, building and marketing medical devices that stimulate nerves and analyze nerve response for EVENTS & PRESENTATIONS Event Details. Summary Toggle. Apr 22, 2021 at 8:00 AM EDT. Q1 2021 NeuroMetrix Inc. Earnings Conference Call. Listen to webcast. Summary Toggle. Jan 28, 2021 at 8:00 AM EST. Q4 and Year-End 2020 NeuroMetrix Inc. Earnings Conference Call. Listen to webcast.PRESS RELEASES
Press Releases. WOBURN, Mass. , April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021 . The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological.INVESTOR FAQS
The phone number for Corporate and Investor Relations is. 1-781-890-9989. . The NeuroMetrix mailing address is. Investor Relations. NeuroMetrix, Inc. 1000 Winter Street. Waltham, MA 02451. You can also contact us through our online investor. NEUROMETRIX AND GLAXOSMITHKLINE CONSUMER HEALTHCARE WALTHAM, Mass., Jan. 17, 2018 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq:NURO) announced today a collaboration with GlaxoSmithKline (NYSE:GSK), one of the world's largest science-led global healthcare companies, involving Quell ® Wearable Pain Relief Technology™. The key elements of the collaboration are as follows: GSK Consumer Healthcare acquires exclusive ownership of QuellNEUROMETRIX, INC.
n e u r o m e t r i x , i n c . a n n u a l r e p o rt o n f o r m 10-k f o r t h e y e a r e n d e d d e c e m b e r 31, 2020 ta b l e of c o n t e n t s
NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeutic NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders. NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain Using NEUROMETRIX, INC. ANNOUNCES DATE FOR FIRST QUARTER 2021 WOBURN, Mass., April 15, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that it plans to issue its 2021 first quarter financial results before the opening of the market on April 22, 2021.The Company will host a conference call at 8:00 a.m., Eastern Time on April 22, 2021 to discuss its financial results as well as business developments affecting the Company. NEUROMETRIX REPORTS QUELL® TECHNOLOGY TO BE USED IN NIH WOBURN, Mass., April 16, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today announced that Quell technology has been selected for use in a large, NIH-funded, randomized, pragmatic clinical trial of TENS for fibromyalgia. Fibromyalgia is a disorder characterized by widespread musculoskeletal pain accompanied by fatigue, sleep and mooddisturbances.
OWNERSHIP PROFILE
The Investor Relations website contains information about NeuroMetrix 's business for stockholders, potential investors, and financialanalysts.
SHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering on EMAIL ALERTS SUBSCRIPTION Email Alerts Subscription. You may automatically receive NeuroMetrix, Inc. financial information by email. Please enter your preferences for email notifications below. Unsubscribe from Investor Relations email alerts. Our email alerts provide automated opt-out methods as well as complete contact information. NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeutic NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders. NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain Using NEUROMETRIX, INC. ANNOUNCES DATE FOR FIRST QUARTER 2021 WOBURN, Mass., April 15, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that it plans to issue its 2021 first quarter financial results before the opening of the market on April 22, 2021.The Company will host a conference call at 8:00 a.m., Eastern Time on April 22, 2021 to discuss its financial results as well as business developments affecting the Company. NEUROMETRIX REPORTS QUELL® TECHNOLOGY TO BE USED IN NIH WOBURN, Mass., April 16, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today announced that Quell technology has been selected for use in a large, NIH-funded, randomized, pragmatic clinical trial of TENS for fibromyalgia. Fibromyalgia is a disorder characterized by widespread musculoskeletal pain accompanied by fatigue, sleep and mooddisturbances.
OWNERSHIP PROFILE
The Investor Relations website contains information about NeuroMetrix 's business for stockholders, potential investors, and financialanalysts.
SHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering on EMAIL ALERTS SUBSCRIPTION Email Alerts Subscription. You may automatically receive NeuroMetrix, Inc. financial information by email. Please enter your preferences for email notifications below. Unsubscribe from Investor Relations email alerts. Our email alerts provide automated opt-out methods as well as complete contact information. OVERVIEW – NEUROMETRIX NeuroMetrix. Founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology, NeuroMetrix is a commercial stage, bioelectrical and digital medicine company driving innovation to address chronic health conditions including chronic pain and diabetes. Building on its roots in diagnostics technology,NeuroMetrix applied
TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.comINVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX, INC. ANNOUNCES DATE FOR FIRST QUARTER 2021 WOBURN, Mass., April 15, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that it plans to issue its 2021 first quarter financial results before the opening of the market on April 22, 2021.The Company will host a conference call at 8:00 a.m., Eastern Time on April 22, 2021 to discuss its financial results as well as business developments affecting the Company. NEUROMETRIX REPORTS Q3 2020 FINANCIAL RESULTS WOBURN, Mass., Oct. 22, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended September 30, 2020.The Company is a leading developer of neurostimulation-based medical devices that are used to diagnose and treat chronic health conditions. NEUROMETRIX REPORTS Q1 2020 FINANCIAL RESULTS Financials: In the first quarter of 2020, total revenues were $2.2 million, down 30% from $3.1 million in the prior year period. Gross margin was $1.6 million versus $0.8 million in Q1 2019. The gross margin rate of 71.4% improved from 48% in Q1 2019, excluding the effect of inventory charges taken in the year ago quarter. NEUROMETRIX REPORTS QUELL® TECHNOLOGY TO BE USED IN NIH WOBURN, Mass., April 16, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today announced that Quell technology has been selected for use in a large, NIH-funded, randomized, pragmatic clinical trial of TENS for fibromyalgia. Fibromyalgia is a disorder characterized by widespread musculoskeletal pain accompanied by fatigue, sleep and mooddisturbances.
NEUROMETRIX SIGNS DPNCHECK® COLLABORATION AGREEMENT WITH WOBURN, Mass., June 25, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported that it has signed a collaboration agreement with Biomedix to comarket DPNCheck for peripheral neuropathy detection within Medicare Advantage and other value-based care markets. NeuroMetrix and Biomedix provide complementary diagnostic tests to physicians and health plans.INVESTOR FAQS
The phone number for Corporate and Investor Relations is. 1-781-890-9989. . The NeuroMetrix mailing address is. Investor Relations. NeuroMetrix, Inc. 1000 Winter Street. Waltham, MA 02451. You can also contact us through our online investor. NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeutic OVERVIEW – NEUROMETRIX NeuroMetrix. Founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology, NeuroMetrix is a commercial stage, bioelectrical and digital medicine company driving innovation to address chronic health conditions including chronic pain and diabetes. Building on its roots in diagnostics technology,NeuroMetrix applied
PRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders.INVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain UsingSHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering on NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeutic OVERVIEW – NEUROMETRIX NeuroMetrix. Founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology, NeuroMetrix is a commercial stage, bioelectrical and digital medicine company driving innovation to address chronic health conditions including chronic pain and diabetes. Building on its roots in diagnostics technology,NeuroMetrix applied
PRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders.INVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain UsingSHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering onPRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital CAREERS – NEUROMETRIX Join our team and help change lives through innovative health technology. NeuroMetrix is a publicly-traded, innovative healthcare company that develops and markets wearable therapeutics and point-of-care tests that help consumers and physicians better managechronic pain,
BOARD OF DIRECTORS
David E. Goodman, M.D. Director. David E. Goodman, M.D. has served as a member of our Board of Directors since June 2004. Since September 2008, Dr. Goodman has served as Executive Vice President of Business Development for Masimo Corporation, a manufacturer of non-invasivepatient monitors.
FINANCIALS & FILINGS 2021. First Quarter. NeuroMetrix Reports Q1 2021 Financial Results. Q1 2021 NeuroMetrix Inc. Earnings Conference Call. Form 10-Q. CONTACT US | NEUROMETRIX The Investor Relations website contains information about NeuroMetrix 's business for stockholders, potential investors, and financialanalysts.
SEC FILINGS
SEC Filings. Report of unscheduled material events or corporate event. Report of unscheduled material events or corporate event. Quarterly report which provides a continuing view of a company's financial position. Form of prospectus disclosing information, facts, events covered in both forms 424B2, 424B3.QUARTERLY RESULTS
Fourth Quarter. NeuroMetrix Reports Q4 and Full Year 2020 Financial Results. Q4 and Year-End 2020 NeuroMetrix Inc. Earnings Conference Call. Form 10-K. Third Quarter. NeuroMetrix Reports Q3 2020 Financial Results. Q3 2020 NeuroMetrix Inc. Earnings Conference Call. Form 10-Q.Second Quarter.
INVESTMENT CALCULATOR Investment Date Original Shares Original Value Current Shares Current Value % Return Split Adjustment Current price; Jan 02, 2014: 1,000.00: $292.00: 12.50: $37.75 NEUROMETRIX AND GLAXOSMITHKLINE CONSUMER HEALTHCARE WALTHAM, Mass., Jan. 17, 2018 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq:NURO) announced today a collaboration with GlaxoSmithKline (NYSE:GSK), one of the world's largest science-led global healthcare companies, involving Quell ® Wearable Pain Relief Technology™. The key elements of the collaboration are as follows: GSK Consumer Healthcare acquires exclusive ownership of Quell NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeutic OVERVIEW – NEUROMETRIX NeuroMetrix. Founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology, NeuroMetrix is a commercial stage, bioelectrical and digital medicine company driving innovation to address chronic health conditions including chronic pain and diabetes. Building on its roots in diagnostics technology,NeuroMetrix applied
PRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders.INVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain UsingSHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering on NEUROMETRIXPRESS RELEASESCAREERSCOMPANY OVERVIEWTECHNOLOGYNEUROMETRIX CUSTOMER PORTAL LOGIN Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. NeuroMetrix was founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology. We are a leading developer and manufacturer of diagnostic and therapeutic OVERVIEW – NEUROMETRIX NeuroMetrix. Founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology, NeuroMetrix is a commercial stage, bioelectrical and digital medicine company driving innovation to address chronic health conditions including chronic pain and diabetes. Building on its roots in diagnostics technology,NeuroMetrix applied
PRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction NEUROMETRIX CUSTOMER PORTAL LOGIN To activate your account, please call Customer Service at 888-786-7287 (781-250-1400). You will be given a Login ID and a Password, which you must change after first login. CONTACT US – NEUROMETRIX CUSTOMER SERVICE. Phone +1 888 786 7287 Mon–Fri, 8:00am–6:00pm EST. customerservice@neurometrix.com TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital NEUROMETRIX REPORTS Q1 2021 FINANCIAL RESULTS WOBURN, Mass., April 22, 2021 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) today reported financial and business highlights for the quarter ended March 31, 2021.The Company is a leader in proprietary non-invasive medical devices for the diagnosis and treatment of pain and neurological disorders.INVESTOR RELATIONS
Investor Relations. Investor Relations. NeuroMetrix is a leading developer and manufacturer of diagnostic and therapeutic neurostimulation-based medical devices that are used throughout the world. The Company has three FDA cleared commercial products. DPNCheck® is a point-of-care test that is used to evaluate peripheralneuropathies.
NEUROMETRIX ANNOUNCES ISSUANCE OF NEW U.S. PATENT FOR NeuroMetrix Announces Issuance of New U.S. Patent for Quell® Wearable Technology. WOBURN, Mass., Sept. 21, 2020 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq: NURO) announced today that the U.S. Patent and Trademark Office will issue on September 22, 2020 U.S. Patent No. 10,780,269 titled “Apparatus and Method for Relieving Pain UsingSHAI GOZANI
Shai N. Gozani M.D., Ph.D. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital backed start-up, through an initial public offering onPRODUCT SERVICES
A point-of-care diagnostic platform for assessing common neuropathies. The ADVANCE System is a NeuroMetrix legacy product. Although the device is no longer marketed, we maintain and supply a large customer base that continues to utilize our disposable electrodes. For users, the ADVANCE System provides efficient and convenient nerve conduction TEAM – NEUROMETRIX Shai N. Gozani M.D., Ph.D. President, Chief Executive Officer and Director. Shai Gozani, founded NeuroMetrix, Inc. in 1996 as a spin-off from the Massachusetts Institute of Technology. He currently serves as Chairman of the board of directors and as our President and Chief Executive Officer. Shai has led the Company from a venture capital CAREERS – NEUROMETRIX Join our team and help change lives through innovative health technology. NeuroMetrix is a publicly-traded, innovative healthcare company that develops and markets wearable therapeutics and point-of-care tests that help consumers and physicians better managechronic pain,
BOARD OF DIRECTORS
David E. Goodman, M.D. Director. David E. Goodman, M.D. has served as a member of our Board of Directors since June 2004. Since September 2008, Dr. Goodman has served as Executive Vice President of Business Development for Masimo Corporation, a manufacturer of non-invasivepatient monitors.
FINANCIALS & FILINGS 2021. First Quarter. NeuroMetrix Reports Q1 2021 Financial Results. Q1 2021 NeuroMetrix Inc. Earnings Conference Call. Form 10-Q. CONTACT US | NEUROMETRIX The Investor Relations website contains information about NeuroMetrix 's business for stockholders, potential investors, and financialanalysts.
SEC FILINGS
SEC Filings. Report of unscheduled material events or corporate event. Report of unscheduled material events or corporate event. Quarterly report which provides a continuing view of a company's financial position. Form of prospectus disclosing information, facts, events covered in both forms 424B2, 424B3.QUARTERLY RESULTS
Fourth Quarter. NeuroMetrix Reports Q4 and Full Year 2020 Financial Results. Q4 and Year-End 2020 NeuroMetrix Inc. Earnings Conference Call. Form 10-K. Third Quarter. NeuroMetrix Reports Q3 2020 Financial Results. Q3 2020 NeuroMetrix Inc. Earnings Conference Call. Form 10-Q.Second Quarter.
INVESTMENT CALCULATOR Investment Date Original Shares Original Value Current Shares Current Value % Return Split Adjustment Current price; Jan 02, 2014: 1,000.00: $292.00: 12.50: $37.75 NEUROMETRIX AND GLAXOSMITHKLINE CONSUMER HEALTHCARE WALTHAM, Mass., Jan. 17, 2018 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (Nasdaq:NURO) announced today a collaboration with GlaxoSmithKline (NYSE:GSK), one of the world's largest science-led global healthcare companies, involving Quell ® Wearable Pain Relief Technology™. The key elements of the collaboration are as follows: GSK Consumer Healthcare acquires exclusive ownership of QuellSearch
* About
* PRODUCTS & TECHNOLOGY * INVESTOR RELATIONSPrevious
*
ADVANCING DIABETES CARE WITH POINT-OF-CARE DIABETIC NEUROPATHY DIAGNOSTIC TECHNOLOGY*
OUR MISSION IS TO IMPROVE HEALTH THROUGH TECHNOLOGY*
WORLD’S FIRST 24/7 WEARABLE TECHNOLOGY FOR MANAGING CHRONIC PAIN*
USING NEUROTECHNOLOGY TO SOLVE A $635 BILLION PROBLEM*
ADVANCING DIABETES CARE WITH POINT-OF-CARE DIABETIC NEUROPATHY DIAGNOSTIC TECHNOLOGY*
OUR MISSION IS TO IMPROVE HEALTH THROUGH TECHNOLOGYNext
At NeuroMetrix, we believe that technology can empower people to reclaim their lives from chronic disease. Through our long-standing history and clinical expertise, we understand that by tapping into the power of the nervous system, we can affect disease without the side effects of drugs. Our mission is to improve health through technology – by investing in meaningful innovation and gathering data, we can use these insights to transform medicine. Founded in 1996 as a spinoff from the Harvard-MIT Division of Health Sciences and Technology, NeuroMetrix is a commercial stage company that integrates neurostimulation and digital medicine innovations to address chronic health conditions including chronic pain, sleep disorders and diabetes.*
THERAPEUTICS
Offering the millions of people who suffer from chronic pain a new wearable technology for pain control utilizing advancedneurotechnology.
*
DIAGNOSTICS
Providing physicians the ability to test for diabetic neuropathy, the most common long term complication of Type 2 diabetes, accurately and rapidly at the point-of-care.*
DIGITAL HEALTH
Helping patients optimize their therapy and decrease the impact of chronic pain on their quality of life.AS FEATURED IN
*
*
*
*
*
*
This product changed my life. I work in a hospital for up to 12 hours a shift and with Quell I am now able to comfortably complete my shift with a lot less discomfort and pain. Quell works so well I wear it almost 24/7. Thank you for allowing me to continue working until Iretire in comfort.*
- “Quellrelief.com Customer”*
Life Changer! I have been suffering with chronic back pain and knee pain to the point I could no longer walk more than 5 mins without severe pain. I was skeptical about trying this but with the money back guarantee, I figured I had nothing to lose. Within the first day I noticed a huge difference. Now after 1 full week, my pain is almost gone and I can walk without pain. It’s so easy to use and set up. I even forget I have it on. If you have pain, you have nothing to lose by trying this. It will change your life!*- Jo P.
*
My Quell was easy to calibrate and position on my calf. My chronic back pain seemed to blur down in intensity. After about two weeks of daily wear, the pain relief was unbelievable. I wear it constantly even when wearing skirts and heels. The real test came the day I forgot to put it on and had a difficult time getting through my normal routine. For non-medicated chronic pain relief, there is nothing more effective than Quell.*- "Amazon Customer"
*
I received my Quell 2.0 4 weeks ago. I immediately put it on and started wearing it 20+ hrs daily. I wasn’t sure it was helping for the first 2 weeks but today is week 4 and my pain is down about 80%! I am sleeping better too. I have not had any problems with the electrodes. They last 2 weeks and I could probably stretch it a few more days. I usually leave the electrodes in the same place during the day and switch to the other leg at bedtime. Quell has made a huge improvement in my quality of life!*- Robin D.
*
This product changed my life. I work in a hospital for up to 12 hours a shift and with Quell I am now able to comfortably complete my shift with a lot less discomfort and pain. Quell works so well I wear it almost 24/7. Thank you for allowing me to continue working until Iretire in comfort.*
- “Quellrelief.com Customer”*
Life Changer! I have been suffering with chronic back pain and knee pain to the point I could no longer walk more than 5 mins without severe pain. I was skeptical about trying this but with the money back guarantee, I figured I had nothing to lose. Within the first day I noticed a huge difference. Now after 1 full week, my pain is almost gone and I can walk without pain. It’s so easy to use and set up. I even forget I have it on. If you have pain, you have nothing to lose by trying this. It will change your life!*- Jo P.
* 1
* 2
* 3
* 4
* Previous
* Next
AWARDS
SXSW Innovation AwardAARP 50+ Innovators
APMA Seal of Approval IDSA Industrial Design Excellence Award Best of CES Finalist CES Last Gadget Standing Finalist Connected Health Symposium Patient Shark Tank Award Medical Design Excellence Award* contact
*Unpaid testimonial. In a 60-day home use test conducted among 88 Quell users with lower extremity and/or low back pain, the majority of participants reported an improvement in their pain with regular use of the device. Results may vary. Copyright © 2020 NeuroMetrix, Inc.* Privacy Policy
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0