Are you over 18 and want to see adult content?
Other Annotations
More Annotations

A complete backup of flywayfareva.com
Are you over 18 and want to see adult content?

A complete backup of rybolovnorsko.cz
Are you over 18 and want to see adult content?
A complete backup of thebalancefinder.com
Are you over 18 and want to see adult content?

A complete backup of projectorscreen.com
Are you over 18 and want to see adult content?

A complete backup of jewish-heritage-europe.eu
Are you over 18 and want to see adult content?

A complete backup of storesoutletcoachonline.com
Are you over 18 and want to see adult content?

A complete backup of sahodayabbsr.com
Are you over 18 and want to see adult content?
Favourite Annotations
A complete backup of mundoprimaria.com
Are you over 18 and want to see adult content?

A complete backup of nihangunacik.com
Are you over 18 and want to see adult content?
A complete backup of inovastconcepts.com
Are you over 18 and want to see adult content?
A complete backup of fondazionecariplo.it
Are you over 18 and want to see adult content?

A complete backup of indiancinemagallery.net
Are you over 18 and want to see adult content?
A complete backup of rightbuildgroup.co.uk
Are you over 18 and want to see adult content?
Text
more.
MY NATERA - PATIENT PORTALLOG INPRODUCTS & SERVICESPRICING & BILLINGCONTACTREGISTER A TEST KITSUBMIT A BLOOD SAMPLE My Natera - Patient Portal. Conceive. Deliver. Thrive. Create an Account. Sign in. Natera's comprehensive menu of genetic tests forWomen's Health.
NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account. NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings.RENASIGHT OVERVIEW
Renasight is a test to determine if there is a genetic cause for an individual’s kidney disease or if there is an increased hereditary risk due to family history. The test uses a blood or saliva sample to test 385 genes associated with chronic kidney disease (CKD). Results are available in approximately 3 weeks. NATERA TO PRESENT NEW COLORECTAL CANCER DATA AT THE 2021 Natera to Present New Colorectal Cancer Data at the 2021 ASCO GI Symposium. Oral presentation showcases the largest early-stage CRC cohort to date, further validating the serial use of Signatera™ for early recurrence detection and MRD assessment. SAN CARLOS, Calif., Jan. 12, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneerand
TRANSFORMING MANAGEMENT OF GENETIC DISEASE Natera is a global leader in cell-free DNA (cfDNA) testing with a focus on women’s health, oncology, and organ health. Natera offers highly accurate solutions for noninvasive prenatal testing (NIPT), genetic-carrier screening, (PGD/PGS), and miscarriage testing. Learnmore.
MY NATERA - PATIENT PORTALLOG INPRODUCTS & SERVICESPRICING & BILLINGCONTACTREGISTER A TEST KITSUBMIT A BLOOD SAMPLE My Natera - Patient Portal. Conceive. Deliver. Thrive. Create an Account. Sign in. Natera's comprehensive menu of genetic tests forWomen's Health.
NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account. NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings.RENASIGHT OVERVIEW
Renasight is a test to determine if there is a genetic cause for an individual’s kidney disease or if there is an increased hereditary risk due to family history. The test uses a blood or saliva sample to test 385 genes associated with chronic kidney disease (CKD). Results are available in approximately 3 weeks. NATERA TO PRESENT NEW COLORECTAL CANCER DATA AT THE 2021 Natera to Present New Colorectal Cancer Data at the 2021 ASCO GI Symposium. Oral presentation showcases the largest early-stage CRC cohort to date, further validating the serial use of Signatera™ for early recurrence detection and MRD assessment. SAN CARLOS, Calif., Jan. 12, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneerand
OUR TECHNOLOGY
Our cell-free DNA technology workflow. Our refined workflow extends the boundaries of what’s possible with cfDNA testing by integrating high-yield DNA extraction with proprietary library preparation and bioinformatics. We’ve set the industry standard by creating cfDNA tests with greater than 99% sensitivity down to a single DNA moleculein
NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings. NATERA TO PRESENT NEW COLORECTAL CANCER DATA AT THE 2021 SAN CARLOS, Calif., Jan. 12, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneer and global leader in cell-free DNA testing, today announced it will present new data on its personalized and tumor-informed molecular residual disease (MRD) test, Signatera, at the American Society of Clinical Oncology’s 2021 Gastrointestinal Cancers Symposium (ASCO GI), taking place January 15-17, 2021. MINIMAL RESIDUAL DISEASE BY CIRCULATING TUMOR DNA ANALYSIS Odds Ratio. 95% CI: P. pT3 vs pT1–2. 5.34. 1.99–14.66: 0.001. pT4 vs pT1 –2: 4.10. 1.21 15.42: 0.03. pN1 –2 vs pN0: 1.07. 0.42 2.71: 0.87. V1 vs V0: 1.63. 0MEDICAL DIRECTORS
Medical Directors | Natera. While DNA is our business. it’s patients who fill our hearts. Natera’s diverse teams blend a passion for patients with deep clinical, scientific, and technological roots. Clinical Experts. Medical Directors. Leadership Team. Clinical Experts. Medical Directors. TUMOR-INFORMED ASSESSMENT OF CIRCULATING TUMOR DNA AND ITS Background • Hepatocellular carcinoma (HCC) and biliary tract cancers represent a heterogeneous group of diseases.¹ • Glycoprotein-based tumor markers like alpha-fetoprotein (AFP) or Carbohydrate antigen 19-9 (CA-19-9), though part of standard-of- HORIZON ADVANCED CARRIER SCREENING Horizon carrier screening helps couples determine the risk of passing on serious genetic conditions to their child. It can be performed either preconception (ideally) or during pregnancy. NEW STUDY SHEDS LIGHT ON RACIAL BARRIERS TO HEREDITARY Testing for heritable cancer mutations (e.g., BRCA1/2 mutations) informs providers about an individual’s risk of hereditary cancers. For example, patients with a mutation in BRCA1 or BRCA2 have a ~70% chance of developing breast cancer in their lifetime. 3 For patients with significant family cancer history or with a hereditary cancer mutation, medical recommendations suggest that these RISK OF RELAPSE WITH MULTIPLE MYELOMA Background • Despite treatment with high-dose chemotherapy followed by autologous stem cell transplantation (AHCT), patients with multiple myeloma (MM) invariably relapse. • Marrow minimal residual disease (MRD) in MM has emerged as the most important prognostic marker. SPECTRUM PREIMPLANTATION GENETICS (PGT) Spectrum preimplantation genetic testing (PGT) evaluates embryos for extra or missing chromosomes (PGT-A), single gene conditions (PGT-M), or structural rearrangements (PGT-SR), improving the chances of a healthy pregnancy. Spectrum tests for chromosomal abnormalities and TRANSFORMING MANAGEMENT OF GENETIC DISEASE Natera is a global leader in cell-free DNA (cfDNA) testing with a focus on women’s health, oncology, and organ health. Natera offers highly accurate solutions for noninvasive prenatal testing (NIPT), genetic-carrier screening, (PGD/PGS), and miscarriage testing. Learnmore.
MY NATERA - PATIENT PORTALLOG INPRODUCTS & SERVICESPRICING & BILLINGCONTACTREGISTER A TEST KITSUBMIT A BLOOD SAMPLE My Natera - Patient Portal. Conceive. Deliver. Thrive. Create an Account. Sign in. Natera's comprehensive menu of genetic tests forWomen's Health.
NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account.RENASIGHT OVERVIEW
Renasight is a test to determine if there is a genetic cause for an individual’s kidney disease or if there is an increased hereditary risk due to family history. The test uses a blood or saliva sample to test 385 genes associated with chronic kidney disease (CKD). Results are available in approximately 3 weeks. NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings.JOB OPENINGS
Join us, make a difference. Behind every Natera test, more than 1,800 Naterans are working together to change the way that disease ismanaged.
LEADERSHIP TEAM
Robert Schueren is an accomplished industry veteran with extensive molecular diagnostics, genomics, and pharma experience. Mr. Schueren has enjoyed an expansive 30-year career and has been a driving force behind rapid business growth at both start-up and large biotechcompanies.
NATERA TO PRESENT NEW COLORECTAL CANCER DATA AT THE 2021 Natera to Present New Colorectal Cancer Data at the 2021 ASCO GI Symposium. Oral presentation showcases the largest early-stage CRC cohort to date, further validating the serial use of Signatera™ for early recurrence detection and MRD assessment. SAN CARLOS, Calif., Jan. 12, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneerand
EMPOWER OVERVIEW
Empower is a genetic test for those who want to know more about their risk of developing cancer or why it might be common in their family. Empower tests up to 53 genes associated with risk for common hereditary cancers using blood or saliva samples. Results are delivered within 2-3 weeks. IMPROVED PATIENT ACCESS Improved patient access Affordable Pricing and Billing Options Order Submission Test Processed Results reported out Insurance claim ˜ledExplanation
TRANSFORMING MANAGEMENT OF GENETIC DISEASE Natera is a global leader in cell-free DNA (cfDNA) testing with a focus on women’s health, oncology, and organ health. Natera offers highly accurate solutions for noninvasive prenatal testing (NIPT), genetic-carrier screening, (PGD/PGS), and miscarriage testing. Learnmore.
MY NATERA - PATIENT PORTALLOG INPRODUCTS & SERVICESPRICING & BILLINGCONTACTREGISTER A TEST KITSUBMIT A BLOOD SAMPLE My Natera - Patient Portal. Conceive. Deliver. Thrive. Create an Account. Sign in. Natera's comprehensive menu of genetic tests forWomen's Health.
NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account.RENASIGHT OVERVIEW
Renasight is a test to determine if there is a genetic cause for an individual’s kidney disease or if there is an increased hereditary risk due to family history. The test uses a blood or saliva sample to test 385 genes associated with chronic kidney disease (CKD). Results are available in approximately 3 weeks. NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings.JOB OPENINGS
Join us, make a difference. Behind every Natera test, more than 1,800 Naterans are working together to change the way that disease ismanaged.
LEADERSHIP TEAM
Robert Schueren is an accomplished industry veteran with extensive molecular diagnostics, genomics, and pharma experience. Mr. Schueren has enjoyed an expansive 30-year career and has been a driving force behind rapid business growth at both start-up and large biotechcompanies.
NATERA TO PRESENT NEW COLORECTAL CANCER DATA AT THE 2021 Natera to Present New Colorectal Cancer Data at the 2021 ASCO GI Symposium. Oral presentation showcases the largest early-stage CRC cohort to date, further validating the serial use of Signatera™ for early recurrence detection and MRD assessment. SAN CARLOS, Calif., Jan. 12, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneerand
EMPOWER OVERVIEW
Empower is a genetic test for those who want to know more about their risk of developing cancer or why it might be common in their family. Empower tests up to 53 genes associated with risk for common hereditary cancers using blood or saliva samples. Results are delivered within 2-3 weeks. IMPROVED PATIENT ACCESS Improved patient access Affordable Pricing and Billing Options Order Submission Test Processed Results reported out Insurance claim ˜ledExplanation
NATERA PORTAL HUB
Visit the appropriate Natera™ portal to check on results, schedule conversations with genetic counselors, place orders, or make payments.OUR TECHNOLOGY
Our cell-free DNA technology workflow. Our refined workflow extends the boundaries of what’s possible with cfDNA testing by integrating high-yield DNA extraction with proprietary library preparation and bioinformatics. We’ve set the industry standard by creating cfDNA tests with greater than 99% sensitivity down to a single DNA moleculein
NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings. NATERA TO PRESENT NEW COLORECTAL CANCER DATA AT THE 2021 SAN CARLOS, Calif., Jan. 12, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneer and global leader in cell-free DNA testing, today announced it will present new data on its personalized and tumor-informed molecular residual disease (MRD) test, Signatera, at the American Society of Clinical Oncology’s 2021 Gastrointestinal Cancers Symposium (ASCO GI), taking place January 15-17, 2021. NATERA ANNOUNCES NEW PROSPERA™ DATA AT THE ATC 2021 AUSTIN, Texas, June 4, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneer and global leader in cell-free DNA (cfDNA) testing, today announced it will present new data on its latest scientific innovations in cfDNA testing at the American Transplant Congress’ (ATC) 2021 Virtual Connect, taking place June 4-9, 2021. SPECTRUM PREIMPLANTATION GENETICS (PGT) Spectrum preimplantation genetic testing (PGT) evaluates embryos for extra or missing chromosomes (PGT-A), single gene conditions (PGT-M), or structural rearrangements (PGT-SR), improving the chances of a healthy pregnancy. Spectrum tests for chromosomal abnormalities and NEW STUDY SHEDS LIGHT ON RACIAL BARRIERS TO HEREDITARY Testing for heritable cancer mutations (e.g., BRCA1/2 mutations) informs providers about an individual’s risk of hereditary cancers. For example, patients with a mutation in BRCA1 or BRCA2 have a ~70% chance of developing breast cancer in their lifetime. 3 For patients with significant family cancer history or with a hereditary cancer mutation, medical recommendations suggest that these MINIMAL RESIDUAL DISEASE BY CIRCULATING TUMOR DNA ANALYSIS Odds Ratio. 95% CI: P. pT3 vs pT1–2. 5.34. 1.99–14.66: 0.001. pT4 vs pT1 –2: 4.10. 1.21 15.42: 0.03. pN1 –2 vs pN0: 1.07. 0.42 2.71: 0.87. V1 vs V0: 1.63. 0 TUMOR-INFORMED ASSESSMENT OF CIRCULATING TUMOR DNA AND ITS Background • Hepatocellular carcinoma (HCC) and biliary tract cancers represent a heterogeneous group of diseases.¹ • Glycoprotein-based tumor markers like alpha-fetoprotein (AFP) or Carbohydrate antigen 19-9 (CA-19-9), though part of standard-of- RISK OF RELAPSE WITH MULTIPLE MYELOMA Background • Despite treatment with high-dose chemotherapy followed by autologous stem cell transplantation (AHCT), patients with multiple myeloma (MM) invariably relapse. • Marrow minimal residual disease (MRD) in MM has emerged as the most important prognostic marker. TRANSFORMING MANAGEMENT OF GENETIC DISEASE Natera is a global leader in cell-free DNA (cfDNA) testing with a focus on women’s health, oncology, and organ health. Natera offers highly accurate solutions for noninvasive prenatal testing (NIPT), genetic-carrier screening, (PGD/PGS), and miscarriage testing. Learnmore.
NATERA PORTAL HUB
Visit the appropriate Natera™ portal to check on results, schedule conversations with genetic counselors, place orders, or make payments.WOMEN'S HEALTH
Women’s Health. Natera ™ offers a full menu of high-quality genetic tests to support the entire spectrum of women’s health needs—from planning a family and MY NATERA - PATIENT PORTAL My Natera - Patient Portal. Conceive. Deliver. Thrive. Create an Account. Sign in. Natera's comprehensive menu of genetic tests forWomen's Health.
NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings.JOB OPENINGS
Join us, make a difference. Behind every Natera test, more than 1,800 Naterans are working together to change the way that disease ismanaged.
NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account.RENASIGHT OVERVIEW
Renasight is a test to determine if there is a genetic cause for an individual’s kidney disease or if there is an increased hereditary risk due to family history. The test uses a blood or saliva sample to test 385 genes associated with chronic kidney disease (CKD). Results are available in approximately 3 weeks.LEADERSHIP TEAM
Robert Schueren is an accomplished industry veteran with extensive molecular diagnostics, genomics, and pharma experience. Mr. Schueren has enjoyed an expansive 30-year career and has been a driving force behind rapid business growth at both start-up and large biotechcompanies.
EMPOWER OVERVIEW
Empower is a genetic test for those who want to know more about their risk of developing cancer or why it might be common in their family. Empower tests up to 53 genes associated with risk for common hereditary cancers using blood or saliva samples. Results are delivered within 2-3 weeks. TRANSFORMING MANAGEMENT OF GENETIC DISEASE Natera is a global leader in cell-free DNA (cfDNA) testing with a focus on women’s health, oncology, and organ health. Natera offers highly accurate solutions for noninvasive prenatal testing (NIPT), genetic-carrier screening, (PGD/PGS), and miscarriage testing. Learnmore.
NATERA PORTAL HUB
Visit the appropriate Natera™ portal to check on results, schedule conversations with genetic counselors, place orders, or make payments.WOMEN'S HEALTH
Women’s Health. Natera ™ offers a full menu of high-quality genetic tests to support the entire spectrum of women’s health needs—from planning a family and MY NATERA - PATIENT PORTAL My Natera - Patient Portal. Conceive. Deliver. Thrive. Create an Account. Sign in. Natera's comprehensive menu of genetic tests forWomen's Health.
NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings.JOB OPENINGS
Join us, make a difference. Behind every Natera test, more than 1,800 Naterans are working together to change the way that disease ismanaged.
NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account.RENASIGHT OVERVIEW
Renasight is a test to determine if there is a genetic cause for an individual’s kidney disease or if there is an increased hereditary risk due to family history. The test uses a blood or saliva sample to test 385 genes associated with chronic kidney disease (CKD). Results are available in approximately 3 weeks.LEADERSHIP TEAM
Robert Schueren is an accomplished industry veteran with extensive molecular diagnostics, genomics, and pharma experience. Mr. Schueren has enjoyed an expansive 30-year career and has been a driving force behind rapid business growth at both start-up and large biotechcompanies.
EMPOWER OVERVIEW
Empower is a genetic test for those who want to know more about their risk of developing cancer or why it might be common in their family. Empower tests up to 53 genes associated with risk for common hereditary cancers using blood or saliva samples. Results are delivered within 2-3 weeks.WOMEN'S HEALTH
Women’s Health. Natera ™ offers a full menu of high-quality genetic tests to support the entire spectrum of women’s health needs—from planning a family and NATERA ANNOUNCES NEW PROSPERA™ DATA AT THE ATC 2021 AUSTIN, Texas, June 4, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneer and global leader in cell-free DNA (cfDNA) testing, today announced it will present new data on its latest scientific innovations in cfDNA testing at the American Transplant Congress’ (ATC) 2021 Virtual Connect, taking place June 4-9, 2021. WOMEN'S HEALTH PRICING & BILLING Anora – Miscarriage Test. Spectrum – Preimplantation Genetics. FOR PATIENTS. Pricing and Billing Information. Women’s Health Portal. FOR CLINICIANS. NateraConnect Provider Portal. Horizon Conditions List. NEVA for Empower – Natera’s Educational Virtual Assistant.CAREERS | NATERA
What people are saying about Natera. “Natera truly cares about making a difference with their products, services and cutting-edge technology. For once, I felt like I truly belonged, and that my efforts were really adding value to the company’s mission. My team was full of amazing people.”. NEW STUDY SHEDS LIGHT ON RACIAL BARRIERS TO HEREDITARY Testing for heritable cancer mutations (e.g., BRCA1/2 mutations) informs providers about an individual’s risk of hereditary cancers. For example, patients with a mutation in BRCA1 or BRCA2 have a ~70% chance of developing breast cancer in their lifetime. 3 For patients with significant family cancer history or with a hereditary cancer mutation, medical recommendations suggest that theseSIGNATERA OVERVIEW
Signatera is a personalized, tumor-informed assay optimized to detect circulating tumor DNA (ctDNA) for molecular residual disease (MRD) assessment and recurrence monitoring for patients previously diagnosed with cancer, with broad utility for cancer management.PROSPERA OVERVIEW
Covered by Medicare, Prospera is a transplant rejection test that uses a simple blood draw to assess the risk of rejection of a transplanted kidney. Through the use of advanced cell-free DNA technology, Prospera increases a provider’s ability to identify otherwise undetected rejection that might lead to kidney loss.NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account.ASCO | NATERA
Explore the three new studies recently presented at the 2020 American Society of Clinical Oncology (ASCO) virtual conference and access additional content below. The oral presentation and 2 posters highlight the unique applications of Signatera to detect recurrence using circulating-tumor DNA (ctDNA).HORIZON FAQ
Board-certified genetic counselors are available to answer questions about Horizon carrier screening Monday through Friday, 6 AM to 5 PM Pacific Time, excluding holidays. Please call 844-778-4700 and ask to speak with the on-call genetic counselor. You may also email gc@natera.com with questions. TRANSFORMING MANAGEMENT OF GENETIC DISEASE Natera is a global leader in cell-free DNA (cfDNA) testing with a focus on women’s health, oncology, and organ health. Natera offers highly accurate solutions for noninvasive prenatal testing (NIPT), genetic-carrier screening, (PGD/PGS), and miscarriage testing. Learnmore.
NATERA PORTAL HUB
Visit the appropriate Natera™ portal to check on results, schedule conversations with genetic counselors, place orders, or make payments.WOMEN'S HEALTH
Women’s health testing can help you plan for a healthy baby. Visit our page to learn about the prenatal and natal genetic testing thatNatera offers.
MY NATERA - PATIENT PORTAL � 2021 Natera, Inc. The test described has been developed and its performance characteristics determined by the CLIA-certified laboratory performing the test.RENASIGHT OVERVIEW
Comprehensive: Leverages next generation sequencing to analyze over 380 genes that were selected by genetic experts to provide clinically actionable information. Convenient: Remote testing services including virtual ordering for providers and at home sample collection for patients (saliva or mobile blood draw).Complimentary information sessions with board-certified genetic counselors. NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings.NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account.JOB OPENINGS
The tests described have been developed and their performance characteristics determined by the CLIA-certified laboratory performingthe test.
LEADERSHIP TEAM
Robert Schueren is an accomplished industry veteran with extensive molecular diagnostics, genomics, and pharma experience. Mr. Schueren has enjoyed an expansive 30-year career and has been a driving force behind rapid business growth at both start-up and large biotechcompanies.
EMPOWER OVERVIEW
Empower is a genetic test for those who want to know more about their risk of developing cancer, why it might be common in their family, or want to inform treatment options following a cancer diagnosis. TRANSFORMING MANAGEMENT OF GENETIC DISEASE Natera is a global leader in cell-free DNA (cfDNA) testing with a focus on women’s health, oncology, and organ health. Natera offers highly accurate solutions for noninvasive prenatal testing (NIPT), genetic-carrier screening, (PGD/PGS), and miscarriage testing. Learnmore.
NATERA PORTAL HUB
Visit the appropriate Natera™ portal to check on results, schedule conversations with genetic counselors, place orders, or make payments.WOMEN'S HEALTH
Women’s health testing can help you plan for a healthy baby. Visit our page to learn about the prenatal and natal genetic testing thatNatera offers.
MY NATERA - PATIENT PORTAL � 2021 Natera, Inc. The test described has been developed and its performance characteristics determined by the CLIA-certified laboratory performing the test.RENASIGHT OVERVIEW
Comprehensive: Leverages next generation sequencing to analyze over 380 genes that were selected by genetic experts to provide clinically actionable information. Convenient: Remote testing services including virtual ordering for providers and at home sample collection for patients (saliva or mobile blood draw).Complimentary information sessions with board-certified genetic counselors. NATERA NEWS: PRESS RELEASES Natera News - your source for the latest Genetics & Genomics news, company information, and business listings.NATERA CONNECT
You appear to be trying to log in with a natera.com email address. If you're a Natera employee, please log in using your Okta account.JOB OPENINGS
The tests described have been developed and their performance characteristics determined by the CLIA-certified laboratory performingthe test.
LEADERSHIP TEAM
Robert Schueren is an accomplished industry veteran with extensive molecular diagnostics, genomics, and pharma experience. Mr. Schueren has enjoyed an expansive 30-year career and has been a driving force behind rapid business growth at both start-up and large biotechcompanies.
EMPOWER OVERVIEW
Empower is a genetic test for those who want to know more about their risk of developing cancer, why it might be common in their family, or want to inform treatment options following a cancer diagnosis.WOMEN'S HEALTH
Women’s health testing can help you plan for a healthy baby. Visit our page to learn about the prenatal and natal genetic testing thatNatera offers.
NATERA ANNOUNCES NEW PROSPERA™ DATA AT THE ATC 2021 AUSTIN, Texas, June 4, 2021 /PRNewswire/ — Natera, Inc. (NASDAQ: NTRA), a pioneer and global leader in cell-free DNA (cfDNA) testing, today announced it will present new data on its latest scientific innovations in cfDNA testing at the American Transplant Congress’ (ATC) 2021 Virtual Connect, taking place June 4-9, 2021. WOMEN'S HEALTH PRICING & BILLING Natera provides personalized cost estimates through our Price Transparency Program (PTP). If you provide your insurance information, and we estimate your out-of-pocket cost to exceed our cash price, we will contact you to discuss alternative payment options.CAREERS | NATERA
Behind our approach is a mosaic of people, ideas, and experiences. Be a part of the synergy of perspectives that drives our innovation and leadership in genetic testing.PROSPERA OVERVIEW
With a 95% negative predictive value, Prospera misses ~3x fewer rejections than serum creatinine. 1 Prospera’s proprietary donor-derived cell-free DNA (dd-cfDNA) technology offers early warning signs of transplant rejection and reduces the likelihood of renal allograft failure with a non-invasive single blood draw. 1,2 NEW STUDY SHEDS LIGHT ON RACIAL BARRIERS TO HEREDITARY Testing for heritable cancer mutations (e.g., BRCA1/2 mutations) informs providers about an individual’s risk of hereditary cancers. For example, patients with a mutation in BRCA1 or BRCA2 have a ~70% chance of developing breast cancer in their lifetime. 3 For patients with significant family cancer history or with a hereditary cancer mutation, medical recommendations suggest that these HORIZON ADVANCED CARRIER SCREENING Horizon carrier screening helps couples determine the risk of passing on serious genetic conditions to their child. It can be performed either preconception (ideally) or during pregnancy.SIGNATERA OVERVIEW
Signatera is a personalized, tumor-informed assay optimized to detect circulating tumor DNA (ctDNA) for molecular residual disease (MRD) assessment and recurrence monitoring for patients previously diagnosed with cancer, with broad utility for cancer management.ASCO | NATERA
A study evaluating the validity of Signatera ctDNA assay to detect molecular residual disease (MRD) in CRC patients who are at high risk of recurrence, during and after adjuvant chemotherapy.HORIZON FAQ
Horizon can help you and your partner learn about the chance to have a child with a genetic disease before or during pregnancy. Many people do not know they are a carrier for an inherited genetic disease until they have an affected child. WE USE COOKIES ON THIS SITE TO ENHANCE YOUR USER EXPERIENCE By clicking any link on this page you are giving your consent for usto set cookies.
No, give me more infoI agree
Skip to main content Google Tag Manager* Careers
* Patient Portal
* Payment Portal
* Provider Portal
Toggle navigation Menu* Products
* HORIZON
Advanced carrier screening* SPECTRUM
Preimplantation genetics* PANORAMA
Next-generation NIPT* VISTARA
Single-gene NIPT
* ANORA
Miscarriage test (POC)* EVERCORD
Newborn stem cell banking* SIGNATERA
Molecular monitoring (MRD)* PROSPERA
Transplant assessment* CONSTELLATION
Technology licensingX
* About Natera
* Our Company
* Our People
* Our Community
* Science & Informatics* Careers
* For Reproductive Testing * Horizon Carrier Screen* Spectrum PGT
* Panorama Prenatal Test* Vistara
* Anora Miscarriage Test * For Cord Blood Banking* Evercord
* Pricing
* For Oncology
* Signatera
* For Organ Transplant* Prospera
* For Laboratories
* Constellation
*
-------------------------* Patient Portal
* Provider Portal
* Resources
* Webinars
* Test Cost Estimator * Find Clinical Specialist * Talk to a Genetic Counselor*
-------------------------* Media
* Investor Relations* Contact Us
*
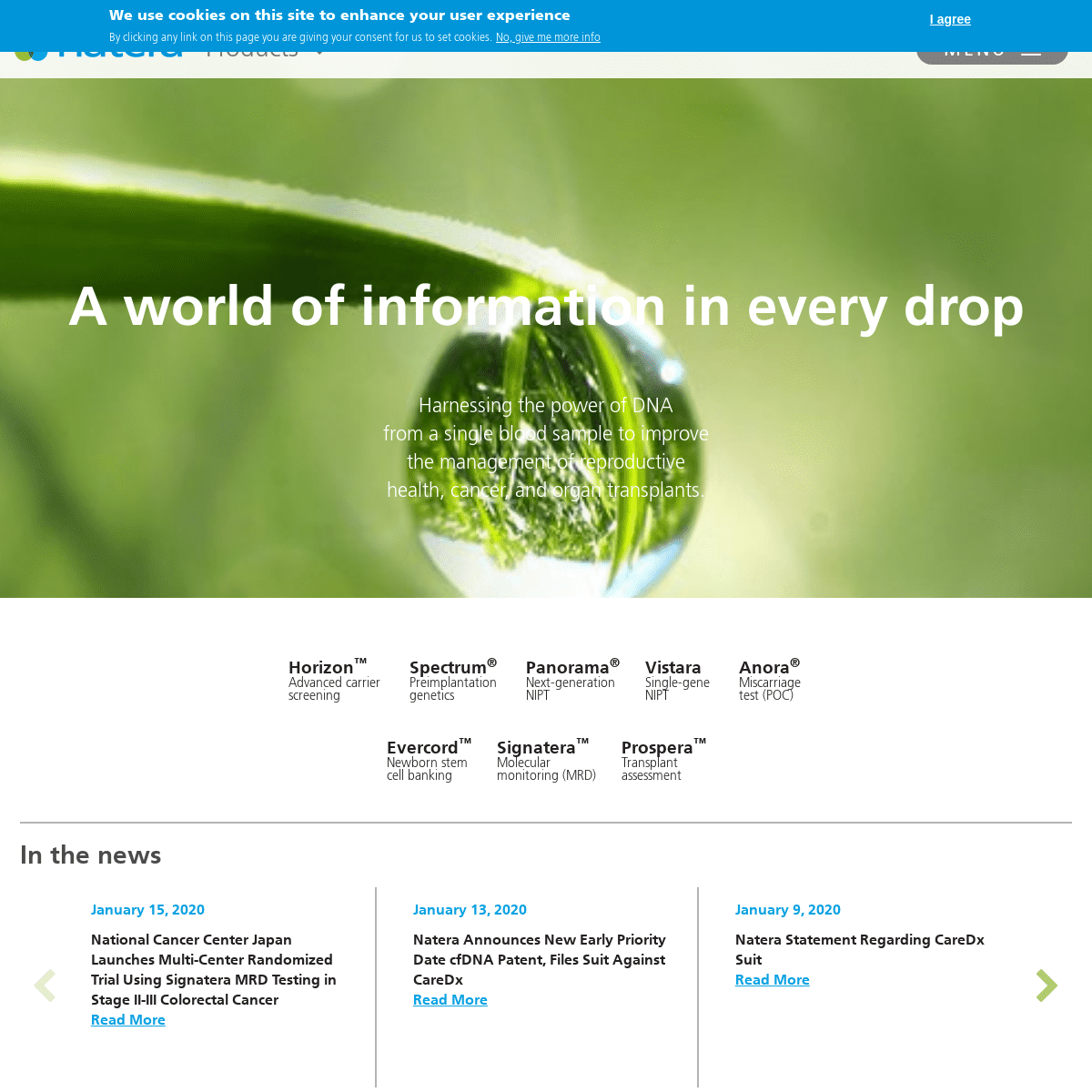
------------------------- A WORLD OF INFORMATION IN EVERY DROP Harnessing the power of DNA from a single blood sample to improve the management of reproductive health, cancer, and organ transplants. A WORLD OF INFORMATION IN EVERY DROP Harnessing the power of DNA from a single blood sample to improve the management of reproductive health, cancer, and organ transplants. A WORLD OF INFORMATION IN EVERY DROP Harnessing the power of DNA from a single blood sample to improve the management of reproductive health, cancer, and organ transplants. A WORLD OF INFORMATION IN EVERY DROP Harnessing the power of DNA from a single blood sample to improve the management of reproductive health, cancer, and organ transplants. A WORLD OF INFORMATION IN EVERY DROP Harnessing the power of DNA from a single blood sample to improve the management of reproductive health, cancer, and organ transplants.Horizon™
Advanced carrier
screening
Spectrum®
Preimplantation
genetics
Panorama®
Next-generation
NIPT
Vistara
Single-gene
NIPT
Anora®
Miscarriage
test (POC)
Evercord™
Newborn stem
cell banking
Signatera™
Molecular
monitoring (MRD)
Prospera™
Transplant
assessment
IN THE NEWS
JANUARY 15, 2020
National Cancer Center Japan Launches Multi-Center Randomized Trial Using Signatera MRD Testing in Stage II-III Colorectal CancerRead More
JANUARY 13, 2020
Natera Announces New Early Priority Date cfDNA Patent, Files SuitAgainst CareDx
Read More
JANUARY 9, 2020
Natera Statement Regarding CareDx SuitRead More
JANUARY 9, 2020
Natera Launches BESPOKE CRC Study to Evaluate Outcomes in 1,000 Signatera Patients with Stage II-III Colorectal CancerRead More
JANUARY 8, 2020
Natera to Webcast Live Presentation at the 38th Annual J.P. Morgan Healthcare ConferenceRead More
JANUARY 6, 2020
Natera Announces Achievement of $55 Million in Pharma Contracts and Issuance of New Oncology PatentsRead More
DECEMBER 19, 2019
Natera Receives Final Medicare Coverage for Prospera™ Organ Transplant Rejection TestRead More
NOVEMBER 27, 2019
Panorama Publication Demonstrates Utility of Measuring Individual Fetal Fractions in Twin PregnanciesRead More
NOVEMBER 6, 2019
Natera Reports Third Quarter 2019 Financial ResultsRead More
OCTOBER 30, 2019
Natera Announces Third Quarter 2019 Earnings Conference CallRead More
OCTOBER 17, 2019
Natera Announces Pricing of Follow-On OfferingRead More
OCTOBER 15, 2019
Natera Launches Proposed Follow-On OfferingRead More
OCTOBER 3, 2019
Natera to Host Investor Conference Call to Discuss Progress in itsOncology Business
Read More
SEPTEMBER 24, 2019
Foundation Medicine and Natera Partner to Advance Personalized CancerMonitoring
Read More
SEPTEMBER 13, 2019
CBR to Acquire Natera's Evercord™ Cord Blood BusinessRead More
SEPTEMBER 9, 2019
Natera Publishes Largest NIPT Outcomes Study Demonstrating Robust Clinical Performance Over a 4-year PeriodRead More
SEPTEMBER 4, 2019
Natera to Present at the Morgan Stanley 17th Annual Global HealthcareConference
Read More
AUGUST 29, 2019
Natera Appoints Rowan Chapman to its Board of DirectorsRead More
AUGUST 28, 2019
Natera to Webcast Live Presentation at the Baird 2019 Global Healthcare ConferenceRead More
AUGUST 22, 2019
Medicare Issues Positive Draft Local Coverage Determination for Natera's Signatera™ MRD Test in Colorectal CancerRead More
AUGUST 22, 2019
Panorama NIPT Achieves 2 Milion Test MilestoneRead More
AUGUST 7, 2019
Natera Reports Second Quarter 2019 Financial ResultsRead More
AUGUST 5, 2019
Publication Demonstrates Unique Ability of Panorama Test to Determine Zygosity in Twin PregnanciesRead More
JULY 31, 2019
Natera Announces Second Quarter 2019 Earnings Conference CallRead More
JUNE 24, 2019
New ProActive™ Study From Natera Will Test for Rejection in 3,000 Kidney Transplant RecipientsRead More
JUNE 3, 2019
Data Presented at ASCO Shows Natera's Signatera Test Detects Immunotherapy Treatment Response in Metastatic CancersRead More
MAY 9, 2019
Natera Reports First Quarter 2019 Financial ResultsRead More
MAY 9, 2019
New Blood Test Detects Colorectal Cancer Recurrence Up to 16.5 MonthsEarlier
Read More
MAY 7, 2019
Signatera™ Blood Test Detects Bladder Cancer Relapse Up to Eight Months Earlier Than Current Clinical StandardsRead More
MAY 6, 2019
FDA Grants Breakthrough Device Designation to Natera's Signatera TestRead More
MAY 2, 2019
Natera Announces First Quarter 2019 Earnings Conference CallRead More
APRIL 16, 2019
New Publication Validates Clinical Performance of Signatera for Recurrence Monitoring and MRD Assessment in Breast CancerRead More
APRIL 10, 2019
Natera Announces Plans to Commercialize Tumor Whole Exome Sequencingfrom Plasma
Read More
MARCH 28, 2019
Medicare Issues Draft Local Coverage Determination for Natera's New Prospera™ Kidney Transplant Rejection TestRead More
MARCH 26, 2019
Natera to Partner with AMAL Therapeutics on Clinical Trial Using Signatera MRD Test to Assess Treatment Response to Colorectal CancerVaccine
Read More
MARCH 12, 2019
Natera Reports Fourth Quarter and Fiscal Year 2018 Financial ResultsRead More
MARCH 11, 2019
Natera and BGI Genomics Announce $50M Partnership to Commercialize Signatera Oncology Test in China and to Develop Reproductive Health Tests in Select Markets on BGI's DNBseq™ Technology PlatformRead More
MARCH 5, 2019
Natera, Inc. Announces Fourth Quarter and Fiscal 2018 EarningsConference Call
Read More
FEBRUARY 22, 2019
Natera Announces Publication of Analytical Validation Study Demonstrating Superior Precision of Its Kidney Transplant RejectionAssay
Read More
FEBRUARY 19, 2019
Natera Presents Outcomes Data at SMFM Annual Meeting Showing Strong Clinical Performance of Its SNP-Based Non-Invasive Prenatal TestRead More
FEBRUARY 1, 2019
Natera Announces Agreement with One Lambda to Co-Distribute Its Kidney Transplant Rejection TestRead More
JANUARY 7, 2019
Natera Announces Publication of Kidney Transplant Validation Study, Demonstrating Superior Data in Detection of Clinical and SubclinicalRejection
Read More
JANUARY 4, 2019
Natera To Webcast Live Presentation at the 37th Annual J.P. Morgan Healthcare ConferenceRead More
JANUARY 4, 2019
Natera Obtains Z-Codes for Signatera MRD and Treatment Monitoring Testin Oncology
Read More
JANUARY 2, 2019
Natera Appoints Steve Chapman CEO and Robert Schueren COO; Matthew Rabinowitz Moves to Executive ChairmanRead More
DECEMBER 6, 2018
Natera Presents Breast Cancer Data at SABCS Showing Ability of Signatera (RUO) to Detect Molecular Residual Disease Up to Two Years Prior to Clinical Relapse and Predict Treatment ResponseRead More
NOVEMBER 29, 2018
Natera Achieves Enrollment Goal of 20,000 Patients in SMART StudyRead More
NOVEMBER 28, 2018
Natera Receives Unique Z-Code for Its Kidney Transplant RejectionMonitoring Test
Read More
NOVEMBER 8, 2018
Natera Reports Third Quarter 2018 Financial ResultsRead More
OCTOBER 18, 2018
Natera and Neon Therapeutics Announce Clinical Trial Collaboration to Assess Treatment Response to Personal Cancer VaccineRead More
JULY 12, 2018
Natera Announces Pricing of Follow-On OfferingRead More
JUNE 21, 2018
Natera Develops Powerful Kidney Transplant Rejection BiomarkerRead More
JUNE 19, 2018
Natera and Institut Jules Bordet Announce Collaboration in NeoadjuvantBreast Cancer
Read More
JUNE 13, 2018
Natera Announces New Clinical Utility Data and Issuance of CPT Code for NIPT in Twin PregnanciesRead More
Previous Next
FAMILY OF REPRODUCTIVE HEALTH GENETIC TESTS NATERA® IS DRIVEN BY A PASSION FOR IMPROVING THE PATH TO PARENTHOOD WITH A SUITE OF HIGH-QUALITY PRODUCTS THAT SUPPORT FAMILIES IN THEIR JOURNEY FROM PRECONCEPTION, TO PREGNANCY, AND BIRTH.LEARN MORE
FAMILY OF REPRODUCTIVE HEALTH GENETIC TESTS NATERA® IS DRIVEN BY A PASSION FOR IMPROVING THE PATH TO PARENTHOOD WITH A SUITE OF HIGH-QUALITY PRODUCTS THAT SUPPORT FAMILIES IN THEIR JOURNEY FROM PRECONCEPTION, TO PREGNANCY, AND BIRTH.LEARN MORE
FAMILY OF REPRODUCTIVE HEALTH GENETIC TESTS NATERA® IS DRIVEN BY A PASSION FOR IMPROVING THE PATH TO PARENTHOOD WITH A SUITE OF HIGH-QUALITY PRODUCTS THAT SUPPORT FAMILIES IN THEIR JOURNEY FROM PRECONCEPTION, TO PREGNANCY, AND BIRTH.LEARN MORE
FAMILY OF REPRODUCTIVE HEALTH GENETIC TESTS NATERA® IS DRIVEN BY A PASSION FOR IMPROVING THE PATH TO PARENTHOOD WITH A SUITE OF HIGH-QUALITY PRODUCTS THAT SUPPORT FAMILIES IN THEIR JOURNEY FROM PRECONCEPTION, TO PREGNANCY, AND BIRTH.LEARN MORE
FAMILY OF REPRODUCTIVE HEALTH GENETIC TESTS Natera® is driven by a passion for improving the path to parenthood with a suite of high-quality products that support families in their journey from preconception, to pregnancy, and birth.LEARN MORE
Applying revolutionary technology to change the management of disease worldwide is at the heart of Natera’s mission.CUTTING-EDGE
ONCOLOGY RESEARCH
NATERA IS DEVELOPING NEW TOOLS TO REVOLUTIONIZE TREATMENT MONITORING, RECURRENCE MONITORING, AND RESIDUAL DISEASE DETECTION ACROSS MANYCANCER TYPES.
LEARN MORE
CUTTING-EDGE
ONCOLOGY RESEARCH
NATERA IS DEVELOPING NEW TOOLS TO REVOLUTIONIZE TREATMENT MONITORING, RECURRENCE MONITORING, AND RESIDUAL DISEASE DETECTION ACROSS MANYCANCER TYPES.
LEARN MORE
CUTTING-EDGE
ONCOLOGY RESEARCH
NATERA IS DEVELOPING NEW TOOLS TO REVOLUTIONIZE TREATMENT MONITORING, RECURRENCE MONITORING, AND RESIDUAL DISEASE DETECTION ACROSS MANYCANCER TYPES.
LEARN MORE
CUTTING-EDGE
ONCOLOGY RESEARCH
NATERA IS DEVELOPING NEW TOOLS TO REVOLUTIONIZE TREATMENT MONITORING, RECURRENCE MONITORING, AND RESIDUAL DISEASE DETECTION ACROSS MANYCANCER TYPES.
LEARN MORE
CUTTING-EDGE
ONCOLOGY RESEARCH
Natera is developing new tools to revolutionize treatment monitoring, recurrence monitoring, and residual disease detection across manycancer types.
LEARN MORE
POWERFUL ORGAN
TRANSPLANT ASSAY
Natera is applying its expertise in cell-free DNA to detect organ transplant injury and rejection earlier, in a non-invasive and moreprecise way.
Learn More
POWERFUL ORGAN
TRANSPLANT ASSAY
Natera is applying its expertise in cell-free DNA to detect organ transplant injury and rejection earlier, in a non-invasive and moreprecise way.
Learn More
POWERFUL ORGAN
TRANSPLANT ASSAY
Natera is applying its expertise in cell-free DNA to detect organ transplant injury and rejection earlier, in a non-invasive and moreprecise way.
Learn More
POWERFUL ORGAN
TRANSPLANT ASSAY
Natera is applying its expertise in cell-free DNA to detect organ transplant injury and rejection earlier, in a non-invasive and moreprecise way.
Learn More
POWERFUL ORGAN
TRANSPLANT ASSAY
Natera is applying its expertise in cell-free DNA to detect organ transplant injury and rejection earlier, in a non-invasive and moreprecise way.
Learn More
FOOTER_DISCLAIMER
FOOTER_DISCLAIMER
�2020 Natera, Inc. These tests have been developed and their performance characteristics determined by the CLIA-certified laboratory performing the test. These tests have not been cleared or approved by the U.S. Food and Drug Administration (FDA). Although FDA does not currently clear or approve laboratory-developed tests in the U.S., certification of the laboratory is required under CLIA to ensure the quality and validity of the tests.FOOTER_LEGAL
* Natera.com
* Careers
* Terms of Use
* Privacy Policy
* Notice of Privacy Practices * Notice of Data Collection for California Residents* Licenses
* Sitemap
__ Back to Top
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0