Are you over 18 and want to see adult content?
More Annotations
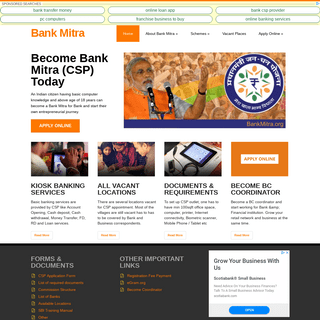
BankMitra.org - Become Bank Mitra CSP today - 100% trusted
Are you over 18 and want to see adult content?
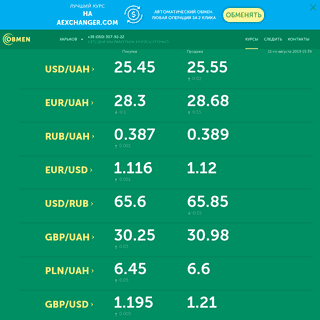
Обмен валюты (Харьков): Круглосуточный обмен валют. Оптовый обмен евро, доллара, фунтов, злотых, крон
Are you over 18 and want to see adult content?

LinuxBabe - Linux Sysadmin | Desktop Linux | Raspberry Pi
Are you over 18 and want to see adult content?

Franchise en France : Créer votre Franchise et Devenir Franchisé en 2019
Are you over 18 and want to see adult content?

The Story of Mathematics - A History of Mathematical Thought from Ancient Times to the Modern Day
Are you over 18 and want to see adult content?

Contrainformacion.es - Periodismo incómodo
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of americasbest.com
Are you over 18 and want to see adult content?
A complete backup of emeraldfloors.com
Are you over 18 and want to see adult content?
A complete backup of americansamoa.gov
Are you over 18 and want to see adult content?
A complete backup of midwifethinking.com
Are you over 18 and want to see adult content?
A complete backup of kamedaseika.co.jp
Are you over 18 and want to see adult content?

A complete backup of python-graph-gallery.com
Are you over 18 and want to see adult content?
A complete backup of paxchristiusa.org
Are you over 18 and want to see adult content?
Text
LYOPHILIZATION
INTRODUCTION. Freeze-drying, also known as lyophilization, is the process of removing ice from a formulation through sublimation. This typically consists of three different steps during the process. First, the solution is cooled to freeze the material. Next, primary drying is conducted to remove bulk ice by establishing a pressure differential BD MEDICAL - PHARMACEUTICAL SYSTEMS - DRUG DEVELOPMENT AND BD Medical – Pharmaceutical Systems Offers Innovative, High-Quality, Clinically Proven & Customized Pre-fillable Parenteral Drug Delivery Systems. BD’s drug delivery systems are designed to protect, package, and deliver drug therapies and to maximize healthcare worker and patient safety. – Pre-fillable Syringes: BD is uniquelypositioned
SPECIAL FEATURE
SPECIAL FEATURE – Platform Technologies: Not Just for Big Pharma. Platform technologies are considered a valuable tool to improve efficiency and quality in drug product development. The basic idea is that a platform, in combination with a risk-based approach, is the most systematic method to leverage prior knowledge for a given newmolecule.
ZIARCO PHARMA ACQUIRES PFIZER DRUGS, SECURES FINANCING Ziarco Pharma Acquires Pfizer Drugs, Secures Financing. Ziarco recently announced the closing of an initial $6-million tranche of Series A financing totalling $27 million. The round was led by Biotechnology Value Fund L.P, with participation by Pfizer Venture Investments. Concurrent with the financing, Ziarco has entered into anagreement with
PATIENT SAFETY NARRATIVES Patient safety narratives are a key element in clinical study reporting. We will look at current regulatory requirements regarding safety narratives, a proposed process for their development, and review and examine ways to simplify the reporting process. These procedures are aimed atCELL & GENE THERAPY
Aldo Romano and Emily Moran, MBA, discuss how the life science industry must bring technology solutions to the table while at the same time securing the capacity to develop and manufacture groundbreaking cures so patients and society can tap the benefits of cell and gene therapies. LACTOSE IN PHARMACEUTICAL APPLICATIONS Lactose in Pharmaceutical Applications. INTRODUCTION Lactose is a naturally occurring simple carbohydrate, or sugar, found only in the milk of mammals. For this reason, it is also commonly referred to as “milk sugar.”. All commercial lactose is obtained from the milk ofcows as a
DRUG DEVELOPMENT & DELIVERY Drug Development & Delivery is a print and online content provider that presents the latest scientific methods in drug development for professionals in R&D, APIs, pre-formulation, formulation development and analytical methods. ABOUT US - DRUG DEVELOPMENT AND DELIVERY About us. Drug Development & Delivery, including its exclusive Specialty Pharma section, is a print publication, conference organizer (Drug Development Networking Summit), and online content provider exclusively committed to advancing the applied science, technology, and related business practices of pharmaceutical and biological drug MAY 2021 - DRUG DEVELOPMENT AND DELIVERY Philip Kuehl, PhD, and Kimberly B. Shepard, PhD, describe the formulation and manufacturing considerations for development of dry-powder therapeutics for local treatment of lung cancer. Two case studies are presented in which two approved drugs are successfully formulated for DPI administration toLYOPHILIZATION
INTRODUCTION. Freeze-drying, also known as lyophilization, is the process of removing ice from a formulation through sublimation. This typically consists of three different steps during the process. First, the solution is cooled to freeze the material. Next, primary drying is conducted to remove bulk ice by establishing a pressure differential BD MEDICAL - PHARMACEUTICAL SYSTEMS - DRUG DEVELOPMENT AND BD Medical – Pharmaceutical Systems Offers Innovative, High-Quality, Clinically Proven & Customized Pre-fillable Parenteral Drug Delivery Systems. BD’s drug delivery systems are designed to protect, package, and deliver drug therapies and to maximize healthcare worker and patient safety. – Pre-fillable Syringes: BD is uniquelypositioned
SPECIAL FEATURE
SPECIAL FEATURE – Platform Technologies: Not Just for Big Pharma. Platform technologies are considered a valuable tool to improve efficiency and quality in drug product development. The basic idea is that a platform, in combination with a risk-based approach, is the most systematic method to leverage prior knowledge for a given newmolecule.
ZIARCO PHARMA ACQUIRES PFIZER DRUGS, SECURES FINANCING Ziarco Pharma Acquires Pfizer Drugs, Secures Financing. Ziarco recently announced the closing of an initial $6-million tranche of Series A financing totalling $27 million. The round was led by Biotechnology Value Fund L.P, with participation by Pfizer Venture Investments. Concurrent with the financing, Ziarco has entered into anagreement with
PATIENT SAFETY NARRATIVES Patient safety narratives are a key element in clinical study reporting. We will look at current regulatory requirements regarding safety narratives, a proposed process for their development, and review and examine ways to simplify the reporting process. These procedures are aimed atCELL & GENE THERAPY
Aldo Romano and Emily Moran, MBA, discuss how the life science industry must bring technology solutions to the table while at the same time securing the capacity to develop and manufacture groundbreaking cures so patients and society can tap the benefits of cell and gene therapies. LACTOSE IN PHARMACEUTICAL APPLICATIONS Lactose in Pharmaceutical Applications. INTRODUCTION Lactose is a naturally occurring simple carbohydrate, or sugar, found only in the milk of mammals. For this reason, it is also commonly referred to as “milk sugar.”. All commercial lactose is obtained from the milk ofcows as a
SPECIAL FEATURE
SPECIAL FEATURE – Platform Technologies: Not Just for Big Pharma. Platform technologies are considered a valuable tool to improve efficiency and quality in drug product development. The basic idea is that a platform, in combination with a risk-based approach, is the most systematic method to leverage prior knowledge for a given newmolecule.
ITEOS THERAPEUTICS ANNOUNCES NEW PHASE 1/2A DATA iTeos Therapeutics, Inc. recently announced new clinical data from its ongoing Phase 1/2a clinical trial of inupadenant (EOS-850), a next-generation adenosine receptor (A 2A R) antagonist, at the American Society of Clinical Oncology (ASCO) Annual Meeting 2021. Updated results from the single-agent dose-escalation and expansion portions of the trial provided evidence of durable antitumor IN8BIO COMPLETES TREATMENT OF FIRST COHORT IN PHASE 1 IN8bio, Inc. recently announced an update from the ongoing Phase 1 clinical trial of INB-200, its DeltEx drug resistant immunotherapy (DRI), MGMT-gene modified gamma delta T NOBLE FURTHER EXPANDS CAPABILITIES OF ITS NEW HUMAN In direct response to the rapid growth of the recently launched Human Factors + (HF+) program, Noble, an Aptar Pharma company, recently announced the appointment of two new members to its internal HF+ team.In the four months since launch, the HF+ service has experienced this growth across each of its patient-administered medical deviceprograms.
EXTRACTABLES & LEACHABLES THE EXTRACTABLES SCREEN. USP chapter Assessment of Extractables Associated with Pharmaceutical Packaging/Delivery Systems is the basis for the chemical safety assessment section of USP , the organic extractables profile of draft USP , and the chemical safety qualification of draft USP .It is, therefore, the cornerstone to determine whether a packaging system is HARPOON THERAPEUTICS PRESENTS UPDATED INTERIM CLINICAL Harpoon Therapeutics, Inc. recently presented interim data from the ongoing dose-escalation portion of a Phase 1/2a trial for HPN424 in patients with metastatic castration-resistant prostate cancer (mCRPC) at the 2021 American Society of Clinical Oncology (ASCO) AnnualMeeting.
BAYER TO ADVANCE TWO FIRST-OF-ITS-KIND CELL & GENE Bayer AG recently announced that BlueRock Therapeutics (BlueRock) successfully administered the first dose of its pluripotent stem cell-derived dopaminergic neurons, named DA01, to a Parkinson’s disease patient in their open-label Phase 1 clinical study. YOURWAY BREAKS GROUND ON MAJOR EXPANSION, INCLUDING PHASE Yourway recently announced an expansion underway at its headquarters in Allentown, PA. An expanded complex is being constructed for Yourway’s global operation, adding an additional 3,500 pallet locations of refrigerated 2°C-8°C space and increasing the footprint of the Allentown campus to over 300,000 sq ft. ADDEX STARTS PHASE 2 CLINICAL STUDY OF ADX71149 FOR EPILEPSY A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices. AKOYA & ASTRAZENECA TO COLLABORATE ON SPATIAL BIOLOGY Akoya Biosciences, Inc. recently announced a collaboration with AstraZeneca to advance new multiplex immunofluorescence (mIF) workflows and spatial biomarker signatures, based on DRUG DEVELOPMENT & DELIVERY Drug Development & Delivery is a print and online content provider that presents the latest scientific methods in drug development for professionals in R&D, APIs, pre-formulation, formulation development and analytical methods. ZIARCO PHARMA ACQUIRES PFIZER DRUGS, SECURES FINANCING Ziarco Pharma Acquires Pfizer Drugs, Secures Financing. Ziarco recently announced the closing of an initial $6-million tranche of Series A financing totalling $27 million. The round was led by Biotechnology Value Fund L.P, with participation by Pfizer Venture Investments. Concurrent with the financing, Ziarco has entered into anagreement with
SPECIAL FEATURE
SPECIAL FEATURE – Platform Technologies: Not Just for Big Pharma. Platform technologies are considered a valuable tool to improve efficiency and quality in drug product development. The basic idea is that a platform, in combination with a risk-based approach, is the most systematic method to leverage prior knowledge for a given newmolecule.
LYOPHILIZATION
INTRODUCTION. Freeze-drying, also known as lyophilization, is the process of removing ice from a formulation through sublimation. This typically consists of three different steps during the process. First, the solution is cooled to freeze the material. Next, primary drying is conducted to remove bulk ice by establishing a pressure differential DARÉ BIOSCIENCE ANNOUNCES RECEIPT OF $1.5 MILLION UNDER A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices. INTAC TECHNOLOGY COMBATS MISUSE OF EXTENDED-RELEASE DRUGS During this year’s Annual Scientific Meeting of the American Pain Society (May 16-19, 2012, Honolulu, HI), Grunenthal presented results from clinical studies utilizing a product based on its INTAC formulation technology.INTAC is designed to be crush-resistant and toprotect
AKORN TO ACQUIRE VERSAPHARM FOR $440 MILLION Akorn to Acquire VersaPharm for $440 Million. Akorn, Inc. recently announced it has entered into a definitive agreement to acquire VPI Holdings Corp., the parent company of VersaPharm Incorporated, for $440 million in cash. VersaPharm, a privately held developer and marketer of multi-source prescription pharmaceuticals, has a focus inthe niche
MARKET BRIEF
The combined AD and MCI populations in the 7MM are expected to reach approximately 48 million people by 2026, which represents an annual growth rate of 2.3% from 2016. In the 2016 base year, 34.5% of market share ($1.1 billion) was attributed to mild AD, which generated the HEART FAILURE MARKET TO SOAR TO $11.8 BILLION BY 2025 Heart Failure Market to Soar to $11.8 Billion by 2025. The heart failure market is set to rise from around $3.2 billion in 2015 to $11.8 billion by 2025, representing a compound annual growth rate of 13.7%, according to research and consulting firm GlobalData. The company’s latest report, PharmaPoint: Heart Failure – Global DrugForecast
INDONESIAN PHARMACEUTICAL MARKET SEES RAPID EXPANSION Indonesian Pharmaceutical Market Sees Rapid Expansion. Driven by increased government healthcare spending and growing life expectancy, the Indonesian pharmaceutical market value will climb from approximately $5 billion in 2013 to $9.9 billion by 2020, at a Compound Annual Growth Rate (CAGR) of 10.2%, forecasts research andconsulting firm
DRUG DEVELOPMENT & DELIVERY Drug Development & Delivery is a print and online content provider that presents the latest scientific methods in drug development for professionals in R&D, APIs, pre-formulation, formulation development and analytical methods. ZIARCO PHARMA ACQUIRES PFIZER DRUGS, SECURES FINANCING Ziarco Pharma Acquires Pfizer Drugs, Secures Financing. Ziarco recently announced the closing of an initial $6-million tranche of Series A financing totalling $27 million. The round was led by Biotechnology Value Fund L.P, with participation by Pfizer Venture Investments. Concurrent with the financing, Ziarco has entered into anagreement with
SPECIAL FEATURE
SPECIAL FEATURE – Platform Technologies: Not Just for Big Pharma. Platform technologies are considered a valuable tool to improve efficiency and quality in drug product development. The basic idea is that a platform, in combination with a risk-based approach, is the most systematic method to leverage prior knowledge for a given newmolecule.
LYOPHILIZATION
INTRODUCTION. Freeze-drying, also known as lyophilization, is the process of removing ice from a formulation through sublimation. This typically consists of three different steps during the process. First, the solution is cooled to freeze the material. Next, primary drying is conducted to remove bulk ice by establishing a pressure differential DARÉ BIOSCIENCE ANNOUNCES RECEIPT OF $1.5 MILLION UNDER A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices. INTAC TECHNOLOGY COMBATS MISUSE OF EXTENDED-RELEASE DRUGS During this year’s Annual Scientific Meeting of the American Pain Society (May 16-19, 2012, Honolulu, HI), Grunenthal presented results from clinical studies utilizing a product based on its INTAC formulation technology.INTAC is designed to be crush-resistant and toprotect
AKORN TO ACQUIRE VERSAPHARM FOR $440 MILLION Akorn to Acquire VersaPharm for $440 Million. Akorn, Inc. recently announced it has entered into a definitive agreement to acquire VPI Holdings Corp., the parent company of VersaPharm Incorporated, for $440 million in cash. VersaPharm, a privately held developer and marketer of multi-source prescription pharmaceuticals, has a focus inthe niche
MARKET BRIEF
The combined AD and MCI populations in the 7MM are expected to reach approximately 48 million people by 2026, which represents an annual growth rate of 2.3% from 2016. In the 2016 base year, 34.5% of market share ($1.1 billion) was attributed to mild AD, which generated the HEART FAILURE MARKET TO SOAR TO $11.8 BILLION BY 2025 Heart Failure Market to Soar to $11.8 Billion by 2025. The heart failure market is set to rise from around $3.2 billion in 2015 to $11.8 billion by 2025, representing a compound annual growth rate of 13.7%, according to research and consulting firm GlobalData. The company’s latest report, PharmaPoint: Heart Failure – Global DrugForecast
INDONESIAN PHARMACEUTICAL MARKET SEES RAPID EXPANSION Indonesian Pharmaceutical Market Sees Rapid Expansion. Driven by increased government healthcare spending and growing life expectancy, the Indonesian pharmaceutical market value will climb from approximately $5 billion in 2013 to $9.9 billion by 2020, at a Compound Annual Growth Rate (CAGR) of 10.2%, forecasts research andconsulting firm
NANOMEDICINES
V Smart Nanomedicines are game changers that are designed, engineered, and customized to deliver non-brain penetrant therapeutic agents across the BBB, target and selectively release at specific brain sites or cells and be administered systemically. Lauren Sciences’ innovative pipeline of transformative drugs in successful developmentfor CNS
CLINICAL TRIALS
Karen Ooms, Msc, believes the consequences of this pandemic on ongoing clinical trials can be objectively assessed, and with the correct mitigation strategies put in place, study integrity can be preserved, optimizing use of the available resources for both patients andsponsors.
MAY 2021 - DRUG DEVELOPMENT AND DELIVERY Philip Kuehl, PhD, and Kimberly B. Shepard, PhD, describe the formulation and manufacturing considerations for development of dry-powder therapeutics for local treatment of lung cancer. Two case studies are presented in which two approved drugs are successfully formulated for DPI administration to BIO PLATFORMS ARCHIVES 6/3/2021 Corbus Pharmaceuticals Licenses Two Integrin Targeting mAbs Further Expanding Pipeline Into Cancer & Fibrotic Diseases. Corbus Pharmaceuticals Holdings, Inc. recently announced licensing deals for two new monoclonal antibodies (mAbs), CRB-601 and CRB-602, that target integrins to inhibit activation of.DRUG DELIVERY
Mikolaj Milewski and Amitava Mitra highlight some of the recent advances in the field of microneedle-mediated transdermal drug delivery and vaccination, including summaries of preclinical pharmacokinetic data and a brief overview of clinical studies, encompassing the time period from 2010 to present day.TOPICAL DEVELOPMENT
THE RISK CHALLENGE. Recent reviews have suggested that it takes the evaluation of 10,000 compounds to produce one approved medicine containing a new chemical entity (NCE), and the development time is typically between 10 and 14 years with costs in excess of $1 billion. 1,2 Nevertheless, the continual discovery of new potential target pathways, the re-purposing of drugs, and ARTICLES - PAGE 250 OF 251 - DRUG DEVELOPMENT AND DELIVERY Corbus Pharmaceuticals Licenses Two Integrin Targeting mAbs Further Expanding Pipeline Into Cancer & Fibrotic Diseases. Corbus Pharmaceuticals Holdings, Inc. recently announced licensing deals for two new monoclonal antibodies (mAbs), CRB-601 and CRB-602, that target integrins to inhibit activation of. CONTRACT SERVICES ARCHIVES 5/26/2021 Evonik Markets New Enteric-Protected Ready-to-Fill Capsules for Fast, High-Performance Drug Development. Evonik recently launched the EUDRACAP platform of easy-to-handle capsules to help the pharmaceutical industry accelerate speed to market for complex oral drug products in early development stages.. PATIENT SAFETY NARRATIVES Patient safety narratives are a key element in clinical study reporting. We will look at current regulatory requirements regarding safety narratives, a proposed process for their development, and review and examine ways to simplify the reporting process. These procedures are aimed atLYOPHILIZATION
INTRODUCTION. The global lyophilization market for pharmaceutical and biotechnology products, valued at $1.97 billion in 2014, is expected to increase to $2.66 billion by 2019, with a compound annual growth rate (CAGR) of 6.2%. 1 About 25% of new injectable drugs, vaccines, and biological products are formulated in a lyophilized form, and for many proteins, peptides, and vaccines, this DRUG DEVELOPMENT & DELIVERY Drug Development & Delivery is a print and online content provider that presents the latest scientific methods in drug development for professionals in R&D, APIs, pre-formulation, formulation development and analytical methods. ZIARCO PHARMA ACQUIRES PFIZER DRUGS, SECURES FINANCING Ziarco recently announced the closing of an initial $6-million tranche of Series A financing totalling $27 million. The round was led by Biotechnology Value Fund L.P, with participation by DARÉ BIOSCIENCE ANNOUNCES RECEIPT OF $1.5 MILLION UNDER A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices.SPECIAL FEATURE
Platform technologies are considered a valuable tool to improve efficiency and quality in drug product development. The basic idea is that a platform, in combination with a risk-based approach, is the most systematic method to leverage prior knowledge for a given newmolecule.
LYOPHILIZATION
A third type of temperature sensor is the Tempris ® system (Figure 3). 3 It is a wireless sensor consisting of a handmade quartz crystal that oscillates in a temperature-dependent frequency. The sensor is excited by a modulated microwave signal, and the response is overlaid with the carrier signal leading to a frequency shift from which the product temperature is derived. INTAC TECHNOLOGY COMBATS MISUSE OF EXTENDED-RELEASE DRUGS During this year’s Annual Scientific Meeting of the American Pain Society (May 16-19, 2012, Honolulu, HI), Grunenthal presented results from clinical studies utilizing a product based on its INTAC formulation technology.INTAC is designed to be crush-resistant and toprotect
MARKET BRIEF
Akiko Fukui is a Neurology and Ophthalmology Healthcare Analyst at GlobalData in London.Prior to joining GlobalData, she was an Associate at PAREXEL Consulting, where she worked on consulting projects advising companies on market access, pricing, and reimbursementdecisions.
AKORN TO ACQUIRE VERSAPHARM FOR $440 MILLION Akorn, Inc. recently announced it has entered into a definitive agreement to acquire VPI Holdings Corp., the parent company of VersaPharm Incorporated, for $440 million in cash. HEART FAILURE MARKET TO SOAR TO $11.8 BILLION BY 2025 The heart failure market is set to rise from around $3.2 billion in 2015 to $11.8 billion by 2025, representing a compound annual growth rate of 13.7%, according to research and INDONESIAN PHARMACEUTICAL MARKET SEES RAPID EXPANSION Driven by increased government healthcare spending and growing life expectancy, the Indonesian pharmaceutical market value will climb from approximately $5 billion in 2013 to $9.9 billion by 2020, at a Compound Annual Growth Rate (CAGR) of 10.2%, forecasts research and consulting firm GlobalData. DRUG DEVELOPMENT & DELIVERY Drug Development & Delivery is a print and online content provider that presents the latest scientific methods in drug development for professionals in R&D, APIs, pre-formulation, formulation development and analytical methods. ZIARCO PHARMA ACQUIRES PFIZER DRUGS, SECURES FINANCING Ziarco recently announced the closing of an initial $6-million tranche of Series A financing totalling $27 million. The round was led by Biotechnology Value Fund L.P, with participation by DARÉ BIOSCIENCE ANNOUNCES RECEIPT OF $1.5 MILLION UNDER A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices.SPECIAL FEATURE
Platform technologies are considered a valuable tool to improve efficiency and quality in drug product development. The basic idea is that a platform, in combination with a risk-based approach, is the most systematic method to leverage prior knowledge for a given newmolecule.
LYOPHILIZATION
A third type of temperature sensor is the Tempris ® system (Figure 3). 3 It is a wireless sensor consisting of a handmade quartz crystal that oscillates in a temperature-dependent frequency. The sensor is excited by a modulated microwave signal, and the response is overlaid with the carrier signal leading to a frequency shift from which the product temperature is derived. INTAC TECHNOLOGY COMBATS MISUSE OF EXTENDED-RELEASE DRUGS During this year’s Annual Scientific Meeting of the American Pain Society (May 16-19, 2012, Honolulu, HI), Grunenthal presented results from clinical studies utilizing a product based on its INTAC formulation technology.INTAC is designed to be crush-resistant and toprotect
MARKET BRIEF
Akiko Fukui is a Neurology and Ophthalmology Healthcare Analyst at GlobalData in London.Prior to joining GlobalData, she was an Associate at PAREXEL Consulting, where she worked on consulting projects advising companies on market access, pricing, and reimbursementdecisions.
AKORN TO ACQUIRE VERSAPHARM FOR $440 MILLION Akorn, Inc. recently announced it has entered into a definitive agreement to acquire VPI Holdings Corp., the parent company of VersaPharm Incorporated, for $440 million in cash. HEART FAILURE MARKET TO SOAR TO $11.8 BILLION BY 2025 The heart failure market is set to rise from around $3.2 billion in 2015 to $11.8 billion by 2025, representing a compound annual growth rate of 13.7%, according to research and INDONESIAN PHARMACEUTICAL MARKET SEES RAPID EXPANSION Driven by increased government healthcare spending and growing life expectancy, the Indonesian pharmaceutical market value will climb from approximately $5 billion in 2013 to $9.9 billion by 2020, at a Compound Annual Growth Rate (CAGR) of 10.2%, forecasts research and consulting firm GlobalData.CLINICAL TRIALS
Karen Ooms, Msc, believes the consequences of this pandemic on ongoing clinical trials can be objectively assessed, and with the correct mitigation strategies put in place, study integrity can be preserved, optimizing use of the available resources for both patients andsponsors.
NANOMEDICINES
Susan Rosenbaum, JD, and Irwin Hollander, PhD, present a unique and novel solution to this greatest challenge in medicine for brain disease treatment, considered the “Holy Grail of Neuroscience,” with their breakthrough innovation. MAY 2021 - DRUG DEVELOPMENT AND DELIVERY COVER SPECIAL FEATURE – PFS & Parenteral Manufacturing: How COVID-19 Changed the Market. Contributor Cindy Dubin explores how device developers and parenteral contract development and manufacturing organizations (CDMOs) are addressing current challenges, as well as advancements in customized device design, the continuous effort to incorporate safety and human factors, and how PATIENT SAFETY NARRATIVES INTRODUCTION. Patient safety narratives are a key element in clinical study reporting. We will look at current regulatory requirements regarding safety narratives, a proposed process for their development, and review and examine ways to simplify the reporting process. These procedures are aimed at reducing the burden of time and cost.. Patient safety narratives should be prepared for allDRUG DELIVERY
Mikolaj Milewski and Amitava Mitra highlight some of the recent advances in the field of microneedle-mediated transdermal drug delivery and vaccination, including summaries of preclinical pharmacokinetic data and a brief overview of clinical studies, encompassing the time period from 2010 to present day. BIO PLATFORMS ARCHIVES 6/3/2021 Corbus Pharmaceuticals Licenses Two Integrin Targeting mAbs Further Expanding Pipeline Into Cancer & Fibrotic Diseases. Corbus Pharmaceuticals Holdings, Inc. recently announced licensing deals for two new monoclonal antibodies (mAbs), CRB-601 and CRB-602, that target integrins to inhibit activation of.TOPICAL DEVELOPMENT
THE RISK CHALLENGE. Recent reviews have suggested that it takes the evaluation of 10,000 compounds to produce one approved medicine containing a new chemical entity (NCE), and the development time is typically between 10 and 14 years with costs in excess of $1 billion. 1,2 Nevertheless, the continual discovery of new potential target pathways, the re-purposing of drugs, and ARTICLES - PAGE 250 OF 251 - DRUG DEVELOPMENT AND DELIVERY Corbus Pharmaceuticals Licenses Two Integrin Targeting mAbs Further Expanding Pipeline Into Cancer & Fibrotic Diseases. Corbus Pharmaceuticals Holdings, Inc. recently announced licensing deals for two new monoclonal antibodies (mAbs), CRB-601 and CRB-602, that target integrins to inhibit activation of. PARTICULATE IDENTIFICATION Dr. Markus Lankers is President and Co-founder of rap-ID Particle Systems GmbH that develops and manufactures instruments for rapid particle identification and siliconization measurements. He researches and develops solutions for particulate analysis. He previously worked with Schering AG. He helped to establish the Visual Inspection of Parenterals Interest Group and serves as program co-chair CONTRACT SERVICES ARCHIVES 5/26/2021 Evonik Markets New Enteric-Protected Ready-to-Fill Capsules for Fast, High-Performance Drug Development. Evonik recently launched the EUDRACAP platform of easy-to-handle capsules to help the pharmaceutical industry accelerate speed to market for complex oral drug products in early development stages.. DRUG DEVELOPMENT & DELIVERY Drug Development & Delivery is a print and online content provider that presents the latest scientific methods in drug development for professionals in R&D, APIs, pre-formulation, formulation development and analytical methods. ABOUT US - DRUG DEVELOPMENT AND DELIVERY Drug Development & Delivery, including its exclusive Specialty Pharma section, is a print publication, conference organizer (Drug Development Networking Summit), and online content provider exclusively committed to advancing the applied science, technology, and related business practices of pharmaceutical and biological drug development, reporting on formulation design, bioavailability and ZIARCO PHARMA ACQUIRES PFIZER DRUGS, SECURES FINANCINGZIARCO PHARMA LTDDR MIKE YEADON PFIZER OPINIONDR YEADON PFIZERMICHAEL YEADON PFIZER Ziarco recently announced the closing of an initial $6-million tranche of Series A financing totalling $27 million. The round was led by Biotechnology Value Fund L.P, with participation byTOPICAL DEVELOPMENT
THE RISK CHALLENGE. Recent reviews have suggested that it takes the evaluation of 10,000 compounds to produce one approved medicine containing a new chemical entity (NCE), and the development time is typically between 10 and 14 years with costs in excess of $1 billion. 1,2 Nevertheless, the continual discovery of new potential target pathways, the re-purposing of drugs, and advantageous DARÉ BIOSCIENCE ANNOUNCES RECEIPT OF $1.5 MILLION UNDER A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices. INTAC TECHNOLOGY COMBATS MISUSE OF EXTENDED-RELEASE DRUGS During this year’s Annual Scientific Meeting of the American Pain Society (May 16-19, 2012, Honolulu, HI), Grunenthal presented results from clinical studies utilizing a product based on its INTAC formulation technology.INTAC is designed to be crush-resistant and toprotect
AKORN TO ACQUIRE VERSAPHARM FOR $440 MILLION Akorn, Inc. recently announced it has entered into a definitive agreement to acquire VPI Holdings Corp., the parent company of VersaPharm Incorporated, for $440 million in cash. WHITE PAPER: CONTROL OF BETA-GLUCANS & ENDOTOXIN IN HIGHBEST SOURCE OF BETA GLUCANSWHAT ARE BETA GLUCANSFOODS HIGH IN BETA GLUCANSBETA GLUCAN DOSAGE FOR CANCERBETA GLUCANS AND CANCERENDOTOXIN ENHANCEMENT Sucrose or saccharose is a disaccharide composed of glucose and fructose. Figure 1, depicts the chemical structure of sucrose. It has been used in protein stabilization particularly biotherapeutics since the past three decades.MARKET BRIEF
Akiko Fukui is a Neurology and Ophthalmology Healthcare Analyst at GlobalData in London.Prior to joining GlobalData, she was an Associate at PAREXEL Consulting, where she worked on consulting projects advising companies on market access, pricing, and reimbursementdecisions.
FROST & SULLIVAN PREDICTS GROWTH FOR THERAPEUTIC A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices. DRUG DEVELOPMENT & DELIVERY Drug Development & Delivery is a print and online content provider that presents the latest scientific methods in drug development for professionals in R&D, APIs, pre-formulation, formulation development and analytical methods. ABOUT US - DRUG DEVELOPMENT AND DELIVERY Drug Development & Delivery, including its exclusive Specialty Pharma section, is a print publication, conference organizer (Drug Development Networking Summit), and online content provider exclusively committed to advancing the applied science, technology, and related business practices of pharmaceutical and biological drug development, reporting on formulation design, bioavailability and ZIARCO PHARMA ACQUIRES PFIZER DRUGS, SECURES FINANCINGZIARCO PHARMA LTDDR MIKE YEADON PFIZER OPINIONDR YEADON PFIZERMICHAEL YEADON PFIZER Ziarco recently announced the closing of an initial $6-million tranche of Series A financing totalling $27 million. The round was led by Biotechnology Value Fund L.P, with participation byTOPICAL DEVELOPMENT
THE RISK CHALLENGE. Recent reviews have suggested that it takes the evaluation of 10,000 compounds to produce one approved medicine containing a new chemical entity (NCE), and the development time is typically between 10 and 14 years with costs in excess of $1 billion. 1,2 Nevertheless, the continual discovery of new potential target pathways, the re-purposing of drugs, and advantageous DARÉ BIOSCIENCE ANNOUNCES RECEIPT OF $1.5 MILLION UNDER A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices. INTAC TECHNOLOGY COMBATS MISUSE OF EXTENDED-RELEASE DRUGS During this year’s Annual Scientific Meeting of the American Pain Society (May 16-19, 2012, Honolulu, HI), Grunenthal presented results from clinical studies utilizing a product based on its INTAC formulation technology.INTAC is designed to be crush-resistant and toprotect
AKORN TO ACQUIRE VERSAPHARM FOR $440 MILLION Akorn, Inc. recently announced it has entered into a definitive agreement to acquire VPI Holdings Corp., the parent company of VersaPharm Incorporated, for $440 million in cash. WHITE PAPER: CONTROL OF BETA-GLUCANS & ENDOTOXIN IN HIGHBEST SOURCE OF BETA GLUCANSWHAT ARE BETA GLUCANSFOODS HIGH IN BETA GLUCANSBETA GLUCAN DOSAGE FOR CANCERBETA GLUCANS AND CANCERENDOTOXIN ENHANCEMENT Sucrose or saccharose is a disaccharide composed of glucose and fructose. Figure 1, depicts the chemical structure of sucrose. It has been used in protein stabilization particularly biotherapeutics since the past three decades.MARKET BRIEF
Akiko Fukui is a Neurology and Ophthalmology Healthcare Analyst at GlobalData in London.Prior to joining GlobalData, she was an Associate at PAREXEL Consulting, where she worked on consulting projects advising companies on market access, pricing, and reimbursementdecisions.
FROST & SULLIVAN PREDICTS GROWTH FOR THERAPEUTIC A Guide to Aseptic Manufacturing The aseptic manufacturing process is complex with each drug candidate's unique set of challenges. Speed to market is necessary, it’s crucial to follow a strong quality risk management, operate in a current cGMP environment with vigilant personnel practices.NANOMEDICINES
Susan Rosenbaum, JD, and Irwin Hollander, PhD, present a unique and novel solution to this greatest challenge in medicine for brain disease treatment, considered the “Holy Grail of Neuroscience,” with their breakthrough innovation.CLINICAL TRIALS
Karen Ooms, Msc, believes the consequences of this pandemic on ongoing clinical trials can be objectively assessed, and with the correct mitigation strategies put in place, study integrity can be preserved, optimizing use of the available resources for both patients andsponsors.
PATIENT SAFETY NARRATIVES INTRODUCTION. Patient safety narratives are a key element in clinical study reporting. We will look at current regulatory requirements regarding safety narratives, a proposed process for their development, and review and examine ways to simplify the reporting process. These procedures are aimed at reducing the burden of time and cost.. Patient safety narratives should be prepared for allSPECIAL FEATURE
Platform technologies are considered a valuable tool to improve efficiency and quality in drug product development. The basic idea is that a platform, in combination with a risk-based approach, is the most systematic method to leverage prior knowledge for a given newmolecule.
DRUG DEVELOPMENT
The prospect of a generation of new cell and gene therapies introduced in succession, many of them offering potentially curative efficacy, is obviously good news for many patients, but it also presents many significant considerations for the optimal strategies in drugdevelopment.
LYOPHILIZATION
John W. Burke, MS, says that success in lyophilization transfer and scale-up projects depends on a structured approach to information sharing between a pharma company and its CDMO partner, and should include extensive details about the APIs or bulk drug substance characteristics and planned development and clinical programs. BIO PLATFORMS ARCHIVES 6/3/2021 Corbus Pharmaceuticals Licenses Two Integrin Targeting mAbs Further Expanding Pipeline Into Cancer & Fibrotic Diseases. Corbus Pharmaceuticals Holdings, Inc. recently announced licensing deals for two new monoclonal antibodies (mAbs), CRB-601 and CRB-602, that target integrins to inhibit activation of. UNIQURE ANNOUNCES POSITIVE RECOMMENDATION TO ADVANCE PHASE uniQure N.V. recently announced the independent Data Safety Monitoring Board (DSMB) overseeing the Phase 1/2 clinical trial of AMT-130 for the treatment of Huntington’s disease has met and reviewed safety data for the fully enrolled first cohort of ten patients. ARTICLES - PAGE 250 OF 251 - DRUG DEVELOPMENT AND DELIVERY Corbus Pharmaceuticals Licenses Two Integrin Targeting mAbs Further Expanding Pipeline Into Cancer & Fibrotic Diseases. Corbus Pharmaceuticals Holdings, Inc. recently announced licensing deals for two new monoclonal antibodies (mAbs), CRB-601 and CRB-602, that target integrins to inhibit activation of. E-BOOK - FIVE DANGERS OF AN INSECURE API SUPPLY CHAIN Securing your API supply chain is key when managing the manufacture and delivery of your products, and a minor delay can derail a whole project. Learn what to* __Subscriptions
* __Current/Back Issues/eBooks * __Market News & Trends * __Executive Interviews__
Select a Section
__
* Analytical/Testing * Bioavailability & Solubility* Bio Platforms
* Drug Delivery
* Devices
* Excipients
* Formulation Development* Contract Services
SIGN UP FOR A
FREE SUBSCRIPTION
SPECIAL FEATURE – EXCIPIENTS: MATCHING INGREDIENTS TO MOLECULES IMPROVES FUNCTIONALITY Contributor Cindy Dubin speaks with several leading excipient manufacturers on how their excipient offerings are improving drug release, solubility, taste, physical characteristics, viscosity, and more for a range of molecules. Bioavailability & Solubility,Drug
Delivery,
Excipients, FeaturedArticles,
Formulation Development FORMULATION FORUM – RATIONAL DESIGN & DEVELOPMENT OF LIPID-FILLEDHARD CAPSULES
Jim Huang, PhD, says with ever increasing percentage of water-insoluble or low permeable new therapeutic entities among drug pipelines, lipid-based delivery systems, together with other main delivery technologies, have become an important tool for the development of those compounds.Drug Delivery,
Excipients, FeaturedArticles,
Formulation Development THERAPEUTIC PEPTIDES – CONTINUOUS MANUFACTURING OF PEPTIDES COULD SPEED UP DEVELOPMENT, REDUCE COSTS & IMPROVE QUALITY Jens Bukrinski, PhD, MSc, says the high-quality, high-process consistency between manufacturing runs and the in-line PAT analytics of the μLOT platform will enable unprecedented robustness of the manufacturing process, significantly retiring the risk of failure to supply due to non-scalability of the manufacturing process.Bio Platforms,
Featured Articles,
Formulation Development CANNABINOID THERAPY – NEURODIRECT EFFECTS(TM) CBD: NON-SYSTEMIC CANNABIDIOL FOR AUTISM SPECTRUM DISORDER Ronald Aung-Din, MD, says NeuroDirect Effects Technology CBD is particularly attractive in this population as it utilizes the body’s own ECS, and in utilizing a non-systemic therapeutic mechanism, there is less likelihood for long-term negative effects in developing brainsof children.
Drug Delivery,
Featured
Articles,
Formulation Development ORAL THIN FILMS – MISCONCEPTIONS, ADVANTAGES & LIMITATIONS ABOUT AN EMERGING DRUG DELIVERY SYSTEM Srinivasan Shanmugam, PhD, says due to advancements in the science behind OTF design, as well as the predicted increase in OTF market valuation, this drug delivery system is becoming increasingly popular. Bioavailability & Solubility,Drug
Delivery,
Featured Articles,
Formulation Development EXECUTIVE INTERVIEW – TEDOR PHARMA, INC.: A STRATEGY SHIFT TO SERVE BOTH GENERIC & BRANDED COMPANIES Doug Drysdale, President & CEO of Tedor Pharma, Inc., discusses the FLEXITAB technology, his strategy for managing a shift in business model, and how Tedor is addressing the needs of small- and medium-size companies developing innovative and repurposed small molecules for unmet medical needs.Contract Services,
Executive Interviews,Featured
Articles,
Formulation Development NATURAL LANGUAGE PROCESSING – HOW LIFE SCIENCES COMPANIES ARE LEVERAGING NLP FROM MOLECULE TO MARKET Jane Z. Reed, PhD, MA, discusses some of the challenges facing pharma researchers and executives; the benefit NLP can bring; and some specific customer-use cases (covering patent landscaping, gene disease associations, access to safety silos, and more).Contract Services,
Featured Articles,
Formulation Development DEVELOPMENT TIMELINES – DRUG DEVELOPMENT TIMES, WHAT IT TAKES –PART 3
Josef Bossart, PhD, analyzes the development and review times of the US FDA’s new drug approvals (NDA and BLA) for the period of 2010through 2018.
Featured Articles,
Formulation Development MARKET BRIEF – ALLERGIC RHINITIS IMMUNOTHERAPY: OPPORTUNITY ANALYSIS& FORECASTS TO 2028
Rose Joachim, PhD, indicates that while a wide variety of OTC therapies exist for the temporary abatement of AR symptoms, allergen-specific immunotherapy is the only truly disease-modifying therapy available for AR.Featured Articles,
Formulation Development STANDARD OPERATING PROCEDURES – HOW WRITING AN EFFECTIVE SOP CAN INFLUENCE COMPLIANCE & BUILD A BETTER ORGANIZATION Heidi Stuttz provides a common outline used in many companies and explores the essential elements of a well-written SOP that can be effective whether developing one from scratch or remediating a current system of SOPs that will foster success in every area of your company in which it is implemented.Contract Services,
Featured Articles
CLINICAL STUDY REPORTING – ASSESSING THE VALUE OF INTERIM ANALYSESIN CLINICAL TRIALS
Paul Stark, MS, ScD, reviews how and when an interim analysis would be valuable and how, with examples and outcomes, it can be applied in a clinical trial setting.Contract Services,
Featured Articles
GLOBAL REPORT – 2019 GLOBAL DRUG DELIVERY & FORMULATION REPORT: PART 2, NOTABLE DRUG DELIVERY & FORMULATION PRODUCT APPROVALS OF 2019 In part 2 of this 4-part series, PharmaCircle, in collaboration with Drug Development & Delivery, focuses on notable drug delivery and formulation product approvals. Devices, Drug Delivery,Featured
Articles,
Formulation DevelopmentVIEW MORE ARTICLES
EXCLUSIVE ONLINE CONTENT4/23/2020
HISTOGEN ANNOUNCES INVESTIGATIONAL DEVICE EXEMPTION APPLICATION Histogen Inc. recently announced it has submitted an Investigational Device Exemption (IDE) application with the US FDA for the initiationof a….
4/23/2020
MOGRIFY & SANGAMO ANNOUNCE COLLABORATION & EXCLUSIVE LICENSEAGREEMENT
Mogrify Ltd and Sangamo Therapeutics recently announced they have executed a collaboration and exclusive license agreement for Sangamoto develop….
4/23/2020
MUSTANG BIO RECEIVES ADVANCED THERAPY MEDICINAL PRODUCT CLASSIFICATION Mustang Bio, Inc. recently announced that the European Medicines Agency (EMA) has granted Advanced Therapy Medicinal Product (ATMP) classification to….4/23/2020
ESPERION ANNOUNCES DEVELOPMENT & COMMERCIALIZATION AGREEMENT WITH OTSUKA PHARMACEUTICAL Esperion recently announced it has entered into a collaboration agreement with Otsuka Pharmaceutical Co., Ltd. for the development and commercialization of….4/23/2020
Q BIOMED INITIATES RAPID DEVELOPMENT OF NOVEL COVID-19 THERAPEUTICS Q BioMed Inc. recently announced that together with its technology partner, Mannin Research, they are accelerating the rapid development of novel drugs for the treatment of life-threatening complications caused by COVID-19 and….4/16/2020
NEW COVID-19 CLINICAL TRIAL SUPPORTED BY OCTAPHARMA USA Octapharma USA is supporting a new investigator initiated clinical trial led by George Sakoulas, MD, of Sharp Memorial Hospital in San Diego, CA, focused on treating the most critical patients at the….4/15/2020
TFF PHARMACEUTICALS ANNOUNCES R&D COLLABORATION FOR A UNIVERSALINFLUENZA VACCINE
TFF Pharmaceuticals, Inc. recently announced that it has entered into a research collaboration with the University of Georgia’s Center for Vaccines and Immunology (CVI), along with….4/15/2020
HALIX ENTERS COLLABORATION FOR GMP MANUFACTURING OF A COVID-19 VACCINE HALIX B.V. has recently joined a consortium of partners under the guidance of the University of Oxford, to provide GMP manufacturing services supporting the large scale production of….4/15/2020
SAMSUNG BIOEPIS ANNOUNCES US LAUNCH OF ONCOLOGY BIOSIMILAR FOR EARLY & METASTATIC HER2-OVEREXPRESSING BREAST CANCER & METASTATIC GASTRICCANCER
Samsung Bioepis Co., Ltd. recently announced today that ONTRUZANT (trastuzumab-dttb), a biosimilar of the reference biologic medicine HERCEPTIN (trastuzumab) for the treatment of….4/9/2020
PRECISION BIOSCIENCES ANNOUNCES DOSING OF FIRST PATIENT IN PHASE 1/2ACLINICAL TRIAL
Precision BioSciences, Inc. recently announced the initiation of patient dosing in a Phase 1/2a clinical trial of its second off-the-shelf (allogeneic) chimeric…. VIEW MORE EXCLUSIVE ONLINE CONTENT MARKET NEWS & TRENDS4/23/2020
CATALYST BIOSCIENCES COMPLETES PHASE 2B TRIAL OF SUBCUTANEOUS FACTOR IX DALCINONACOG ALFA (DALCA)4/23/2020
INMUNE BIO ANNOUNCES INITIATION OF CLINICAL PROGRAM TO DETERMINE IF COMPANY’S PLATFORM MAY PREVENT COMPLICATIONS OF CYTOKINE STORMCAUSED BY COVID-19
4/23/2020
VAXART ANNOUNCES POSITIVE PRECLINICAL DATA FOR ITS ORAL COVID-19VACCINE PROGRAM
4/23/2020
ASCENDIA JOINS THE FIGHT AGAINST COVID-19; DEVELOPING FORMULATIONS4/23/2020
ARCUTIS ANNOUNCES ENROLLMENT OF FIRST PATIENT IN PHASE 1/2B STUDY4/20/2020
COVID-19: GATTEFOSSÉ TAKES MEASURES & CONTINUES TO BE OPERATIONAL4/15/2020
HOVIONE’S INHALER FOR HIGH-DOSE DELIVERY EARNS PRODUCT DESIGN AWARD4/15/2020
GUARDION HEALTH SCIENCES ANNOUNCES LAUNCH OF IMMUNO-SUPPORTIVE/ANTI-OXIDANT COMPLEX4/15/2020
BIOMARKERS HEAVILY USED AS DIAGNOSTIC TOOLS IN COVID-19 TRIALS4/15/2020
CELSION REPORTS SUFFICIENT EVENTS HAVE BEEN REACHED FOR THE SECOND INTERIM ANALYSIS OF THE PHASE 3 OPTIMA STUDY VIEW MORE MARKET NEWSVIDEO SPOTLIGHT __
COMPANY PROFILES
* AbbVie Contract Manufacturing * ABITEC Corporation * Adare Pharmaceuticals * Ajinomoto Bio-Pharma Services* Alcami
* ARL Bio Pharma
* Ascendia Pharmaceuticals* BASF Corporation
* Baxter BioPharma Solutions * BD Medical – Pharmaceutical Systems* BioDuro
* Bioiberica S.A.U
* Capsugel – Lonza * Captisol, a Ligand Company * Catalent Pharma Solutions * Credence MedSystems Inc.* Croda Health Care
* Datwyler Pharma Packaging USA Inc.* DDL
* Element Materials Technology * Eurofins BioPharma Product Testing * Excite Pharma Services * Foster Delivery Science * Frontage Laboratories, Inc.* Gattefosse
* Haselmeier, Inc.
* HERMES PHARMA
* ILC Dover
* Integral BioSystems LLC* JRS Pharma
* Lonza Pharma & Biotech * Lyophilization Technology, Inc. * Metrics Contract Services* MilliporeSigma
* Mitsubishi Gas Chemical Company, Inc.* Nemera
* Noble
* Pace Analytical Life Sciences* Pfanstiehl
* Pfizer CentreOne
* PharmaCircle
* Pii
* PMI Biopharma Solutions * Portal Instruments * Proveris Scientific* Quotient Sciences
* Recipharm
* SGS Life Sciences
* SGW Pharma Marketing* Singota Solutions
* Vetter Pharma International* Yourway
EBOOKS __
* Analytical Testing e-Book 2019 * Respiratory e-Book 2019INDUSTRY EVENTS
RDD (Respiratory Drug Delivery) April 26, 2020-Digital ConferenceINTERPHEX
July 15-17, 2020, New York CityCPhI North America
September 9-11, 2020, Philadelphia, PABIO2020 Digital
June 8-11, 2020, San Diego, CAPDA ANNUAL MEETING
July 20-22, 2020, Raleigh, NCMARKET SEGMENTS
* Analytical/Testing * Bioavailability & Solubility* Bio Platforms
* Drug Delivery
* Devices
* Excipients
* Formulation Development* Contract Services
CORPORATE
* SUBMISSION GUIDELINES* ABOUT US
* BACK ISSUES
* MEDIA PLANNER
* CONTACT
* FREE PRINT SUBSCRIPTION DRUG DEVELOPMENT & DELIVERY 219 Changebridge RoadMontville, NJ 07045
Tel: (973) 299-1200
Fax: (973) 299-7937
Copyright © 2018 Drug Development & Delivery. All Rights Reserved. Privacy Policy/ Terms and
Conditions
This material may not be published, broadcast, rewritten, or redistributed without permission . Web design and development by WalkerTek .Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0